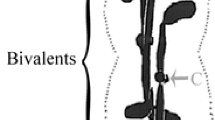
Summary
Variable numbers of bivalents and sex chromosomes do not attach to the spindle when prophase or early prometaphase cranefly spermatocytes (2n=8) are treated with cytochalasin D or latrunculin. The unattached bivalents lie in the cytoplasm or at the spindle pole, and they do not delay onset of autosomal anaphase; sometimes they disjoin at the same time as the attached bivalents, so they respond to the global signals that initiate anaphase. Unattached sex chromosomes do not delay autosomal anaphase, either. Of various interpretations of these data, we think the best explanation is that the checkpoint system responds to physical rather than chemical cues; we think that the spindle is a “tensegral” structure, that chromosomes need to interact with the spindle in order to be recognised by the anaphase-onset “checkpoint control”, and that the physical interaction of chromosomes with spindle acts as a signalling network. Cytochalasin D and latrunculin treatments delay onset of sex chromosome anaphase (which normally occurs about 15 min after autosomal anaphase) and cause altered patterns of sex-chromosome segregation.
Similar content being viewed by others
References
Adames KA, Forer A (1996) Evidence for poleward forces on chromosome arms during anaphase. Cell Motil Cytoskeleton 34: 13–25
Callaini G, Dallai R, Riparbelli MG (1997) Wolbachia-induced delay of paternal chromatin condensation does not prevent maternal chromosomes from entering anaphase in incompatible crosses of Drosophila stimulans. J Cell Sci 110: 271–280
Campbell MS, Gorbsky GJ (1995) Microinjection of mitotic cells with the 3F3/2 anti-phosphoepitope antibody delays the onset of anaphase. J Cell Biol 129: 1195–1204
Dietz R (1956) Die Spermatocytenteilungen der Tipuliden II: graphische Analyse der Chromosomenbewegung während der Prometaphase I im Leben. Chromosoma 8: 183–211
Dietz R (1969) Bau und Funktion des Spindelapparats. Naturwissenschaften 56: 237–248
— (1972) Die Assembly-Hypothese der Chromosomenbewegung und die Veränderungen der Spindellänge während der Anaphase I in Spermatocyten von Pales ferruginea (Tipulidae, Diptera). Chromosoma 38: 11–76
Forer A (1972) A method for making preparations of living crane fly spermatocytes for study with light microscopy followed by electron microscopy. Cytobiologie 6: 403–409
— (1980) Chromosome movements in the meiosis of insects, especially crane-fly spermatocytes. In: Blackman RL, Hewitt GM, Ashburner M (ed) Insect cytogenetics. Blackwell, Oxford, pp 85–95
— (1982) Crane fly spermatocytes and spermatids: a system for studying cytoskeletal components. Methods Cell Biol 25: 227–252
—, Koch C (1973) Influence of autosome movements and of sexchromosome movements on sex-chromosome segregation in crane fly spermatocytes. Chromosoma 40: 417–442
- Pickett-Heaps JD (1998) Cytochalasin D and latrunculin affect chromosome behaviour during meiosis in crane-fly spermatocytes. Chromosome Res (in press)
—, Spurck T, Pickett-Heaps JD (1997) Ultraviolet microbeam irradiations of spindle fibres in crane-fly spermatocytes and newt epithelial cells: resolution of previously conflicting observations. Protoplasma 197: 230–240
Gorbsky GJ (1995) Kinetochores, microtubules and the metaphase checkpoint. Trends Cell Biol 5: 143–148
—, Ricketts WA (1993) Differential expression of a phosphoepitope at the kinetochores of moving chromosomes. J Cell Biol 122: 1311–1321
Ilagan AI, Forer A (1997) Effects of ultraviolet-microbeam irradiation of kinetochores in crane-fly spermatocytes. Cell Motil Cytoskeleton 36: 266–275
Ingber DE (1993) Cellular tensegrity: defining new rules of biological design that govern the cytoskeleton. J Cell Sci 104: 613–627
Jang JK, Messina L, Erdman MB, Arbel T, Hawley RS (1995) Induction of metaphase arrest in Drosophila oocytes by chiasmabased kinetochore tension. Science 268: 1917–1919
Janicke MA, LaFountain JR Jr (1982) Chromosome segregation in crane-fly spermatocytes: cold treatment and cold recovery induce anaphase lag. Chromosoma 85: 619–631
— — (1984) Malorientation in half-bivalents at anaphase: analysis of autosomal laggards in untreated, cold-treated, and cold-recovering crane fly spermatocytes. J Cell Biol 98: 859–869
— — (1986) Bivalent orientation and behavior in crane-fly spermatocytes recovering from cold exposure. Cell Motil Cytoskeleton 6: 492–501
Ladrach KS, LaFountain JR Jr (1986) Malorientation and abnormal segregation of chromosomes during recovery from colcemid and nocodazole. Cell Motil Cytoskeleton 6: 419–427
LaFountain JR Jr (1985a) Chromosome segregation and spindle structure in crane fly spermatocytes following colcemid treatment. Chromosoma 91: 329–336
— (1985b) Malorientation in half-bivalents at anaphase in crane fly spermatocytes following colcemid treatment. Chromosoma 91: 337–346
—, Janicke MA, Balczon R, Rickards GK (1992) Cytochalasin induces abnormal anaphase in crane-fly spermatocytes and causes altered distribution of actin and centromeric antigens. Chromosoma 101: 425–441
Li X, Nicklas RB (1995) Mitotic forces control a cell-cycle checkpoint. Nature 373: 630–632
— — (1997) Tension-sensitive kinetochore phosphorylation and the chromosome distribution checkpoint in praying mantid spermatocytes. J Cell Sci 110: 537–545
Maniotis AJ, Chen CS, Ingber DE (1997) Demonstration of mechanical connections between integrins, cytoskeletal filaments, and nucleoplasm that stabilize nuclear structure. Proc Natl Acad Sci USA 94: 849–854
McIntosh JR (1991) Structural and mechanical control of mitotic progression. Cold Spring Harbor Symp Quant Biol 56: 613–619
McKim KS, Hawley RS (1995) Chromosomal control of meiotic cell division. Science 270: 1595–1601
Nicklas RB (1997) How cells get the right chromosomes. Science 275: 632–637
—, Ward SC, Gorbsky GJ (1995) Kinetochore chemistry is sensitive to tension and may link mitotic forces to a cell cycle checkpoint. J Cell Biol 130: 929–939
Pangilinan F, Spencer F (1996) Abnormal kinetochore structure activates the spindle assembly checkpoint in budding yeast. Mol Biol Cell 7: 1195–1208
Pickett-Heaps JD, Spurck T (1982) Studies on kinetochore function in mitosis I: the effects of colchicine and cytochalasin on mitosis in the diatom Hantzschia amphioxys. Eur J Cell Biol 28: 77–82
—, Forer A, Spurck T (1996) Rethinking anaphase: where “Pac-Man” fails and why a role for the spindle matrix is likely. Protoplasma 192: 1–10
— — — (1997) The traction fibre: toward a “tensegral” model of the spindle. Cell Motil Cytoskeleton 37: 1–6
Pluta AF, Mackay AM, Ainsztein AM, Goldberg IG, Earnshaw WC (1995) The centromere: hub of chromosomal activities. Science 270: 1591–1594
Rieder CL, Schulz A, Cole R, Sluder G (1994) Anaphase onset in vertebrate somatic cells is controlled by a checkpoint that monitors sister kinetochore attachment to the spindle. J Cell Biol 127: 1301–1310
—, Cole RW, Khodjakov A, Sluder G (1995) The checkpoint delay in anaphase in response to chromosome monoorientation is mediated by an inhibitory signal produced by unattached kinetochores. J Cell Biol 130: 941–948
—, Khodjakov A, Paliulis LV, Fortier TM, Cole RW, Sluder G (1997) Mitosis in vertebrate somatic cells with two spindles: implications for the metaphase/anaphase transition checkpoint and cleavage. Proc Natl Acad Sci USA 94: 5107–5112
Rudner AD, Murray AW (1996) The spindle assembly checkpoint. Curr Opin Cell Biol 8: 773–780
Sampson K, Pickett-Heaps JD, Forer A (1996) Cytochalasin D blocks chromosomal attachment to the spindle in the green alga Oedogonium. Protoplasma 192: 130–144
Sillers PJ, Forer A (1981) Autosomal spindle fibres influence subsequent sex-chromosome movements in crane-fly spermatocytes. J Cell Sci 49: 51–67
Sluder G, Miller FJ, Thompson EA, Wolf DE (1994) Feedback control of the metaphase-anaphase transition in sea urchin zygotes: role of maloriented chromosomes. J Cell Biol 126: 189–198
—, Thompson EA, Miller FJ, Hayes J, Rieder CL (1997) The checkpoint control for anaphase onset does not monitor excess numbers of spindle poles or bipolar spindle symmetry. J Cell Sci 110: 421–429
Spurck T, Forer A, Pickett-Heaps J (1997) Ultraviolet microbeam irradiations of epithelial and spermatoctye spindles suggest that forces act on the kinetochore fibre and are not generated by its disassembly. Cell Motil Cytoskeleton 36: 136–148
Wells WAE (1996) The spindle-assembly checkpoint: aiming for a perfect mitosis, every time. Trends Cell Biol 6: 228–234
—, Murray AW (1996) Aberrantly segregating centromeres activate the spindle assembly checkpoint in budding yeast. J Cell Biol 133: 75–84
Wilson PJ, Forer A, Leggiadro C (1994) Evidence that kinetochore microtubules in crane-fly spermatocytes disassemble during anaphase primarily at the poleward end. J Cell Sci 107: 3015–3027
Wrench GA, Snyder JA (1997) Cytochalasin J treatment significantly alters mitotic spindle organization and kinetochore structure in PtK1 cells. Cell Motil Cytoskeleton 36: 112–124
Yen TJ, Schaar BT (1996) Kinetochore function: molecular motors, switches and gates. Curr Opin Cell Biol 8: 381–388
Zhang D, Nicklas RB (1996) “Anaphase” and cytokinesis in the absence of chromosomes. Nature 382: 466–468
Zirkle RE (1957) Partial-cell irradiation. Adv Biol Med Phys 5: 103–146
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Forer, A., Pickett-Heaps, J.D. Checkpoint control in crane-fly spermatocytes: unattached chromosomes induced by cytochalasin D or latrunculin treatment do not prevent or delay the start of anaphase. Protoplasma 203, 100–111 (1998). https://doi.org/10.1007/BF01280592
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/BF01280592