Abstract
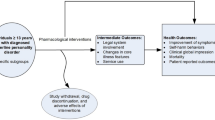
In this double-blind, randomized, placebo-controlled, prospective study, the clinical efficacy and tolerability of oral Hypericum extract STW 3-VI (Laif®) 900 mg once daily was compared with that of placebo. A total of 140 outpatients (94 women; 46 men) with moderate depressive disorders and a 17-item Hamilton Depression Scale (HAMD-17) score of 20 to 24 were enrolled in this study. Following a single-blind placebo run-in period of 7 days, the patients were randomized to Hypericum extract 900 mg or placebo for the 6-week treatment period. Nineteen patients have been excluded from the per protocol collective because of violations of protocol regarding the scheduling of study visits and intake of study medication. The primary endpoint for treatment efficacy was the change in total HAMD-17 score at the end of the 6-week treatment period. The HAMD-17 total score decreased significantly from baseline by approximately 11.1 ±4.5 points (from 22.8±1.1 to 11.8±4.4) in the Hypericum group and by approximately 3.4±3.9 points (from 22.6±1.2 to 19.2±3.8) in the placebo group (P<.001). Comparable group differences in favor of Hypericum were revealed by an additional responder analysis, the von Zerssen’s Adjective Mood Scale, the Clinical Global Impressions scale, and a global efficacy assessment. Tolerability was very good in both groups; neither serious adverse events nor clinically relevant changes in safety parameters were observed, and only 2 cases demonstrated a possible connection between an adverse event and the study medication. The final safety assessment showed no differences between the Hypericum extract and placebo groups. The study provided evidence that Hypericum extract STW 3-VI in a once-daily dosing regimen may be an effective and well-tolerated option for patients with moderate depressive disorders.
Similar content being viewed by others
References
Judd L. Mood disorders in the general population represent an important and worldwide public health problem.Int Clin Psychopharmacol. 1995;10(suppl 4):5–10.
Bilia AK, Gallori S, Vincieri FF. St. John’s wort and depression: efficacy, safety and tolerability—an update.Life Sci. 2002;70:3077–3096.
Laakmann G, Schüle C, Baghai T, Kieser M. St. John’s wort in mild to moderate depression: the relevance of hyperforin for the clinical efficacy.Pharmacopsychiat. 1998;31(suppl):54–59.
Kalb R, Trautmann-Sponsel RD, Kieser M. Efficacy and tolerability of hypericum extract WS 5572 versus placebo in mildly to moderately depressed patients.Pharmacopsyciat. 2001;34:96–103.
Laakmann G, Jahn G, Schüle C. Hypericum-perforatum-Extrakt bei der Behandlung leichter bis mittelschwerer Depressionen: Klinische und pharmakologische Aspekte.Nervenarzt. 2002;73: 600–612.
Hippius H. St. John’s wort (Hypericum perforatum): a herbal antdipressant.Curr Med Res Opin. 1998;14:171–184.
Müller WE, Rolli M, Schäfer C, Hafner U. Effects of Hypericum Extract (LI 160) in Biochemical Models of Antidepressant Activity.Pharmacopsychiat. 1997;30(suppl):102–107.
Uebelhack R, Franke L. In vitro effects of St. John’s Wort Extract and hyperforin on 5HT uptake and efflux in human blood platelets.Pharmacopsychiatry. 2001;34(suppl 1):S146-S147.
Schrader E, Meier B, Brettström A. Hypericum treatment of mild-moderate depression in a placebo controlled study: a prospective, double-blind, randomized, placebo-controlled, multicentre study.Human Psychopharmacol. 1998;13:163–169.
Hänsgen KD, Vesper J. Antidepressive Wirksamkeit eines hochdosierten Hypericum-Extraktes.Münch Med Wschr. 1996;138:29–33.
Vorbach EU, Hübner WD, Arnold KH. Wirksamkeit und Verträglichkeit des Hypericum-Extraktes LI 160 im Vergleich mit Imipramin.Nervenheilkunde. 1993;12:290–296.
Philipp M, Kohen R Hiller KO. Hypericum extract versus imipramine or placebo in patients with moderate depression: randomized multicentre study of treatment for eight weeks.BMJ. 1999;319:1534–1538.
Linde K, Ramirez G, Mulrow D, Pauls A, Weidenhammer W, Melchart D. St. John’s wort for depression. An overview and meta-analysis of randomized clinical trials.BMJ. 1996;313:253–258.
Schulz V. Clinical trials with Hypericum extracts in patients with depression:results, compar isons, conclusions for therapy with antidepressant drugs.Phytomedicine. 2002;9:468–474.
Volz HP. Controlled clinical trials of hypericum extracts in depressed patients. An overview.Pharmacopsychiat. 1997;30(suppl 2):72–76.
Gaster B, Holroyd J. St. John’s Wort for Depression. A Systematic Review.Arch Intern Med. 2000; 160:152–156.
Brockmöller J, Reum T, Bauer S, Kerb R, Hübner WH, Roots J. Hypericin and pseudohypericin: pharmacokinetics and effects on photosensitivity in humans.Pharmacopsychiat. 1997;30(suppl): 94–101.
Hänsgen KD, Vesper J, Ploch M. Multizentrische Doppelblindstudie zur antidepressiven Wirksamkeit des Hypericum-Extraktes LI 160.Nervenheilkunde. 1993;12:285–289.
Sommer H. Besserung psychovegetativer Beschwerden durch Hypericum im Rahmen einer multizentrischen Doppelblindstudie.Nervenheilkunde. 1991;10:308–310.
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Uebelhack, R., Gruenwald, J., Graubaum, HJ. et al. Efficacy and tolerability of Hypericum extract STW 3-VI in patients with moderate depression: A double-blind, randomized, placebo-controlled clinical trial. Adv Therapy 21, 265–275 (2004). https://doi.org/10.1007/BF02850158
Issue Date:
DOI: https://doi.org/10.1007/BF02850158