Abstract
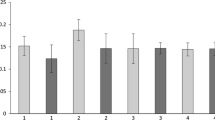
The various uses of copper or copper compounds in industrial, biocidal or pesticidal products lead to inputs of the metal into aquatic environments. To assess effects of copper ions on non-target organisms, the freshwater snail Theodoxus fluviatilis was used as test organism for a three-week laboratory experiment. Snails were exposed to four copper concentrations ranging from 4 to 39 µg Cu2+/L, and besides mortality, several sublethal parameters were evaluated. Concerning survival, an aqueous copper concentration of 6 µg/L was determined as NOEC, and 16 (± 0.1) µg/L as LC50. Negative sublethal effects on reproduction, activity and pathological modifications in the snails were detected in the treatment with an aqueous copper concentration of 15 µg/L. Our results using T. fluviatilis as a mere grazer exclusively feeding on biofilms contribute to findings that field relevant copper concentrations have a significant effect on non-target organisms in aquatic environments.


Similar content being viewed by others
References
ATSDR (Agency for Toxic Substances and Disease Registry) (2004) Toxicological profile for copper. Department for Health and Human Services, Washington, DC
Bhaskar BV, Bhosle NB (2006) Bacterial extracellular polymeric substance (EPS): a carrier of heavy metals in the marine food chain. Environ Int 32(2):191–198
Brix KV, Esbaugh AJ, Grosell M (2011) The toxicity and physiological effects of copper on the freshwater pulmonate snail, Lymnaea stagnalis. Comp Biochem A Physiol 154:261–267
Cuppett JD, Duncan SE, Dietrich AM (2006) Evaluation of copper speciation and water quality factors that affect aqueous copper tasting response. Chem Senses 31:689–697
Das S, Khangarot BS (2011) Bioaccumulation of copper and toxic effects on feeding, growth, fecundity and development of pond snail Lymnaea luteola L. J Hazard Mater 185:295–305
European Copper Institute (ECI) (2008) Voluntary risk assessment of copper, copper (II) sulphate pentahydrate, copper (I) oxide, copper (II) oxide, dicopper chloride trihydroxide. ECHA, Helsinki
Flemming CA, Trevors JT (1989) Copper toxicity and chemistry in the environment: a review. Water Air Soil Pollut 44:143–158
Gergs R, Rothhaupt KO (2008) Feeding rates, assimilation efficiencies and growth of two amphipod species on biodeposited material from zebra mussels. Freshw Biol 53:2494–2503
Hansen BH, Garmo OA, Olsvik PA, Andersen RA (2007) Gill metal binding and stress gene transcription in brown trout (Salmo trutta) exposed to metal environments: the effect of pre-exposure in natural populations. Environ Toxicol Chem 26:944–953
Kahle M, Nöh I (2009) Biozide in Gewässern. Eintragspfade und Informationen zur Belastungssituation und deren Auswirkungen. Texte 09/2009. Federal Environment Agency, Dessau-Roßlau
Kiaune L, Singhasemanon N (2011) Pesticidal copper (I) oxide: environmental fate and aquatic toxicity. Rev Environ Contam Toxicol 213:1–26
Leland HV, Carter JL (1984) Effects of copper on species composition of periphyton in a Sierra Nevada, California, stream. Freshwater Biol 14:281–296
Mehta R, Templeton DM, O’Brian PJ (2006) Mitochondrial involvement in genetically determined transition metal toxicity II. Copper toxicity Chem 163:77–85
Morin S, Duong TT, Herlory O, Feurtet-Mazel A, Coste M (2008) Cadmium toxicity and bioaccumulation in freshwater biofilms. Arch Environ Contam Toxicol 54:173–186
Neumann D (1961) Ernährungsbiologie einer rhipidoglossen Kiemenschnecke. Hydrobiologia 17:133–151
Piola RF, Dafforn KA, Johnston EL (2009) The influence of antifouling practices on marine invasions. Biofouling 25:633–644
R Development Core Team (2006) R: a language and environment for statistical computing. https://www.R-project.com. R Foundation for Statistical Computing, Vienna, Austria
Schaller J, Dharamshi J, Dudel EG (2011) Enhanced metal and metalloid concentrations in the gut system comparing to remaining tissues of Gammarus pulex L. Chemosphere 83:627–631
Skoog G (1978) Influence of natural food items on growth and egg production in brackish water populations of Lymnea peregra and Theodoxus fluviatilis (Mollusca). Oikos 31:340–348
Watermann B, Daehne D, Fürle C, Thomsen A (2017) Antifouling biocides in German coastal & inland waters – How reliable are exposure prognoses of EU-scenario models for marinas? FKZ 3711 67 432. Texte 88/2017. English version. Federal Environment Agency, Dessau-Roßlau
Watermann B, Thomsen A, Kolodzey H, Daehne B, Meemken M, Pijanowska U, Liebezeit G (2008) Histopathological lesions of molluscs in the harbour of Norderney, Lower Saxony, North Sea (Germany). Helgol Mar Res 62:167–175
Zettler ML (2008) Zur Taxonomie und Verbreitung der Gattung Theodoxus Montfort, 1810 in Deutschland. Darstellung historischer und rezenter Daten einschließlich einer Bibliografie. Mollusca 26:13–72
Acknowledgement
We thank Ina Janthur and Bonny Alscher for conducting analyses of nutrients and major ions and Jennifer Bartz for those of copper. The LimnoMar Laboratory for Freshwater and Marine Research Hamburg performed the histopathological analysis, here we are grateful to Anja Thomsen for preparation of snails. During this study Karsten Grabow at University of Education Karlsruhe supported the experimental design with expert knowledge regarding freshwater gastropods.
Author information
Authors and Affiliations
Corresponding author
Additional information
Publisher’s Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Electronic supplementary material
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Rothmeier, L.M., Martens, A., Watermann, B. et al. Effects of Copper Ions on Non-target Species: A Case Study Using the Grazer Theodoxus fluviatilis (Gastropoda: Neritidae). Bull Environ Contam Toxicol 105, 62–66 (2020). https://doi.org/10.1007/s00128-020-02913-x
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00128-020-02913-x