Abstract
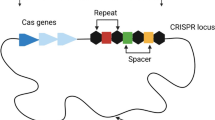
A subculture of S.epidermidis strain ATCC35984 that is amenable to genetically manipulate was occasionally found in our laboratory. This mutant exhibited susceptibility to methicillin in contrast to its parent strain. To unveil the underlying mechanism, whole-genome sequencing of the mutant was performed. A comparative analysis revealed that a large DNA fragment encompassing the CRISPR–Cas system, type I R-M system and the SCCmec element was deleted from the mutant. The large chromosomal deletion associated with CRISPR–Cas system was also observed to occur spontaneously in S. epidermidis in another independent laboratory, or artificially induced by introducing engineering crRNAs in other bacterial species. These findings imply the CRISPR–Cas systems can affect bacterial genome remodeling through deletion of the integrated MGEs (mobile genetic elements). Further bioinformatics analysis identified a higher carriage rate of SCCmec element in the S. epidermidis strains harboring the CRISPR–Cas system. MLST typing and phylogenetic analysis of those CRIPSR–Cas-positive S. epidermidis strains revealed multiple origins. In addition, distinct types of SCCmec carried in those strains suggested that acquisition of this MGE originated from multiple independent recombination events. Intriguingly, CRISPR–Cas systems are found to be always located in the vicinity of orfX gene among staphylococci. Allelic analysis of CRISPR loci flanking cas genes disclosed that the loci distal to the orfX gene are considerably stable and conserved, which probably serve as recombination hotspot between CRISPR–Cas system and phage or plasmid. Therefore, the findings generally support the notion that incomplete immune protection of CRISPR–Cas system can promote dissemination of its neighboring SCCmec element.



source of these strains are listed as follows: BAL bronchoalveolar lavage, CSF cerebrospinal fluid, HM human milk, AC human alar crease, HC housing complex, IB indoor biome. Asterisks indicate SCCmec-negative strains


Similar content being viewed by others
References
Angelopoulou A, Warda AK, O’Connor PM, Stockdale SR, Shkoporov AN, Field D, Draper LA, Stanton C, Hill C, Ross RP (2020) Diverse bacteriocins produced by strains from the human milk microbiota. Front Microbiol 11:788. https://doi.org/10.3389/fmicb.2020.00788
Argemi X, Nanoukon C, Affolabi D, Keller D, Hansmann Y, Riegel P, Baba-Moussa L, Prevost G (2018) Comparative genomics and identification of an enterotoxin-bearing pathogenicity island, SEPI-1/SECI-1, in Staphylococcus epidermidis pathogenic strains. Toxins (basel). https://doi.org/10.3390/toxins10030093
Barrangou R, Horvath P (2017) A decade of discovery: CRISPR functions and applications. Nat Microbiol 2:17092. https://doi.org/10.1038/nmicrobiol.2017.92
Barrangou R, Fremaux C, Deveau H, Richards M, Boyaval P, Moineau S, Romero DA, Horvath P (2007) CRISPR provides acquired resistance against viruses in prokaryotes. Science 315:1709–1712. https://doi.org/10.1126/science.1138140
Bloemendaal AL, Brouwer EC, Fluit AC (2010) Methicillin resistance transfer from Staphylocccus epidermidis to methicillin-susceptible Staphylococcus aureus in a patient during antibiotic therapy. PLoS ONE 5:e11841. https://doi.org/10.1371/journal.pone.0011841
Buttner H, Mack D, Rohde H (2015) Structural basis of Staphylococcus epidermidis biofilm formation: mechanisms and molecular interactions. Front Cell Infect Microbiol 5:14. https://doi.org/10.3389/fcimb.2015.00014
Cabrera-Contreras R, Santamaria RI, Bustos P, Martinez-Flores I, Melendez-Herrada E, Morelos-Ramirez R, Barbosa-Amezcua M, Gonzalez-Covarrubias V, Silva-Herzog E, Soberon X, Gonzalez V (2019) Genomic diversity of prevalent Staphylococcus epidermidis multidrug-resistant strains isolated from a Children’s Hospital in Mexico City in an eight-years survey. PeerJ 7:e8068. https://doi.org/10.7717/peerj.8068
Cao L, Gao CH, Zhu J, Zhao L, Wu Q, Li M, Sun B (2016) Identification and functional study of type III-A CRISPR–Cas systems in clinical isolates of Staphylococcus aureus. Int J Med Microbiol 306:686–696. https://doi.org/10.1016/j.ijmm.2016.08.005
Chlebowicz MA, Maslanova I, Kuntova L, Grundmann H, Pantucek R, Doskar J, van Dijl JM, Buist G (2014) The Staphylococcal Cassette Chromosome mec type V from Staphylococcus aureus ST398 is packaged into bacteriophage capsids. Int J Med Microbiol 304:764–774. https://doi.org/10.1016/j.ijmm.2014.05.010
Conlan S, Mijares LA, Program NCS, Becker J, Blakesley RW, Bouffard GG, Brooks S, Coleman H, Gupta J, Gurson N, Park M, Schmidt B, Thomas PJ, Otto M, Kong HH, Murray PR, Segre JA (2012) Staphylococcus epidermidis pan-genome sequence analysis reveals diversity of skin commensal and hospital infection-associated isolates. Genome Biol 13:R64. https://doi.org/10.1186/gb-2012-13-7-r64
Dy RL, Pitman AR, Fineran PC (2013) Chromosomal targeting by CRISPR–Cas systems can contribute to genome plasticity in bacteria. Mob Genet Elements 3:e26831. https://doi.org/10.4161/mge.26831
Fluit AC, Carpaij N, Majoor EA, Bonten MJ, Willems RJ (2013) Shared reservoir of ccrB gene sequences between coagulase-negative staphylococci and methicillin-resistant Staphylococcus aureus. J Antimicrob Chemother 68:1707–1713. https://doi.org/10.1093/jac/dkt121
Garcia-Gutierrez E, Walsh CJ, Sayavedra L, Diaz-Calvo T, Thapa D, Ruas-Madiedo P, Mayer MJ, Cotter PD, Narbad A (2020) Genotypic and phenotypic characterization of fecal Staphylococcus epidermidis isolates suggests plasticity to adapt to different human body sites. Front Microbiol 11:688. https://doi.org/10.3389/fmicb.2020.00688
Gill SR, Fouts DE, Archer GL, Mongodin EF, Deboy RT, Ravel J, Paulsen IT, Kolonay JF, Brinkac L, Beanan M, Dodson RJ, Daugherty SC, Madupu R, Angiuoli SV, Durkin AS, Haft DH, Vamathevan J, Khouri H, Utterback T, Lee C, Dimitrov G, Jiang L, Qin H, Weidman J, Tran K, Kang K, Hance IR, Nelson KE, Fraser CM (2005) Insights on evolution of virulence and resistance from the complete genome analysis of an early methicillin-resistant Staphylococcus aureus strain and a biofilm-producing methicillin-resistant Staphylococcus epidermidis strain. J Bacteriol 187:2426–2438. https://doi.org/10.1128/JB.187.7.2426-2438.2005
Gotz F (2002) Staphylococcus and biofilms. Mol Microbiol 43:1367–1378. https://doi.org/10.1046/j.1365-2958.2002.02827.x
Guan J, Wang W, Sun B (2017) Chromosomal targeting by the type III-A CRISPR-Cas system can reshape genomes in Staphylococcus aureus. mSphere. https://doi.org/10.1128/mSphere.00403-17
Hatoum-Aslan A, Samai P, Maniv I, Jiang W, Marraffini LA (2013) A ruler protein in a complex for antiviral defense determines the length of small interfering CRISPR RNAs. J Biol Chem 288:27888–27897. https://doi.org/10.1074/jbc.M113.499244
Hatoum-Aslan A, Maniv I, Samai P, Marraffini LA (2014) Genetic characterization of antiplasmid immunity through a type III-A CRISPR–Cas system. J Bacteriol 196:310–317. https://doi.org/10.1128/JB.01130-13
Hille F, Charpentier E (2016) CRISPR–Cas: biology, mechanisms and relevance. Philos Trans R Soc Lond B Biol Sci. https://doi.org/10.1098/rstb.2015.0496
Hille F, Richter H, Wong SP, Bratovic M, Ressel S, Charpentier E (2018) The biology of CRISPR–Cas: backward and forward. Cell 172:1239–1259. https://doi.org/10.1016/j.cell.2017.11.032
Horvath P, Barrangou R (2010) CRISPR/Cas, the immune system of bacteria and archaea. Science 327:167–170. https://doi.org/10.1126/science.1179555
Jia N, Mo CY, Wang C, Eng ET, Marraffini LA, Patel DJ (2019) Type III-A CRISPR– and autoimmunity. Mol Cell 73:264-277 e265. https://doi.org/10.1016/j.molcel.2018.11.007
Jiang W, Samai P, Marraffini LA (2016) Degradation of phage transcripts by CRISPR-associated RNases enables type III CRISPR–Cas immunity. Cell 164:710–721. https://doi.org/10.1016/j.cell.2015.12.053
Kirstahler P, Bjerrum SS, Friis-Moller A, la Cour M, Aarestrup FM, Westh H, Pamp SJ (2018) Genomics-based identification of microorganisms in human ocular body fluid. Sci Rep 8:4126. https://doi.org/10.1038/s41598-018-22416-4
Lee AS, de Lencastre H, Garau J, Kluytmans J, Malhotra-Kumar S, Peschel A, Harbarth S (2018) Methicillin-resistant Staphylococcus aureus. Nat Rev Dis Primers 4:18033. https://doi.org/10.1038/nrdp.2018.33
Lee JYH, Carter GP, Pidot SJ, Guerillot R, Seemann T, Goncalves da Silva A, Foster TJ, Howden BP, Stinear TP, Monk IR (2019) Mining the methylome reveals extensive diversity in Staphylococcus epidermidis restriction modification. Mbio. https://doi.org/10.1128/mBio.02451-19
Li Q, Xie X, Yin K, Tang Y, Zhou X, Chen Y, Xia J, Hu Y, Ingmer H, Li Y, Jiao X (2016) Characterization of CRISPR–Cas system in clinical Staphylococcus epidermidis strains revealed its potential association with bacterial infection sites. Microbiol Res 193:103–110. https://doi.org/10.1016/j.micres.2016.09.003
Lin L, He X, Zhao T, Gu L, Liu Y, Liu X, Liu H, Yang F, Tu M, Tang L, Ge X, Liu C, Zhao J, Song Z, Qu J, Gu F (2018) Engineering the direct repeat sequence of crRNA for optimization of FnCpf1-mediated genome editing in human cells. Mol Ther 26:2650–2657. https://doi.org/10.1016/j.ymthe.2018.08.021
Makarova KS, Wolf YI, Alkhnbashi OS, Costa F, Shah SA, Saunders SJ, Barrangou R, Brouns SJ, Charpentier E, Haft DH, Horvath P, Moineau S, Mojica FJ, Terns RM, Terns MP, White MF, Yakunin AF, Garrett RA, van der Oost J, Backofen R, Koonin EV (2015) An updated evolutionary classification of CRISPR–Cas systems. Nat Rev Microbiol 13:722–736. https://doi.org/10.1038/nrmicro3569
Makarova KS, Wolf YI, Iranzo J, Shmakov SA, Alkhnbashi OS, Brouns SJJ, Charpentier E, Cheng D, Haft DH, Horvath P, Moineau S, Mojica FJM, Scott D, Shah SA, Siksnys V, Terns MP, Venclovas C, White MF, Yakunin AF, Yan W, Zhang F, Garrett RA, Backofen R, van der Oost J, Barrangou R, Koonin EV (2020) Evolutionary classification of CRISPR–Cas systems: a burst of class 2 and derived variants. Nat Rev Microbiol 18:67–83. https://doi.org/10.1038/s41579-019-0299-x
Marraffini LA, Sontheimer EJ (2008) CRISPR interference limits horizontal gene transfer in staphylococci by targeting DNA. Science 322:1843–1845. https://doi.org/10.1126/science.1165771
McDonald ND, Regmi A, Morreale DP, Borowski JD, Boyd EF (2019) CRISPR–Cas systems are present predominantly on mobile genetic elements in Vibrio species. BMC Genomics 20:105. https://doi.org/10.1186/s12864-019-5439-1
Monk IR, Foster TJ (2012) Genetic manipulation of Staphylococci-breaking through the barrier. Front Cell Infect Microbiol 2:49. https://doi.org/10.3389/fcimb.2012.00049
Nobrega FL, Walinga H, Dutilh BE, Brouns SJJ (2020) Prophages are associated with extensive CRISPR–Cas auto-immunity. Nucleic Acids Res 48:12074–12084. https://doi.org/10.1093/nar/gkaa1071
Nunez JK, Kranzusch PJ, Noeske J, Wright AV, Davies CW, Doudna JA (2014) Cas1-Cas2 complex formation mediates spacer acquisition during CRISPR–Cas adaptive immunity. Nat Struct Mol Biol 21:528–534. https://doi.org/10.1038/nsmb.2820
Nussenzweig PM, Marraffini LA (2020) Molecular mechanisms of CRISPR–Cas immunity in bacteria. Annu Rev Genet 54:93–120. https://doi.org/10.1146/annurev-genet-022120-112523
Otto M (2009) Staphylococcus epidermidis–the “accidental” pathogen. Nat Rev Microbiol 7:555–567. https://doi.org/10.1038/nrmicro2182
Otto M (2013) Coagulase-negative staphylococci as reservoirs of genes facilitating MRSA infection: Staphylococcal commensal species such as Staphylococcus epidermidis are being recognized as important sources of genes promoting MRSA colonization and virulence. BioEssays 35:4–11. https://doi.org/10.1002/bies.201200112
Peacock SJ, Paterson GK (2015) Mechanisms of methicillin resistance in Staphylococcus aureus. Annu Rev Biochem 84:577–601. https://doi.org/10.1146/annurev-biochem-060614-034516
Ray MD, Boundy S, Archer GL (2016) Transfer of the methicillin resistance genomic island among staphylococci by conjugation. Mol Microbiol 100:675–685. https://doi.org/10.1111/mmi.13340
Roach DJ, Burton JN, Lee C, Stackhouse B, Butler-Wu SM, Cookson BT, Shendure J, Salipante SJ (2015) A year of infection in the intensive care unit: prospective whole genome sequencing of bacterial clinical isolates reveals cryptic transmissions and novel microbiota. PLoS Genet 11:e1005413. https://doi.org/10.1371/journal.pgen.1005413
Rossi CC, Souza-Silva T, Araujo-Alves AV, Giambiagi-deMarval M (2017) CRISPR–Cas systems features and the gene-reservoir role of coagulase-negative staphylococci. Front Microbiol 8:1545. https://doi.org/10.3389/fmicb.2017.01545
Scharn CR, Tenover FC, Goering RV (2013) Transduction of staphylococcal cassette chromosome mec elements between strains of Staphylococcus aureus. Antimicrob Agents Chemother 57:5233–5238. https://doi.org/10.1128/AAC.01058-13
Selle K, Klaenhammer TR, Barrangou R (2015) CRISPR-based screening of genomic island excision events in bacteria. Proc Natl Acad Sci U S A 112:8076–8081. https://doi.org/10.1073/pnas.1508525112
Sorek R, Kunin V, Hugenholtz P (2008) CRISPR–a widespread system that provides acquired resistance against phages in bacteria and archaea. Nat Rev Microbiol 6:181–186. https://doi.org/10.1038/nrmicro1793
Turner NA, Sharma-Kuinkel BK, Maskarinec SA, Eichenberger EM, Shah PP, Carugati M, Holland TL, Fowler VG Jr (2019) Methicillin-resistant Staphylococcus aureus: an overview of basic and clinical research. Nat Rev Microbiol 17:203–218. https://doi.org/10.1038/s41579-018-0147-4
Varble A, Meaden S, Barrangou R, Westra ER, Marraffini LA (2019) Recombination between phages and CRISPR–cas loci facilitates horizontal gene transfer in staphylococci. Nat Microbiol 4:956–963. https://doi.org/10.1038/s41564-019-0400-2
Vercoe RB, Chang JT, Dy RL, Taylor C, Gristwood T, Clulow JS, Richter C, Przybilski R, Pitman AR, Fineran PC (2013) Cytotoxic chromosomal targeting by CRISPR/Cas systems can reshape bacterial genomes and expel or remodel pathogenicity islands. PLoS Genet 9:e1003454. https://doi.org/10.1371/journal.pgen.1003454
Watson BNJ, Staals RHJ, Fineran PC (2018) CRISPR–Cas-mediated phage resistance enhances horizontal gene transfer by transduction. Mbio. https://doi.org/10.1128/mBio.02406-17
Wiedenheft B, Sternberg SH, Doudna JA (2012) RNA-guided genetic silencing systems in bacteria and archaea. Nature 482:331–338. https://doi.org/10.1038/nature10886
Wielders CL, Vriens MR, Brisse S, de Graaf-Miltenburg LA, Troelstra A, Fleer A, Schmitz FJ, Verhoef J, Fluit AC (2001) In-vivo transfer of mecA DNA to Staphylococcus aureus [corrected]. Lancet 357:1674–1675. https://doi.org/10.1016/s0140-6736(00)04832-7
Wimmer F, Beisel CL (2019) CRISPR–Cas systems and the paradox of self-targeting spacers. Front Microbiol 10:3078. https://doi.org/10.3389/fmicb.2019.03078
Zhu T, Zhao Y, Wu Y, Qu D (2017) The Staphylococcus epidermidis gdpS regulates biofilm formation independently of its protein-coding function. Microb Pathog 105:264–271. https://doi.org/10.1016/j.micpath.2017.02.045
Acknowledgements
We thank Professor Ting Xue from Anhui Agriculture University for kindly providing the original S. epidermidis ATCC 35984 strain
Funding
This work was funded by the National Natural Science Foundation of China (Grant No.81802071), the Support Program for University Outstanding Youth Talent of Anhui Province (gxyq2019043), the Key Program of Educational Commission of Anhui Province (KJ2020A0602), Open Research Fund Program of Key Laboratory of Medical Molecular Virology (MOE/NHC), Fudan University (FDMV-2020005).
Author information
Authors and Affiliations
Contributions
TZ and YZ conceived and designed the study. TZ performed experiments described in this study. TZ and YZ wrote the draft and revised it. All authors approved the final version.
Corresponding author
Ethics declarations
Conflict of interest
The authors have no conflict of interest.
Additional information
Communicated by Erko Stackebrandt.
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Below is the link to the electronic supplementary material.
203_2021_2595_MOESM1_ESM.tif
Supplementary file 1: Table S1 The location of orfX and cas genes in chromosome of S.epidermidis with large enough scaffold or contig (TIF 6953 KB)
203_2021_2595_MOESM2_ESM.docx
Supplementary file 2: Figure S1 Schematic diagram showing the genetic organization of CRISPR–Cas system and SCCmec element in S .epidermidis strains FDAARGOS 153 (A) and BPH0723 (B) with complete genome sequences. The Type IIIA CRISPR–Cas system, Type I R-M system and type IV SCCmec element are denoted in yellow, blue and red, respectively (DOCX 15 KB)
203_2021_2595_MOESM3_ESM.tif
Supplementary file 3: Figure S2 Multiple genome alignment of CRISPR–Cas- SCCmec-negative staphylococci showing the relative location of CRISPR–Cas system and orfX gene (TIF 14570 KB)
Rights and permissions
About this article
Cite this article
Zhu, T., Zhao, Y. Correlation between type IIIA CRISPR–Cas system and SCCmec in Staphylococcus epidermidis. Arch Microbiol 203, 6275–6286 (2021). https://doi.org/10.1007/s00203-021-02595-x
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00203-021-02595-x