Abstract
Rationale
Tramadol, an atypical μ-opioid receptor agonist, as a psychoactive drug, is frequently abused by human beings. Understanding the neurobiological mechanisms of drug-associated learning and memory formation may help prevent drug addiction and relapse. Previous study revealed that dorsal hippocampus (CA1) plays a crucial role in the retrieval of tramadol-associated memory and that its role depends on the expression of CA1 N-methyl-d-aspartate (NMDA) receptors (Jafari-Sabet et al. Can J Physiol Pharmacol 96:45-50, 2018).
Objective
To clarify the exact mechanisms involved, the activation of CA1 nitric oxide (NO) signaling pathway by l-arginine (a nitric oxide precursor) on the interaction between tramadol and MK-801 in memory retrieval was examined. The dorsal hippocampal CA1 regions of adult male NMRI mice were bilaterally cannulated and a single-trial step-down inhibitory avoidance apparatus was used for the assessment of memory retrieval.
Results
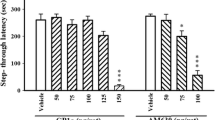
Post-training and/or pre-test microinjection of tramadol (0.5 and 1 μg/mouse) and/or a non-competitive NMDA receptor antagonist, MK-801 (0.25 and 0.5 μg/mouse), induced amnesia which were reversed when the same doses of the drugs were administered 24 h later in a pre-test session, suggesting tramadol state-dependent learning (SDL) and MK-801 SDL. The amnesia induced by post-training microinjection of tramadol (1 μg/mouse) was reversed by pre-test microinjection of MK-801 (0.25 and 0.5 μg/mouse). Pre-test microinjection of MK-801 (0.125 and 0.25 μg/mouse) with an ineffective dose of tramadol (0.25 μg/mouse) potentiated tramadol SDL. The amnesia induced by post-training microinjection of MK-801 (0.5 μg/mouse) was reversed by pre-test microinjection of tramadol (0.5 and 1 μg/mouse). Pre-test microinjection of tramadol (0.25 and 0.5 μg/mouse) with an ineffective dose of MK-801 (0.125 μg/mouse) potentiated MK-801 SDL. Pre-test microinjection of ineffective doses of l-arginine (0.125, 025, and 0.5 μg/mouse) improved amnesia induced by the co-administration of tramadol and MK-801. Pre-test microinjection of l-arginine (0.125, 025, and 0.5 μg/mouse) could not reverse amnesia induced by post-training microinjection of tramadol while same doses of l-arginine improved MK-801 response on tramadol SDL.
Conclusion
The results strongly propose that activation of CA1 NO signaling pathway has a pivotal role in cross SDL among tramadol and MK-801.






Similar content being viewed by others
References
Alijanpour S, Rezayof A, Zarrindast MR (2013) Dorsal hippocampal cannabinoid CB1 receptors mediate the interactive effects of nicotine and ethanol on passive avoidance learning in mice. Addict Biol 18:241–251
Athos J, Impey S, Pineda VV, Chen X, Storm DR (2002) Hippocampal CRE mediated gene expression is required for contextual memory formation. Nat Neurosci 5:1119–1120
Babalonis S, Lofwall MR, Nuzzo PA, Siegel AJ, Walsh SL (2013) Abuse liability and reinforcing efficacy of oral tramadol in humans. Drug Alcohol Depend 129:116–124
Barbosa J, Faria J, Queirós O, Moreira R, Carvalho F, Dinis-Oliveira RJ (2016) Comparative metabolism of tramadol and tapentadol: a toxicological perspective. Drug Metab Rev 15:1–16
Barros DM, Mello e Souza T, De David T, Choi H, Aguzzoli A, Madche C, Ardenghi P, Medina JH, Izquierdo I (2001) Simultaneous modulation of retrieval by dopaminergic D(1), beta-noradrenergic, serotonergic-1A and cholinergic muscarinic receptors in cortical structures of the rat. Behav Brain Res 124:1–7
Bassiony MM, Youssef UM, Hassan MS, Salah El-Deen GM, El-Gohari H, Abdelghani M, Abdalla A, Ibrahim DH (2017) Cognitive impairment and tramadol dependence. J Clin Psychopharmacol 37:61–66
Bodnar RJ (2016) Endogenous opiates and behavior: 2014. Peptides 75:18–70
Brigman JL, Wright T, Talani G, Prasad-Mulcare S, Jinde S, Seabold GK, Mathur P, Davis MI, Bock R, Gustin RM, Colbran RJ, Alvarez VA, Nakazawa K, Delpire E, Lovinger DM, Holmes A (2010) Loss of GluN2B-containing NMDA receptors in CA1 hippocampus and cortex impairs long-term depression, reduces dendritic spine density, and disrupts learning. J Neurosci 30:4590–4600
Cai Z, Xiao F, Lee B, Paul IA, Rhodes PG (1999) Prenatal hypoxia-ischemia alters expression and activity of nitric oxide synthase in the young rat brain and causes learning deficits. Brain Res Bull 49:359–365
Cammarota M, Bevilaqua LR, Ardenghi P, Paratcha G, Levi de Stein M, Izquierdo I, Medina JH (2000) Learning-associated activation of nuclear MAPK, CREB and Elk-1, along with Fos production, in the rat hippocampus after a one-trial avoidance learning: abolition by NMDA receptor blockade. Brain Res Mol Brain Res 76:36–46
Castellano C, Cestari V, Ciamei A (2001) NMDA receptors and learning and memory processes. Curr Drug Targets 2:273–283
Ceretta AP, Camera K, Mello CF, Rubin MA (2008) Arcaine and MK-801make recall state-dependent in rats. Psychopharmacology 201:405–411
Cooke SF, Bliss TV (2006) Plasticity in the human central nervous system. Brain 129:1659–1673
De Vries TJ, Shippenberg TS (2002) Neural systems underlying opiate addiction. J Neurosci 22:3321–3325
Epstein DH, Preston KL, Jasinski DR (2006) Abuse liability, behavioral pharmacology, and physical-dependence potential of opioids in humans and laboratory animals: lessons from tramadol. Biol Psychol 73:90–99
Feil R, Kleppisch T (2008) NO/cGMP-dependent modulation of synaptic transmission. Handb Exp Pharmacol 184:529–560
Ghasemzadeh Z, Rezayof A (2016) Role of hippocampal and prefrontal cortical signaling pathways in dextromethorphan effect on morphine-induced memory impairment in rats. Neurobiol Learn Mem 128:23–32
Harooni HE, Naghdi N, Sepehri H, Rohani AH (2009) The role of hippocampal nitric oxide (NO) on learning and immediate, short- and long-term memory retrieval in inhibitory avoidance task in male adult rats. Behav Brain Res 201:166–172
Harrod SB, Flint RW, Riccio DC (2001) MK-801 induced retrieval, but not acquisition, deficits for passive avoidance conditioning. Pharmacol Biochem Behav 69:585–593
Ishide T, Mancini M, Maher TJ, Chayaikul P, Ally A (2000) Rostral ventrolateral medulla opioid receptor activation modulates glutamate release and attenuates the exercise pressor reflex. Brain Res 865:177–185
Izquierdo I, Bevilaqua LR, Rossato JI, Bonini JS, Medina JH, Cammarota M (2006) Different molecular cascades in different sites of the brain control memory consolidation. Trends Neurosci 29:496–505
Jafari-Sabet M (2006a) NMDA receptor blockers prevents the facilitatory effects of post-training intra-dorsal hippocampal NMDA and physostigmine on memory retention of passive avoidance learning in rats. Behav Brain Res 169:20–127
Jafari-Sabet M (2006b) NMDA receptor antagonists antagonize the facilitatory effects of post-training intra-basolateral amygdala NMDA and physostigmine on passive avoidance learning. Eur J Pharmacol 529:122–128
Jafari-Sabet M (2011) Involvement of dorsal hippocampal muscarinic cholinergic receptors on muscimol state-dependent memory of passive avoidance in mice. Life Sci 88:1136–1141
Jafari-Sabet M, Jannat-Dastjerdi I (2009) Muscimol state-dependent memory: involvement of dorsal hippocampal mu-opioid receptors. Behav Brain Res 202:5–10
Jafari-Sabet M, Karimi AM (2017) Cross state-dependency of learning between arachidonylcyclopropylamide (ACPA) and muscimol in the mouse dorsal hippocampus. Pharmacol Biochem Behav 163:66–73
Jafari-Sabet M, Zarrindast MR, Rezayat M, Rezayof A, Djahanguiri B (2005) The influence of NMDA receptor agonist and antagonist on morphine state-dependent memory of passive avoidance in mice. Life Sci 78:57–163
Jafari-Sabet M, Banafshe HR, Khodadadnejad MA (2013) Modulation of muscimol state-dependent memory by α2-adrenoceptors of the dorsal hippocampal area. Eur J Pharmacol 710:92–99
Jafari-Sabet M, Khodadadnejad MA, Ghoraba S, Ataee R (2014) Nitric oxide in the dorsal hippocampal area is involved on muscimol state-dependent memory in the step-down passive avoidance test. Pharmacol Biochem Behav 117:37–143
Jafari-Sabet M, Jafari-Sabet AR, Dizaji-Ghadim A (2016) Tramadol state-dependent memory: involvement of dorsal hippocampal muscarinic acetylcholine receptors. Behav Pharmacol 27:470–478
Jafari-Sabet M, Mofidi H, Attarian-Khosroshahi MS (2018) NMDA receptors in the dorsal hippocampal area are involved in tramadol state-dependent memory of passive avoidance learning in mice. Can J Physiol Pharmacol 96:45–50
Jamali-Raeufy N, Nasehi M, Ebrahimi-Ghiri M, Zarrindast MR (2011) Cross state-dependency of learning between WIN55, 212-2 and scopolamine in rat dorsal hippocampus. Neurosci Lett 491:227–231
Korpi ER, den Hollander B, Farooq U, Vashchinkina E, Rajkumar R, Nutt DJ, Hyytiä P, Dawe GS (2015) Mechanisms of action and persistent neuroplasticity by drugs of abuse. Pharmacol Rev 67:872–1004
Lanier RK, Lofwall MR, Mintzer MZ, Bigelow GE, Strain EC (2010) Physical dependence potential of daily tramadol dosing in humans. Psychopharmacology 211:457–466
Lansink CS, Goltstein PM, Lankelma JV, McNaughton BL, Pennartz CM (2009) Hippocampus leads ventral striatum in replay of place-reward information. PLoS Biol 7:e1000173
Ledo A, Frade J, Barbosa RM, Laranjinha J (2004) Nitric oxide in brain: diffusion, targets and concentration dynamics in hippocampal subregions. Mol Asp Med 25:75–89
de Lima MN, Laranja DC, Bromberg E, Roesler R, Schröder N (2005) Pre- or post-training administration of the NMDA receptor blocker MK-801 impairs object recognition memory in rats. Behav Brain Res 156:139–143
Lisman J (2003) Long-term potentiation: outstanding questions and attempted synthesis. Philos Trans Royal Soc London B: Bio Sci 358:829–842
Ma YY, Meng L, Guo CY, Han JS, Lee DY, Cui CL (2009) Dose- and time dependent, context-induced elevation of dopamine and its metabolites in the nucleus accumbens of morphine-induced CPP rats. Behav Brain Res 204:192–199
Majlessi N, Choopani S, Bozorgmehr T, Azizi Z (2008) Involvement of hippocampal nitric oxide in spatial learning in the rat. Neurobiol Learn Mem 90:413–419
Martin JL, Itzhak Y (2000) 7-Nitroindazole blocks nicotine-induced conditioned place preference but not LiCl-induced conditioned place aversion. Neuroreport 11:947–949
McGaugh JL (2013) Making lasting memories: remembering the significant. Proc Natl Acad Sci U S A 110:10402–10407
Naqvi S, Martin KJ, Arthur JS (2014) CREB phosphorylation at Ser133 regulates transcription via distinct mechanisms downstream of cAMP and MAPK signaling. Biochem J 458:469–479
Neves G, Cooke SF, Bliss TV (2008) Synaptic plasticity, memory and the hippocampus: a neural network approach to causality. Nat Rev Neurosci 9:65–75
Nossaman VE, Ramadhyani U, Kadowitz PJ, Nossaman BD (2010) Advances in perioperative pain management: use of medications with dual analgesic mechanisms, tramadol & tapentadol. Anesthesiol Clin 28:647–666
O’Connor EC, Mead AN (2010) Tramadol acts as a weak reinforcer in the rat self-administration model, consistent with its low abuse liability in humans. Pharmacol Biochem Behav 96:279–286
Oliva P, Aurilio C, Massimo F, Grella A, Maione S, Grella E, Scafuro M, Rossi F, Berrino L (2002) The antinociceptive effect of tramadol in the formalin test is mediated by the serotonergic component. Eur J Pharmacol 445:179–185
Paoletti P, Neyton J (2007) NMDA receptor subunits: function and pharmacology. Curr Opin Pharmacol 7:39–47
Paxinos G, Franklin KBJ (2001) The mouse brain in stereotaxic coordinates, 2nd edn. Academic Press, New York
Place R, Farovik A, Brockmann M, Eichenbaum H (2016) Bidirectional prefrontal-hippocampal interactions support context-guided memory. Nat Neurosci 19:992–994
Peters J, De Vries TJ (2012) Glutamate mechanisms underlying opiate memories. Cold Spring Harb Perspect Med 2:a012088
Pittenger C, Huang YY, Paletzki RF, Bourtchouladze R, Scanlin H, Vronskaya S, Kandel ER (2002) Reversible inhibition of CREB/ATF transcription factors in region CA1 of the dorsal hippocampus disrupts hippocampus-dependent spatial memory. Neuron 34:447–462
Pu L, Bao GB, Xu NJ, Ma L, Pei G (2002) Hippocampal long-term potentiation is reduced by chronic opiate treatment and can be restored by re-exposure to opiates. J Neurosci 22:1914–1921
Rosa J, Myskiw JC, Furini CR, Sapiras GG, Izquierdo I (2014) Fear extinction can be made state-dependent on peripheral epinephrine: role of norepinephrine in the nucleus tractus solitarius. Neurobiol Learn Mem 113:55–61
Shen F, Wang XW, Ge FF, Li YJ, Cui CL (2016) Essential role of the NO signaling pathway in the hippocampal CA1 in morphine-associated memory depends on glutaminergic receptors. Neuropharmacology 102:216–228
Shiga Y, Minami K, Shiraishi M, Uezono Y, Murasaki O, Kaibara M, Shigematsu A (2002) The inhibitory effects of tramadol on muscarinic receptor-induced responses in Xenopus oocytes expressing cloned M(3) receptors. Anesth Analg 95:1269–1273
Shiraishi M, Minami K, Uezono Y, Yanagihara N, Shigematsu A (2001) Inhibition by tramadol of muscarinic receptor-induced responses in cultured adrenal medullary cells and in Xenopus laevis oocytes expressing cloned M1 receptors. J Pharmacol Exp Ther 299:255–260
Solati J (2011) Dorsal hippocampal N-methyl-D-aspartate glutamatergic and μ-opioidergic systems modulate anxiety behaviors in rats in a non-interactive manner. Kaohsiung J Med Sci 27:485–493
Wagner JJ, Etemad LR, Thompson AM (2001) Opioid-mediated facilitation of long-term depression in rat hippocampus. J Pharmacol Exp Ther 296:776–781
Whitlock JR, Heynen AJ, Shuler MG, Bear MF (2006) Learning induces long-term potentiation in the hippocampus. Science 313:1093–1097
Zarrindast MR, Rezayof A (2004) Morphine state-dependent learning: sensitization and interactions with dopamine receptors. Eur J Pharmacol 497:197–204
Zarrindast MR, Jafari-Sabet M, Rezayat M, Djahanguiri B, Rezayof A (2006a) Involvement of NMDA receptors in morphine state-dependent learning in mice. Int J Neurosci 116:731–743
Zarrindast MR, Nouraei N, Khallilzadeh A, Askari E (2006b) Influence of acute and sub-chronic nicotine pretreatment on morphine state-dependent learning. Behav Brain Res 173:268–273
Zarrindast MR, Ownegh V, Rezayof A, Ownegh F (2014) The involvement of dorsal hippocampus in dextromethorphan-induced state-dependent learning in mice. Pharmacol Biochem Behav 116:90–95
Zhang M, Jing L, Liu Q, Wen RT, Li JX, Li YL, Gong Q, Liang JH (2012) Tramadol induces conditioned place preference in rats: interactions with morphine and buprenorphine. Neurosci Lett 520:87–91
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
All animal experiments were conducted in accordance with the UK Animals (Scientific Procedures) Act, 1986 and associated guidelines and EU Directive 2010/63/EU for animal experiments and approved by the local ethical committee.
Conflict of interest
The authors declare that they have no conflict of interest.
Additional information
Research Highlights
• Tramadol and/or MK-801 induced amnesia and also state-dependent learning (SDL).
• Cross SDL occurred between tramadol and MK-801 in the mouse dorsal hippocampus.
• Intra-CA1 microinjection of l-arginine dose-dependently improved tramadol/MK-801-induced amnesia.
• Intra-CA1 microinjection of l-arginine dose-dependently increased MK-801 response upon tramadol-SDL.
Rights and permissions
About this article
Cite this article
Jafari-Sabet, M., Amiri, S. & Ataee, R. Cross state-dependency of learning between tramadol and MK-801 in the mouse dorsal hippocampus: involvement of nitric oxide (NO) signaling pathway. Psychopharmacology 235, 1987–1999 (2018). https://doi.org/10.1007/s00213-018-4897-5
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00213-018-4897-5