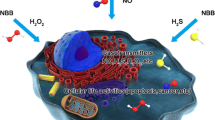
Abstract
Nitric oxide (NO) and its by-products are important biological signals in human physiology and pathology particularly in the vascular and immune systems. Thus, in situ determination of the NO-related molecule (NOx) levels using embedded sensors is of high importance particularly in the context of cellular biocompatibility testing. However, NOx analytical reference method dedicated to the evaluation of biomaterial biocompatibility testing is lacking. Herein, we demonstrate a PAPA-NONOate-based reference method for the calibration of NOx sensors. After, the validation of this reference method and its potentialities were demonstrated for the detection of the oxidative stress-related NO secretion of vascular endothelial cells in a 3D tissue issued from 3D printing. Such NOx detection method can be an integral part of cell response to biomaterials.

Graphical abstract











Similar content being viewed by others
References
Kim J, Koo BK, Knoblich JA. Human organoids: model systems for human biology and medicine. Nat Rev Mol Cell Biol [Internet]. Springer US; 2020;21(10):571–84. Available from: https://doi.org/10.1038/s41580-020-0259-3.
Azizipour N, Avazpour R, Rosenzweig DH, Sawan M, Ajji A. Evolution of biochip technology: a review from lab-on-a-chip to organ-on-a-chip. Micromachines. 2020;11(6):599–614.
Pollet AMAO, den Toonder JMJ. Recapitulating the vasculature using organ-on-chip technology. Bioengineering. 2020;7(1):1–18.
Kodzius R, Schulze F, Gao X, Schneider MR. Organ-on-chip technology: current state and future developments. Genes (Basel). 2017;8(266):8–10.
Shrestha J, Ghadiri M, Shanmugavel M, Razavi Bazaz S, Vasilescu S, Ding L, et al. A rapidly prototyped lung-on-a-chip model using 3D-printed molds. Organs-on-a-Chip [Internet]. Elsevier Ltd; 2019;1:100001. Available from: https://doi.org/10.1016/j.ooc.2020.100001.
Wu S, Chen-Yu C, Kimberly L, Gregory FP, Bentley WE. Chip mudularity enables molecular information acess from organ-on-chip devices with quality control. Sensors Actuators B Chem. 2019;295:30–9.
Zhang YS, Aleman J, Shin SR, Kilic T, Kim D, Shaegh SAM, et al. Multisensor-integrated organs-on-chips platform for automated and continual in situ monitoring of organoid behaviors. Proc Natl Acad Sci U S A. 2017;114(12):E2293–302.
Salzitsa A, Kassanos Panagiotis YG-Z. Multi-parametric rigid and flexible, low-cost, disposable sensing platforms for biomedica applications. Biosens Bioelectron. 2018;102:668–75.
Moncada S, Higgs EA. The discovery of nitric oxide and its role in vascular biology. Br J Pharmacol. 2006;147(1):193–201.
Luiking YC, Engelen MPKJ, Deutz NE. Regulation of nitric oxide production in health and disease. Curr Opin Clin Nutr Metab Care [Internet]. 2010;13(1):97–104. Available from: https://www.ncbi.nlm.nih.gov/pmc/articles/PMC2953417/.
Coleman JW. Nitric oxide in immunity and inflammation. Int Immunopharmacol. 2001;1(8):1397–406.
Farah C, Michel LYM, Balligand JL. Nitric oxide signalling in cardiovascular health and disease. Nat Rev Cardiol. 2018;15(5):292–316.
Schuman EM, Madison DV. Nitric oxide and angiogenesis. J Neuro-Oncol. 2000;272(50):139–48.
Wang J, Song M, Chen B, Wang L, Zhu R. Effects of pH and H2O2 on ammonia, nitrite, and nitrate transformations during UV254nm irradiation: implications to nitrogen removal and analysis. Chemosphere [Internet]. Elsevier Ltd; 2017;184:1003–11. Available from: https://doi.org/10.1016/j.chemosphere.2017.06.078.
Dubbe H, Bühner F, Nieken U. Rapid estimation of NO oxidation kinetic data from experiments. Chemie-Ingenieur-Technik. 2019;91(1):167–9.
Hakim TS, Sugimori K, Camporesi EM, Andersen G. Half-life of nitric oxide in aqueous solutions with and without haemoglobin. Physiol Meas. 1996;17(4):267–77.
Evan HM, Mark SH. Analytical chemistry of nitric oxide. Annu Rev Anal Chem. 2009;3:409–33.
Pinder AG, Rogers SC, Khalatbari A, Ingram TE, James PE. The measurement of nitric oxide and its metabolites in biological samples by ozone-based chemiluminescence. Methods Mol Biol. 2008;476:11–28. https://doi.org/10.1007/978-1-59745-129-1_2.
Tsui CT, MacGillivray SR, Weber SM, McAllister L, Churchward MA, Dennison CR, et al. Applying a novel 3D hydrogel cell culture to investigate activation of microglia due to rotational kinematics associated with mild traumatic brain injury. J Mech Behav Biomed Mater [Internet]. Elsevier Ltd; 2020;(in press). Available from: https://doi.org/10.1016/j.jmbbm.2020.104176.
Khazaei MR, Nasr-Esfahani MH, Chobsaz F, Khazaei M. Noscapine inhibiting the growth and angiogenesis of human eutopic endometrium of endometriosis patients through expression of apoptotic genes and nitric oxide reduction in three-dimensional culture model. Iran J Pharm Res. 2019;18(2):836–45.
Wang B, Chen R, Gao H, Lv X, Chen L, Wang W, et al. A comparative study unraveling the effects of TNF-α stimulation on endothelial cells between 2D and 3D culture. Biomed Mater. 2020;15(6):065018. https://doi.org/10.1088/1748-605X/ab95e3.
Hurst RD, Clark JB. The utility of the nitric oxide electrochemical sensor in biomedical research. Sensors. 2003;3(8):321–9.
Zhang Q, Wei D, Liu J. In vivo reversal of doxorubicin resistance by (-)-epigallocatechin gallate in a solid human carcinoma xenograft. Cancer Lett. 2004;208(2):179–86.
Brown MD, Schoenfisch MH. Electrochemical nitric oxide sensors: principles of design and characterization. Chem Rev. 2019;119(22):11551–75.
Park K, Jeong H, Tanum J, Yoo JC, Hong J. Poly-L-lysine/poly-L-glutamic acid-based layer-by-layer self-assembled multilayer film for nitric oxide gas delivery. J Ind Eng Chem [Internet]. The Korean Society of Industrial and Engineering Chemistry; 2019;69:263–8. Available from: https://doi.org/10.1016/j.jiec.2018.09.005.
Mukosera GT, Liu T, Ishtiaq Ahmed AS, Li Q, Sheng MHC, Tipple TE, et al. Detection of dinitrosyl iron complexes by ozone-based chemiluminescence. Nitric Oxide Biol Chem. 2018;79(July):57–67.
Li H, Liu W, Kan R. A compact low-noise photodiode detection system for chemiluminescence nitric oxide analyzer. Rev Sci Instrum [Internet]. 2019;90(4):2–5. Available from: https://doi.org/10.1063/1.5082400.
Schiller B, Hammer J, Barben J, Trachsel D. Comparability of a hand-held nitric oxide analyser with online and offline chemiluminescence-based nitric oxide measurement. Pediatr Allergy Immunol. 2009;20(7):679–85.
Korn S, Wilk M, Voigt S, Weber S, Keller T, Buhl R. Measurement of fractional exhaled nitric oxide: comparison of three different analysers. Respiration. 2020;99(1):1–8.
Murthy VR, Escobar H, Norberg M, Lachica CI, Gratny LL, Sherman AK, et al. A novel method of measuring fractional exhaled nitric oxide in tracheostomized ventilator-dependent children. Respir Care. 2017;62(5):595–601.
Förstermann U, Sessa WC. Nitric oxide synthases: regulation and function. Eur Heart J. 2012;33(7):829–37.
Stuehr DJ. Enzymes of the L-arginine to nitric oxide pathway. J Nutr. 2004;134(10):2748S–51S.
Bradley SA, Steinert JR. Characterisation and comparison of temporal release profiles of nitric oxide generating donors. J Neurosci Methods [Internet]. Elsevier B.V.; 2015;245:116–24. Available from: https://doi.org/10.1016/j.jneumeth.2015.02.024.
Coneski PN, Schoenfisch MH. Nitric oxide release: part III. Measurement and reporting. Chem Soc Rev. 2012;41(10):3753–8.
Cao GJ, Fisher CM, Jiang X, Chong Y, Zhang H, Guo H, et al. Platinum nanoparticles: an avenue for enhancing the release of nitric oxide from: S -nitroso- N -acetylpenicillamine and S -nitrosoglutathione. Nanoscale. 2018;10(23):11176–85.
Keefer LK, Nims RW, Davies KM, Wink DA. NONOates as nitric oxide donors: convenient nitric oxide dosage forms. Methods Enzymol. 1996;268:281–93.
Hrabie JA, Klose JR, Wink DA, Keefer LK. New nitric oxide-releasing zwitterions derived from polyamines. J Org Chem. 1993;58(6):1472–6.
Micro T, Vessels R. Reduction of biological effluents in purge and trap micro reatcion vessels and detection of endothelium-derived nitric oxide (Endo) by Chemiluminescence. J Mol Cell Cardiol. 1991;23:389–93.
Samouilov A, Zweier JL. Development of chemiluminescence-based methods for specific quantitation of nitrosylated thiols. Anal Biochem. 1998;258(2):322–30.
Castiaux AD, Spence DM, Martin RS. Review of 3D cell culture with analysis in microfluidic systems. Anal Methods R Soc Chem. 2019;11(33):4220–32.
Kapałczyńska M, Kolenda T, Przybyła W, Zajączkowska M, Teresiak A, Filas V, et al. 2D and 3D cell cultures – a comparison of different.pdf. Arch Med Sci. 2018;14(4):910–9.
Vrana NE, Cahill PA, McGuinness GB. Endothelialization of PVA/gelatin cryogels for vascular tissue engineering: effect of disturbed shear stress conditions. J Biomed Mater Res - Part A. 2010;94(4):1080–90.
Knopf-Marques H, Barthes J, Lachaal S, Mutschler A, Muller C, Dufour F, et al. Multifunctional polymeric implant coatings based on gelatin, hyaluronic acid derivative and chain length-controlled poly(arginine). Mater Sci Eng. 2019;104:109898. https://doi.org/10.1016/j.msec.2019.109898.
Knopf-Marques H, Barthes J, Wolfova L, Vidal B, Koenig G, Bacharouche J, et al. Auxiliary biomembranes as a directional delivery system to control biological events in cell-laden tissue-engineering scaffolds. ACS Omega. 2017;2(3):918–29.
Funding
PANBioRA project has received funding from the European Union’s Horizon 2020 research and innovation program under grant agreement No 760921.
Author information
Authors and Affiliations
Corresponding authors
Ethics declarations
Conflict of interest
The authors declare that there are no conflicts of interest.
Additional information
Publisher’s note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Chmayssem, A., Monsalve-Grijalba, K., Alias, M. et al. Reference method for off-line analysis of nitrogen oxides in cell culture media by an ozone-based chemiluminescence detector. Anal Bioanal Chem 413, 1383–1393 (2021). https://doi.org/10.1007/s00216-020-03102-9
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00216-020-03102-9