Abstract
Backgound
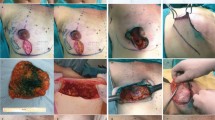
Nipple-sparing mastectomy with immediate implant reconstruction is an increasingly popular technique for both treatment of breast cancer and risk-reducing surgery, with an evolving body of evidence confirming low complication rates and satisfactory patient outcomes. Immediate implant reconstruction usually requires use of one of many available meshes for complete implant coverage. The aim of this study was to assess outcomes after nipple-sparing mastectomy using synthetic absorbable TIGR mesh.
Methods
A retrospective review of a prospectively maintained database of 164 skin and nipple-sparing mastectomies with immediate implant reconstruction using TIGR mesh was performed. Data was retrieved and cross-checked with electronic patient records. Data was analysed with regard to patient demographics, indications for surgery, surgical procedure, complication rates and locoregional recurrence rates.
Results
Of 164 implant reconstructions, forty-three were performed after nipple-sparing mastectomy. No differences in outcomes were seen between the two groups except for a higher incidence of skin or nipple necrosis in the nipple-sparing group (12% versus 2%). There was no nipple loss in this cohort. Infection rate in the nipple-sparing group was 9% versus 11% in the skin-sparing group, with implant loss rates of 9% and 6%, respectively. Mean follow-up was 23.6 months.
Conclusions
Our study has shown that immediate implant reconstruction after nipple-sparing mastectomy using TIGR mesh is safe and feasible, with low rates of early and medium-term complications.
Level of evidence: Level III, therapeutic study.

Similar content being viewed by others
References
Colwell AS, Tessler O, Lin AM et al (2014) Breast reconstruction following nipple sparing mastectomy: predictors of complications, reconstruction outcomes and 5-year trends. Plastic Reconstr Surg J 133(3):496
Gerber B, Krause A, Reimer T et al (2003) Skin-sparing mastectomy with conservation of the nipple-areola complex and autologous reconstruction is an oncologically safe procedure. Ann Surg 238:120
Sacchini V, Pinotti JA, Barros AC et al (2006) Nipple-sparing mastectomy for breast cancer and risk reduction: oncologic or technical problem? J Am Coll Surg 293:704
Crowe JP, Kim JA, Metman R, Banbury J, Patrick RJ, Baynes D (2004) Nipple-sparing mastectomy: technique and results of 54 procedures. Arch Surg 139:148
Bishop CC, Singh S, Nash AG (1990) Mastectomy and breast reconstruction preserving the nipple. Ann R Coll Surg Engl 72:87
de Alcantara FP, Capko D, Barry JM, Morrow M, Pusic A, Sacchini VS (2011) Nipple-sparing kmastectomy for breast cancer and risk-reducing surgery: the memorial Sloan-Kettering Cancer Centre experience. Ann Surg Oncol 18:3117
Spear SL, Willey SC, Fedlman ED et al (2011) Nipple-sparing mastectomy for prophylactic and therapeutic indications. Plast Reconstr Surg 128:1005
Mitchell SD, Willey SC, Beitsch P, Feldman S (2017) Evidence based outcomes of the American Society of Breast Surgeons Nipple Sparing Mastectomy Registry. Gland Surg 7(3):247
Agha RA, Omran YA, Wellstead G, Sagoo H et al (2019) Systematic review of therapeutic nipple-sparing versus skin-sparing mastectomy. BJS Open 3:135
Ashikari AY, Keleman PR, Tastan B, Salzberg CA, Ashikari RH (2017) Nipple sparing mastectomy technique: a literature review and an inframammary technique. Gland Surg 7(3):273
Tousimis E, Haslinger M (2017) Overview of indications for nipple sparing mastectomy. Gland Surg 7(3):288
Gunnarsson GL, Heidemann LN, Bille C, Sorenson JA, Thomsen JB (2017) Nipple sparing mastectomy and the evolving direct to implant beast reconstruction. Gland Surg 7(3):267
Bertozzi N, Pesce M, Santi P, Raposio E (2017) One-stage immediate breast reconstruction: a concise review. Biomed Res Int 2017:6486859
Komorowski AL, Zanini V, Regolo L, Carolei A, Wusocki WM, Costa A (2006) Necrotic complications after nipple- and areola-sparing mastectomy. World J Surg 30(8):1410–1413
Wang ED, Lanier ST, Yilmaz T et al (2010) Acellular dermal matrix in tissue expander/implant breast reconstruction. Plast Reconstr Surg 126:111
Wang ED, Lanier ST, Phillips BT et al (2010) Risks and rewards of acellular dermal matrix in tissue expander/implant breast reconstruction. Plast Reconstr Surg 125:27
Wong L, Buseman J, Oldham J (2011) Infectious complications with acellular dermal matrices and breast reconstruction. Plast Reconstr Surg 127:91
Barber MD, Williams L, Anderson EDC, Neades GT, Raine C, Young O, Kulkarni D, Young I, Dixon JM (2015) Outcome of the use of acellular-dermal matrix to assist implant-based breast reconstruction in a single Centre. Eur J Surg Oncol 41(1):100–105
Becker H, Lind JG (2013) The use of synthetic mesh in reconstructive, revision, and cosmetic breast surgery. Aesthet Plast Surg 37:914
Hallberg H, Elander A, Kolby L, Hansso E (2019) A biological or a synthetic mesh in immediate breast reconstruction? A cohort-study of long-term health-related quality of life (HrQoL). Eur J Surg Oncol. https://doi.org/10.1016/j.ejso.2019.03.013
Wellisch DK, Schain WS, Noone RB, Little JW 3rd (1981) The psychological contribution of nipple addition in breast reconstruction. Plast Reconstr Surg 80(5):699
Colwell AS, Christensen JM (2017) Nipple-sparing mastectomy and direct-to-implant breast reconstruction. Plast Reconstr Surg 140(5S):44S
Mori H, Uemura N, Okazaki M, Nakagawa T, Sato T (2016) Nipple malposition after nipple-sparing mastectomy and expander-implant reconstruction. Breast Cancer 23(5):740–744
Galimberti V, Vicini E, Corso G, Morigi C, Fontana S, Sacchini V, Veronesi P (2017) Nipple-sparing and skin-sparing mastectomy: review of aims, oncological safety and contraindications. Breast 34(S1):S82
Wijayanayagam A, Kumar AS, Foster RD, Esserman LJ (2008) Optimizing the total skin-sparing mastectomy. Arch Surg 143(1):38–45
Pompeii S, Evangelidou D, Arelli F, Ferrnate G (2018) The use of TIGR matrix in breast aesthetic and reconstructive surgery. Clin Past Surg 45(1):65
Sharma S, Van Bafsel S, Barry M, Kell MR (2016) De novo experience of resorbable woven mesh in immediate breast recosntruction post-mastectomy. Eur J Plast Surg 40(1):17–22
Hallbaerg H, Lewin R, Elander A, Hansson E (2018) TIGR matrix surgical mesh - a two-year follow-up study and complication analysis in 65 immediate breast reconstructions. J Plast Surg Hand Surg 52(4):253
Potter S, Conroy EJ, Cutress RI, Williamson PR, Whisker L, Thrush S, Skillman J, Barnes NLP, Mylvaganam S, Teasdale E, Jain A, Gardiner MD, Blazeby JM, Holcombe C, iBRA Steering Group, Breast Reconstruction Research Collaborative (2019) Short-term safety outcomes of mastectomy and immediate implant-based breast reconstruction with and without mesh (iBRA): a multicentre, prospective cohort study. Lancet Oncol 20(2):254–266
Gschwantler-Laulich D, Leser C, Salma M (2018) Singer CF (2018) direct-to-implant breast reconstruction: higher complication rate vs cosmetic benefits. Breast J 24(6):957
Heidemann LN, Gunnarsson GL, Salzberg CA, Sorenson JA, Thomsen JB (2018) Complications following nipple-sparing mastectomy and immediate acellular dermal matrix implant-based breast reconstruction – a systematic review and meta-analysis. Plast Reconstr Surg Glob Open 6(1):e1625
Tran BNN, Fadayomi A, Lin SJ, Singhal D, Lee BT (2017) Cost analysis of postmastectomy reconstruction: a comparison of two staged implant reconstruction using tissue expander and acellular dermal matrix with abdominal-based perforated free flaps. J Surg Oncol 116(4):439
Krishnan NM, Chatterjee A, Rosenkranz KM, Powell SG, Nigriny JF, Vidal DC (2014) The cost effectiveness of acellular dermal matrix in expander-implant immediate reconstruction. J Plast Reconstr Aesthet Surg 67(4):468
Dieterich M, Paepke S, Zwiefel K et al (2013) Implant-based breast reconstruction using a titanium-coated polypropylene mesh (TiLOOP Bra): a multicenter study of 231 cases. Plast Reconstr Surg 132:8e
Baldelli I, Cardoni G, Franchelli S, Fregatti P, Friedman D, Pesce M (2016) Implant-based breast reconstruction using a polyester mesh (Surgimesh-PET): a retrospective single-center study. Plast Reconstr Surg 137(6):931e
Pukancsik D, Kelemen P, Gulyas G, Ujhely M, Kovacs E, Eles K et al (2017) Clinical experiences with the use of ULTRAPRO® mesh in single-stage direct-to-implant immediate postmastectomy breast reconstruction in 102 patients: a retrospective cohort study. Eur J Surg Oncol 43(7):1244–1251
Tessler O, Reish RG, Maman DY, Smith BL, Austen WG Jr (2014) Beyond biologics: absorbable mesh as a low-cost, low-complication sling for implant-based breast reconstruction. Plast Reconstr Surg 133:90e–99e
Kim T, Cho H (2013) The suitability of absorbable mesh insertion for oncoplastic breast surgery in patients with breast cancer scheduled to be irradiated. J Breast Cancer 16:84
Logan Ellis H, Asaolu O, Nebo V, Kasem A (2016) Biological and synthetic mesh use in breast reconstructive surgery: a literature review. World J Surg Oncol 14:121
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
The authors declare no conflict of interest.
Ethical statement
Due to the retrospective database audit analysis undertaken in this study, ethics committee approval and informed consent was not required.
Additional information
Publisher’s note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Quinn, E.M., Barry, M. & Kell, M. Immediate implant reconstruction using absorbable TIGR mesh after nipple-sparing mastectomy. Eur J Plast Surg 43, 279–284 (2020). https://doi.org/10.1007/s00238-019-01603-0
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00238-019-01603-0