Abstract
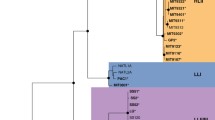
Prochlorococcus is the most abundant photosynthetic prokaryote on our planet. The extensive ecological literature on the Prochlorococcus collective (PC) is based on the assumption that it comprises one single genus comprising the species Prochlorococcus marinus, containing itself a collective of ecotypes. Ecologists adopt the distributed genome hypothesis of an open pan-genome to explain the observed genomic diversity and evolution patterns of the ecotypes within PC. Novel genomic data for the PC prompted us to revisit this group, applying the current methods used in genomic taxonomy. As a result, we were able to distinguish the five genera: Prochlorococcus, Eurycolium, Prolificoccus, Thaumococcus, and Riococcus. The novel genera have distinct genomic and ecological attributes.



Similar content being viewed by others
References
Chisholm SW, Frankel SL, Goericke R et al (1992) Prochiorococcus marinus nov. gen. nov. sp.: an oxyphototrophic marine prokaryote containing divinyl chlorophyll a and b. Arch Microbiol:297–300
Chisholm SW (2017) Prochlorococcus. Curr Biol 27:R447–R448. https://doi.org/10.1016/j.cub.2017.02.043
Braakman R, Follows MJ, Chisholm SW (2017) Metabolic evolution and the self-organization of ecosystems. Proc Natl Acad Sci 114:E3091–E3100. https://doi.org/10.1073/pnas.1619573114
Ting CS, Ramsey ME, Wang YL, Frost AM, Jun E, Durham T (2009) Minimal genomes, maximal productivity: comparative genomics of the photosystem and light-harvesting complexes in the marine cyanobacterium, Prochlorococcus. Photosynth Res 101:1–19
Scanlan DJ, Ostrowski M, Mazard S, Dufresne A, Garczarek L, Hess WR, Post AF, Hagemann M, Paulsen I, Partensky F (2009) Ecological genomics of marine picocyanobacteria. Microbiol Mol Biol Rev 73:249–299. https://doi.org/10.1128/MMBR.00035-08
Ralf G, Repeta DJ (1992) The pigments of Prochlorococcus marinus: the presence of divinylchlorophyll a and b in a marine procaryote. Limnol Oceanogr 37:425–433
Partensky F, Garczarek L (2010) Prochlorococcus: advantages and limits of minimalism. Ann Rev Mar Sci 2:305–331
Mühling M (2012) On the culture-independent assessment of the diversity and distribution of Prochlorococcus. Environ Microbiol 14:567–579
Coleman ML, Sullivan MB, Martiny AC et al (2006) Genomic islands and the ecology and evolution of Prochlorococcus. Science (80- ) 311:1768–1770. https://doi.org/10.1126/science.1122050
Coleman ML, Chisholm SW (2007). Code and context: Prochlorococcus as a model for cross-scale biology. 15:15–407. https://doi.org/10.1016/j.tim.2007.07.001
Kashtan N, Roggensack SE, Rodrigue S et al (2014) Single-cell genomics reveals hundreds of coexisting subpopulations in wild Prochlorococcus. Science (80- ) 344:416–420. https://doi.org/10.1126/science.1248575
Biller SJ, Berube PM, Berta-Thompson JW, Kelly L, Roggensack SE, Awad L, Roache-Johnson KH, Ding H, Giovannoni SJ, Rocap G, Moore LR, Chisholm SW (2014) Genomes of diverse isolates of the marine cyanobacterium Prochlorococcus. Sci Data 1:140034
Kashtan N, Roggensack SE, Berta-Thompson JW, Grinberg M, Stepanauskas R, Chisholm SW (2017) Fundamental differences in diversity and genomic population structure between Atlantic and Pacific Prochlorococcus. ISME J 11:1997–2011. https://doi.org/10.1038/ismej.2017.64
Berube PM, Biller SJ, Hackl T, Hogle SL, Satinsky BM, Becker JW, Braakman R, Collins SB, Kelly L, Berta-Thompson J, Coe A, Bergauer K, Bouman HA, Browning TJ, de Corte D, Hassler C, Hulata Y, Jacquot JE, Maas EW, Reinthaler T, Sintes E, Yokokawa T, Lindell D, Stepanauskas R, Chisholm SW (2018) Data descriptor: single cell genomes of Prochlorococcus, Synechococcus, and sympatric microbes from diverse marine environments. Sci Data 5:1–11. https://doi.org/10.1038/sdata.2018.154
Chisholm SW (2012) The life and times of the ocean’s smallest photosynthetic cell. In: Kolter R, Maloy S (eds) Microbes and evolution: the world that Darwin never saw. ASM Press, pp 165–172
Thompson CC, Silva GGZ, Vieira NM, Edwards R, Vicente ACP, Thompson FL (2013) Genomic taxonomy of the genus Prochlorococcus. Microb Ecol 66:752–762. https://doi.org/10.1007/s00248-013-0270-8
Coutinho F, Tschoeke DA, Thompson F, Thompson C (2016) Comparative genomics of Synechococcus and proposal of the new genus Parasynechococcus. PeerJ 4:e1522. https://doi.org/10.7717/peerj.1522
Walter JM, Coutinho FH, Dutilh BE, Swings J, Thompson FL, Thompson CC (2017) Ecogenomics and taxonomy of Cyanobacteria phylum. Front Microbiol 8. https://doi.org/10.3389/fmicb.2017.02132
Tettelin H, Riley D, Cattuto C, Medini D (2008) Comparative genomics: the bacterial pan-genome. Curr Opin Microbiol 11:472–477
Lapierre P, Gogarten JP (2009) Estimating the size of the bacterial pan-genome. Trends Genet 25:107–110
Rosselló-Móra R, Whitman WB (2019) Dialogue on the nomenclature and classification of prokaryotes. Syst Appl Microbiol 42:5–14. https://doi.org/10.1016/j.syapm.2018.07.002
De Vos P, Thompson F, Thompson C, Swings J (2017) A flavor of prokaryotic taxonomy: systematics revisited. Microbial resources: from functional existence in nature to applications. Elsevier Inc., pp 29–44
Sun Z, Blanchard JL (2014) Strong genome-wide selection early in the evolution of Prochlorococcus resulted in a reduced genome through the loss of a large number of small effect genes. PLoS One 9:e88837
Johnson ZI, Zinser ER, Coe A et al (2006) Niche partitioning among Prochlorococcus ecotypes along ocean-scale environmental gradients. Science (80- ) 311:1737–1740
Garczarek L, Dufresne A, Rousvoal S, West NJ, Mazard S, Marie D, Claustre Hé, Raimbault P, Post AF, Scanlan DJ, Partensky Féé (2007) High vertical and low horizontal diversity of Prochlorococcus ecotypes in the Mediterranean Sea in summer. FEMS Microbiol Ecol 60:189–206. https://doi.org/10.1111/j.1574-6941.2007.00297.x
Rusch DB, Martiny AC, Dupont CL, Halpern AL, Venter JC (2010) Characterization of Prochlorococcus clades from iron-depleted oceanic regions. Proc Natl Acad Sci 107:16184–16189
Larkin AA, Blinebry SK, Howes C, Lin Y, Loftus SE, Schmaus CA, Zinser ER, Johnson ZI (2016) Niche partitioning and biogeography of high light adapted Prochlorococcus across taxonomic ranks in the North Pacific. ISME J 10:1555–1567
West NJ, Lebaron P, Strutton PG, Suzuki MT (2011) A novel clade of Prochlorococcus found in high nutrient low chlorophyll waters in the South and Equatorial Pacific Ocean. ISME J 5:933–944
Huang S, Wilhelm SW, Harvey HR, Taylor K, Jiao N, Chen F (2012) Novel lineages of prochlorococcus and synechococcus in the global oceans. ISME J 6:285–297. https://doi.org/10.1038/ismej.2011.106
Kent AG, Dupont CL, Yooseph S, Martiny AC (2016) Global biogeography of Prochlorococcus genome diversity in the surface ocean. ISME J 10:1856–1865. https://doi.org/10.1038/ismej.2015.265
Moore LR, Chisholm SW (1999) Photophysiology of the marine cyanobacterium Prochlorococcus: ecotypic differences among cultured isolates. Limnol Oceanogr 44:628–638
Biller SJ, Berube PM, Lindell D, Chisholm SW (2015) Prochlorococcus: the structure and function of collective diversity. Nat Rev Microbiol 13:13–27. https://doi.org/10.1038/nrmicro3378
Rocap G, Larimer FW, Lamerdin J, Malfatti S, Chain P, Ahlgren NA, Arellano A, Coleman M, Hauser L, Hess WR, Johnson ZI, Land M, Lindell D, Post AF, Regala W, Shah M, Shaw SL, Steglich C, Sullivan MB, Ting CS, Tolonen A, Webb EA, Zinser ER, Chisholm SW (2003) Genome divergence in two Prochlorococcus ecotypes reflects oceanic niche differentiation. Nature 424:1042–1047
Chandler JW, Lin Y, Gainer PJ, Post AF, Johnson ZI, Zinser ER (2016) Variable but persistent coexistence of Prochlorococcus ecotypes along temperature gradients in the ocean’s surface mixed layer. Environ Microbiol Rep 8:272–284
Binder BJ, Chisholm SW, Olson RJ, Frankel SL, Worden AZ (1996) Dynamics of picophytoplankton, ultraphytoplankton and bacteria in the central equatorial Pacific. Deep Sea Res Part II Top Stud Oceanogr 43:907–931
Ferris MJ, Palenik B (1998) Niche adaptation in ocean cyanobacteria. Nature 396:226–228
García-Fernández JM, Hess WR, Houmard J, Partensky F (1998) Expression of the psbA gene in the marine oxyphotobacteria Prochlorococcus spp. Arch Biochem Biophys 359:17–23
Fuhrman JA, McCallum K, Davis AA (1993) Phylogenetic diversity of subsurface marine microbial communities from the Atlantic and Pacific Oceans. Appl Environ Microbiol 59:1294–1302
Martiny AC, Coleman ML, Chisholm SW (2006) Phosphate acquisition genes in Prochlorococcus ecotypes: evidence for genome-wide adaptation. Proc Natl Acad Sci 103:12552–12557
Martiny AC, Huang Y, Li W (2009) Occurrence of phosphate acquisition genes in Prochlorococcus cells from different ocean regions. Environ Microbiol 11:1340–1347
Moore LR, Post AF, Rocap G, Chisholm SW (2002) Utilization of different nitrogen sources by the marine cyanobacteria Prochlorococcus and Synechococcus. Limnol Oceanogr 47:989–996
Sullivan MB, Waterbury JB, Chisholm SW (2003) Cyanophages infecting the oceanic cyanobacterium Prochlorococcus. Nature 424:1047–1051
Sher D, Thompson JW, Kashtan N, Croal L, Chisholm SW (2011) Response of Prochlorococcus ecotypes to co-culture with diverse marine bacteria. ISME J 5:1125–1132
Biller SJ, Coe A, Chisholm SW (2016) Torn apart and reunited: impact of a heterotroph on the transcriptome of Prochlorococcus. ISME J 10:2831–2843
Biller SJ, Berube PM, Dooley K, Williams M, Satinsky BM, Hackl T, Hogle SL, Coe A, Bergauer K, Bouman HA, Browning TJ, de Corte D, Hassler C, Hulston D, Jacquot JE, Maas EW, Reinthaler T, Sintes E, Yokokawa T, Chisholm SW (2018) Data descriptor: marine microbial metagenomes sampled across space and time. Sci Data 5:1–7. https://doi.org/10.1038/sdata.2018.176
Muñoz-marín MC, Gómez-baena G, López-lozano A et al (2020) Mixotrophy in marine picocyanobacteria: use of organic compounds by Prochlorococcus and Synechococcus. ISME J. 14:1065–1073. https://doi.org/10.1038/s41396-020-0603-9
Vandamme P, Pot B, Gillis M et al (1996) Polyphasic taxonomy, a consensus approach to bacterial systematics. Microbiol Mol Biol Rev 60:407–438
Wayne LG, Brenner DJ, Colwell RR et al (1987) Report of the Ad Hoc Committee on Reconciliation of Approaches to Bacterial Systematics. Int J Syst Evol Microbiol 37:463–464. https://doi.org/10.1099/00207713-37-4-463
Stackebrandt E, Frederiksen W, Garrity GM, Grimont PAD, Kämpfer P, Maiden MCJ, Nesme X, Rosselló-Mora R, Swings J, Trüper HG, Vauterin L, Ward AC, Whitman WB (2002) Report of the ad hoc committee for the re-evaluation of the species definition in bacteriology. Int J Syst Evol Microbiol 52:1043–1047
Gevers D, Cohan FM, Lawrence JG, Spratt BG, Coenye T, Feil EJ, Stackebrandt E, de Peer YV, Vandamme P, Thompson FL, Swings J (2005) Defining prokaryotic species Reevaluating prokaryotic species. Nat Rev Microbiol 3:733–739
Thompson CC, Chimetto L, Edwards RA, Swings J, Stackebrandt E, Thompson FL (2013) Microbial genomic taxonomy. BMC Genomics 14:913. https://doi.org/10.1186/1471-2164-14-913
Thompson CC, Amaral GR, Campeão M, Edwards RA, Polz MF, Dutilh BE, Ussery DW, Sawabe T, Swings J, Thompson FL (2015) Microbial taxonomy in the post-genomic era: rebuilding from scratch? Arch Microbiol 197:359–370
Castelle CJ, Banfield JF (2018) Major new microbial groups expand diversity and alter our understanding of the tree of life. Cell 172:1181–1197
Parks DH, Chuvochina M, Waite DW, Rinke C, Skarshewski A, Chaumeil PA, Hugenholtz P (2018) A standardized bacterial taxonomy based on genome phylogeny substantially revises the tree of life. Nat Biotechnol 36:996–1004. https://doi.org/10.1038/nbt.4229
Hyatt D, Chen G-L, LoCascio PF et al (2010) Prodigal: prokaryotic gene recognition and translation initiation site identification. BMC Bioinf 11:119
Parks DH, Imelfort M, Skennerton CT, Hugenholtz P, Tyson GW (2015) CheckM: assessing the quality of microbial genomes recovered from isolates, single cells, and metagenomes. Genome Res 25:1043–1055
Konstantinidis KT, Tiedje JM (2005) Towards a genome-based taxonomy for prokaryotes. J Bacteriol 187:6258–6264
Camacho C, Coulouris G, Avagyan V, Ma N, Papadopoulos J, Bealer K, Madden TL (2009) BLAST+: architecture and applications. BMC Bioinformatics 10:421
Katoh K, Asimenos G, Toh H (2009) Multiple alignment of DNA sequences with MAFFT. Methods Mol Biol. https://doi.org/10.1007/978-1-59745-251-9_3
Price MN, Dehal PS, Arkin AP (2010) FastTree 2 - approximately maximum-likelihood trees for large alignments. PLoS One. 5:e9490. https://doi.org/10.1371/journal.pone.0009490
Huerta-Cepas J, Serra F, Bork P (2016) ETE 3: Reconstruction, analysis, and visualization of phylogenomic data. Mol Biol Evol. 33:1635–1638. https://doi.org/10.1093/molbev/msw046
Lee MD, Ponty Y (2019) GToTree: A user-friendly workflow for phylogenomics. Bioinformatics. 35:4162–4164. https://doi.org/10.1093/bioinformatics/btz188
Eddy SR (2011) Accelerated profile HMM searches. PLoS Comput Biol. 7:e1002195. https://doi.org/10.1371/journal.pcbi.1002195
Edgar RC (2004) MUSCLE: Multiple sequence alignment with high accuracy and high throughput. Nucleic Acids Res. 32:1792–1797. https://doi.org/10.1093/nar/gkh340
Capella-Gutiérrez S, Silla-Martínez JM, Gabaldón T (2009) trimAl: a tool for automated alignment trimming in large-scale phylogenetic analyses. Bioinformatics. 25:1972–1973. https://doi.org/10.1093/bioinformatics/btp348
Tamura K, Stecher G, Peterson D et al (2013) MEGA6: molecular evolutionary genetics analysis version 6.0. Mol Biol Evol. https://doi.org/10.1093/molbev/mst197
Tange O (2011) GNU parallel: the command-line power tool. ;login USENIX Mag. https://doi.org/10.5281/zenodo.16303
Altschul SF, Madden TL, Schäffer AA et al (1997) Gapped BLAST and PSI-BLAST: a new generation of protein database search programs. Nucleic Acids Res 25:3389–3402
Hildebrand F, Meyer A, Eyre-Walker A (2010) Evidence of selection upon genomic GC-content in bacteria. PLoS Genet 6:e1001107
Hershberg R, Petrov DA (2010) Evidence that mutation is universally biased towards AT in bacteria. PLoS Genet 6:e1001115
Berube PM, Rasmussen A, Braakman R, Stepanauskas R, Chisholm SW (2019) Emergence of trait variability through the lens of nitrogen assimilation in Prochlorococcus. Elife 8:1–28. https://doi.org/10.7554/elife.41043
Schirrmeister BE, Antonelli A, Bagheri HC (2011) The origin of multicellularity in cyanobacteria. BMC Evol Biol 11:45
Di Rienzi SC, Sharon I, Wrighton KC et al (2013) The human gut and groundwater harbor non-photosynthetic bacteria belonging to a new candidate phylum sibling to Cyanobacteria. Elife 2:e01102
Soo RM, Skennerton CT, Sekiguchi Y, Imelfort M, Paech SJ, Dennis PG, Steen JA, Parks DH, Tyson GW, Hugenholtz P (2014) An expanded genomic representation of the phylum Cyanobacteria. Genome Biol Evol 6:1031–1045
Soo RM (2015) In search of non-photosynthetic Cyanobacteria. Univ Queensl
Cohan FM (2002) What are bacterial species? Annu Rev Microbiol 56:457–487
Cohan FM (2016) Bacterial speciation: genetic sweeps in bacterial species. Curr Biol 26:R112–R115. https://doi.org/10.1016/j.cub.2015.10.022
Cohan FM, Kopac SM (2017) A theory-based pragmatism for discovering and classifying newly divergent species of bacterial pathogens. Genet Evol Infect Dis:25
Baumann L, Baumann P, Mandel M et al (1972) Taxonomy of aerobic marine. Microbiology 110:402–429
Deming JW, Somers LK, Straube WL, Swartz DG, Macdonell MT (1988) Isolation of an obligately barophilic bacterium and description of a new genus, Colwellia gen. nov. Syst Appl Microbiol 10:152–160. https://doi.org/10.1016/S0723-2020(88)80030-4
Kurahashi M, Yokota A (2007) Endozoicomonas elysicola gen. nov., sp. nov., a γ-proteobacterium isolated from the sea slug Elysia ornata. Syst Appl Microbiol 30:202–206. https://doi.org/10.1016/j.syapm.2006.07.003
GAUTHIER MJ, LAFAY B, CHRISTEN BR et al (1992) Marinobacter hydrocarbonoclasticus gen. nov,, sp. nov., a new, extremely halotolerant, hydrocarbon-degrading marine bacterium. Int J Syst Bacteriol 42:568–576. https://doi.org/10.1373/49.4.644
GONZALEZ JM, MAYER F, MORAN MA et al (2009) Microbulbifer hydrolyticus gen. nov., sp. nov., and Marinobacterium georgiense gen. nov., sp. nov., two marine bacteria from a lignin-rich pulp mill waste enrichment community. Int J Syst Bacteriol 47:369–376. https://doi.org/10.1099/00207713-47-2-369
LANDSCHOOT A VAN, LEY J DE (1983) Intra- and intergeneric similarities of the rRNA cistrons of Alteromonas, Murimmonas (gen. nov.) and some other Gram-negative bacteria. J Gen Microbiol 129:3057–3074
Gauthier G, Gauthier M, R C (1995) Phylogenetic analysis of the genera Alteromonas, Shewanella, and Moritella using genes coding for small-subunit rRNA sequences and division of the genus Alteromonas into two genera, Alteromonas (Emended) and Pseudoalteromonas gen. nov., and proposal of Tw. Int J Syst Bacteriol 45:755–761
Shieh WY, Te Lin Y, Jean WD (2004) Pseudovibrio denitrificans gen. nov., sp. nov., a marine, facultatively anaerobic, fermentative bacterium capable of denitrification. Int J Syst Evol Microbiol 54:2307–2312. https://doi.org/10.1099/ijs.0.63107-0
Shiba T (1991) Roseobacter litoralis gen. nov., sp. nov., and Roseobacter denitrificans sp. nov., aerobic pink-pigmented bacteria which contain bacteriochlorophyll a. Syst Appl Microbiol 14:140–145. https://doi.org/10.1016/S0723-2020(11)80292-4
Uchino Y, Hirata A, Yokota A, Sugiyama J (2005) Reclassification of marine Agrobacterium species: proposals of Stappia stellulata gen. nov., comb. nov., Stappia aggregata sp. nov., nom. rev., Ruegeria atlantica gen. nov., comb. nov., Ruegeria gelatinovora comb. nov., Ruegeria algicola comb. nov., and A. J Gen Appl Microbiol 44:201–210. https://doi.org/10.2323/jgam.44.201
MacDonell MT, Colwell RR (1985) Phylogeny of the Vibrionaceae, and recommendation for two new genera, Listonella and Shewanella. Syst Appl Microbiol 6:171–182. https://doi.org/10.1016/S0723-2020(85)80051-5
Maldonado LA, Fenical W, Jensen PR, Kauffman CA, Mincer TJ, Ward AC, Bull AT, Goodfellow M (2005) Salinispora arenicola gen. nov., sp. nov. and Salinispora tropica sp. nov., obligate marine actinomycetes belonging to the family Micromonosporaceae. Int J Syst Evol Microbiol 55:1759–1766. https://doi.org/10.1099/ijs.0.63625-0
Waksman SA, Henrici AT (1895) The nomenclature and classification of the actinomycetes. J Am Med Assoc XXIV:452. https://doi.org/10.1001/jama.1895.02430120030007
Macia MC, Ludwig W, Schleifer KH et al (2001) Thalassomonas viridans gen . nov ., sp . nov ., a novel marine γ-proteobacterium. Int J Syst Evol Microbiol 51:1283–1289
Pacini F (1854) Osservazione microscopiche e deduzioni patologiche sul cholera asiatico. Gaz Medicale Ital Toscano Firenze 6:405–4012
Inagaki F, Takai K, Nealson KH, Horikoshi K (2004) Sulfurovum lithotrophicum gen. nov., sp. nov., a novel sulfur-oxidizing chemolithoautotroph within the E-Proteobacteria isolated from Okinawa Trough hydrothermal sediments. Int J Syst Evol Microbiol 54:1477–1482. https://doi.org/10.1099/ijs.0.03042-0
Inagaki F, Takai K, Kobayashi H et al (2003) Sulfurimonas autrotrophica gen. nov., sp. nov., a novel sulfur-oxidizing E-proteobacterium isolated from hydrothermal sediments in the Mid-Okinawa Trough. Int J Syst Evol Microbiol 53:1801–1805. https://doi.org/10.1099/ijs.0.02682-0
Luo H, Moran MA (2014) Evolutionary ecology of the marine Roseobacter clade. Microbiol Mol Biol Rev 78:573–587. https://doi.org/10.1128/MMBR.00020-14
Barka EA, Vatsa P, Sanchez L et al (2016) Taxonomy, physiology, and natural products of Actinobacteria. Am Soc Microbiol 80:1–43. https://doi.org/10.1128/MMBR.00019-15.Address
Clark K, Karsch-Mizrachi I, Lipman DJ et al (2015) GenBank. Nucleic Acids Res 44:D67–D72
Kauff F, Büdel B (2010) Phylogeny of cyanobacteria: an overview. In: Progress in botany 72. Springer, pp 209–224
STANIER RY, SISTROM WR, HANSEN TA et al (1978) Proposal to place the nomenclature of the Cyanobacteria (blue-green algae) under the rules of the International Code of Nomenclature of Bacteria. Int J Syst Evol Microbiol 28:335–336. https://doi.org/10.1099/00207713-28-2-335
Oren A (2004) A proposal for further integration of the cyanobacteria under the Bacteriological Code. Int J Syst Evol Microbiol 54:1895–1902. https://doi.org/10.1099/ijs.0.03008-0
Oren A, Garrity GM (2014) Then and now: a systematic review of the systematics of prokaryotes in the last 80 years. Antonie Van Leeuwenhoek 106:43–56
Tindall BJ (1999) Note: Proposals to update and make changes to the Bacteriological Code. Int J Syst Evol Microbiol 49:1309–1312
De Vos P, Trüper HG (2000) Judicial commission of the international committee on systematic bacteriology; IXth international (IUMS) congress of bacteriology and applied microbiology. Int J Syst Evol Microbiol 50:2239–2244
Labeda DP (2000) International committee on systematic bacteriology; IXth international (IUMS) congress of bacteriology and applied microbiology. Int J Syst Evol Microbiol 50:2245–2247
Hoffmann L, Komárek J, Kaštovsky J (2005) System of cyanoprokaryotes (cyanobacteria)--state in 2004. Arch Hydrobiol Suppl Algol Stud 117:95–115
Oren A, Tindall BJ (2005) Nomenclature of the cyanophyta/cyanobacteria/cyanoprokaryotes under the International Code of Nomenclature of Prokaryotes. Arch Hydrobiol Suppl Algol Stud 117:39–52
Oren A, Komárek J, Hoffmann L (2009) Nomenclature of the Cyanophyta/Cyanobacteria/Cyanoprokaryotes--what has happened since IAC Luxembourg? Arch Hydrobiol Suppl Algol Stud 130:17–26
Oren A, Ventura S (2017) The current status of cyanobacterial nomenclature under the “prokaryotic” and the “botanical” code. Antonie Van Leeuwenhoek 110:1257–1269
Parte AC (2014) LPSN--list of prokaryotic names with standing in nomenclature. Nucleic Acids Res 42:D613–D616. https://doi.org/10.1093/nar/gkt1111
Gaget V, Welker M, Rippka R, de Marsac NT (2015) A polyphasic approach leading to the revision of the genus Planktothrix (Cyanobacteria) and its type species, P. agardhii, and proposal for integrating the emended valid botanical taxa, as well as three new species, Planktothrix paucivesiculata sp. nov. I. Syst Appl Microbiol 38:141–158
Gaget V, Welker M, Rippka R, de Marsac TN (2015) Response to: “Comments on:” A polyphasic approach leading to the revision of the genus Planktothrix (Cyanobacteria) and its type species, P. agardhii, and proposal for integrating the emended valid botanical taxa, as well as three new species, Planktothri. Syst Appl Microbiol 38:368–370
Boone DR, Castenholz RW (2001) The Archaea and the deeply branching and phototrophic bacteria. In: Garrity G, Boone DR, Castenholz RW (eds) Bergey’s manual of systematic bacteriology volume one. Springer-Verlag, New York, pp 473–487
Rippka R, Deruelles J, Waterbury JB, et al (1979) Generic assignments, strain histories and properties of pure cultures of cyanobacteria. Microbiology 111:1–61, 1
Gugger MF, Hoffmann L (2004) Polyphyly of true branching cyanobacteria (Stigonematales). Int J Syst Evol Microbiol 54:349–357
Hugenholtz P, Skarshewski A, Parks DH (2016) Genome-based microbial taxonomy coming of age. Cold Spring Harb Perspect Biol 8:a018085
Shih PM, Wu D, Latifi A, Axen SD, Fewer DP, Talla E, Calteau A, Cai F, Tandeau de Marsac N, Rippka R, Herdman M, Sivonen K, Coursin T, Laurent T, Goodwin L, Nolan M, Davenport KW, Han CS, Rubin EM, Eisen JA, Woyke T, Gugger M, Kerfeld CA (2013) Improving the coverage of the cyanobacterial phylum using diversity-driven genome sequencing. Proc Natl Acad Sci 110:1053–1058. https://doi.org/10.1073/pnas.1217107110
Komarek J, Kastovsky J, Mares J, Johansen JR (2014) Taxonomic classification of cyanoprokaryotes (cyanobacterial genera) 2014, using a polyphasic approach. Preslia 86:295–335
Kozlov AM, Zhang J, Yilmaz P, Glöckner FO, Stamatakis A (2016) Phylogeny-aware identification and correction of taxonomically mislabeled sequences. Nucleic Acids Res 44:5022–5033
Choudoir MJ, Campbell AN, Buckley DH (2012) Grappling with Proteus: population level approaches to understanding microbial diversity. Front Microbiol 3:336
Becraft ED, Wood JM, Rusch DB et al (2015) The molecular dimension of microbial species: 1. Ecological distinctions among, and homogeneity within, putative ecotypes of Synechococcus inhabiting the cyanobacterial mat of Mushroom Spring, Yellowstone National Park. Front Microbiol 6:590. https://doi.org/10.3389/fmicb.2015.00590
Farrant GK, Doré H, Cornejo-Castillo FM, Partensky F, Ratin M, Ostrowski M, Pitt FD, Wincker P, Scanlan DJ, Iudicone D, Acinas SG, Garczarek L (2016) Delineating ecologically significant taxonomic units from global patterns of marine picocyanobacteria. Proc Natl Acad Sci 113:E3365–E3374. https://doi.org/10.1073/pnas.1524865113
Dufresne A, Salanoubat M, Partensky F, Artiguenave F, Axmann IM, Barbe V, Duprat S, Galperin MY, Koonin EV, le Gall F, Makarova KS, Ostrowski M, Oztas S, Robert C, Rogozin IB, Scanlan DJ, de Marsac NT, Weissenbach J, Wincker P, Wolf YI, Hess WR (2003) Genome sequence of the cyanobacterium Prochlorococcus marinus SS120, a nearly minimal oxyphototrophic genome. Proc Natl Acad Sci 100:10020–10025
Kettler GC, Martiny AC, Huang K, Zucker J, Coleman ML, Rodrigue S, Chen F, Lapidus A, Ferriera S, Johnson J, Steglich C, Church GM, Richardson P, Chisholm SW (2007) Patterns and implications of gene gain and loss in the evolution of Prochlorococcus. PLoS Genet 3:2515–2528. https://doi.org/10.1371/journal.pgen.0030231
Zinser ER, Lindell D, Johnson ZI, Futschik ME, Steglich C, Coleman ML, Wright MA, Rector T, Steen R, McNulty N, Thompson LR, Chisholm SW (2009) Choreography of the transcriptome, photophysiology, and cell cycle of a minimal photoautotroph, Prochlorococcus. PLoS One 4:e5135
García-Fernandez JM, Marsac NT, Diez J (2004) Streamlined regulation and gene loss as adaptive mechanisms in prochlorococcus for optimized nitrogen utilization in oligotrophic environments. Microbiol Mol Biol Rev 68:630–638. https://doi.org/10.1128/MMBR.68.4.630
Parks DH, Chuvochina M, Chaumeil P-A, Rinke C, Mussig AJ, Hugenholtz P (2020) A complete domain-to-species taxonomy for Bacteria and Archaea. Nat Biotechnol. https://doi.org/10.1038/s41587-020-0501-8
Chaumeil P-A, Mussig AJ, Hugenholtz P, Parks DH (2019) GTDB-Tk: a toolkit to classify genomes with the Genome Taxonomy Database. Bioinformatics. https://doi.org/10.1093/bioinformatics/btz848
Chisholm SW, Frankel SL, Goericke R, Olson RJ, Palenik B, Waterbury JB, West-Johnsrud L, Zettler ER (1992) Prochlorococcus marinus nov. gen. nov. sp.: an oxyphototrophic marine prokaryote containing divinyl chlorophyll a and b. Arch Microbiol 157:297–300
Giovannoni SJ, Halsey KH, Saw J, Muslin O, Suffridge CP, Sun J, Lee CP, Moore ER, Temperton B, Noell SE (2019) A parasitic arsenic cycle that shuttles energy from phytoplankton to heterotrophic bacterioplankton. MBio 10
Saunders JK, Rocap G (2016) Genomic potential for arsenic efflux and methylation varies among global Prochlorococcus populations. ISME J 10:197–209. https://doi.org/10.1038/ismej.2015.85
Acknowledgments
The authors thank CNPq, CAPES and FAPERJ.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Tschoeke, D., Salazar, V.W., Vidal, L. et al. Unlocking the Genomic Taxonomy of the Prochlorococcus Collective. Microb Ecol 80, 546–558 (2020). https://doi.org/10.1007/s00248-020-01526-5
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00248-020-01526-5