Abstract
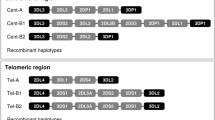
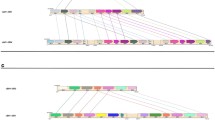
KIR2DL4 is an important immune modulator expressed in natural killer cells; HLA-G is its main ligand. We have characterized the KIR2DL4 genetic diversity by considering the promoter, all exons, and all introns in a highly admixed Brazilian population sample and by using massively parallel sequencing. We introduce a molecular method to amplify and to sequence the complete KIR2DL4 gene. To avoid the mapping bias and genotype errors commonly observed in gene families, we have developed and validated a bioinformatic pipeline designed to minimize these errors and applied it to survey the variability of 220 individuals from the State of São Paulo, southeastern Brazil. We have also compared the KIR2DL4 genetic diversity in the Brazilian cohort with the diversity previously reported by the 1000Genomes consortium. KIR2DL4 presents high linkage disequilibrium throughout the gene, with coding sequences associated with specific promoters. There are few but divergent promoter haplotypes. We have also detected many new KIR2DL4 sequences, all bearing nucleotide exchanges in introns and encoding previously described proteins. Exons 3 and 4, which encode the external domains, are the most variable. The ancestry background influences the KIR2DL4 allele frequencies and must be considered for association studies regarding KIR2DL4.

Similar content being viewed by others
Data availability
The VCF file with phased genotypes is available upon request.
Code availability
(Software application or custom code): hla-mapper (www.castelli-lab.net/apps/hla-mapper/), vcfx (www.castelli-lab.net/apps/vcfx), phasex (available upon request); the GATK package is provided by the Broad Institute (https://gatk.broadinstitute.org).
References
Alicata C, Ashouri E, Nemat-Gorgani N et al (2020) KIR variation in Iranians combines high haplotype and allotype diversity with an abundance of functional inhibitory receptors. Front Immunol 11 https://doi.org/10.3389/fimmu.2020.00556
Auton A, Abecasis GR, Altshuler DM et al (2015) A global reference for human genetic variation. Nature 526:68–74. https://doi.org/10.1038/nature15393
Barrett JC, Fry B, Maller J, Daly MJ (2005) Haploview: analysis and visualization of LD and haplotype maps. Bioinformatics 21:263–265. https://doi.org/10.1093/bioinformatics/bth457
Benson DA, Cavanaugh M, Clark K et al (2018) GenBank. Nucleic Acids Res 46:D41–D47. https://doi.org/10.1093/nar/gkx1094
Boyington JC, Sun PD (2002) A structural perspective on MHC class I recognition by killer cell immunoglobulin-like receptors. Mol Immunol 38:1007–1021. https://doi.org/10.1016/S0161-5890(02)00030-5
Brandt DYC, Aguiar VRC, Bitarello BD et al (2015) Mapping bias overestimates reference allele frequencies at the HLA genes in the 1000 genomes project phase I data. G3 Genes. Genomes, Genet 5:931–941. https://doi.org/10.1534/g3.114.015784
Browning BL, Zhou Y, Browning SR (2018) A one-penny imputed genome from next-generation reference panels. Am J Hum Genet 103:338–348. https://doi.org/10.1016/j.ajhg.2018.07.015
Bruijnesteijn J, van der Wiel MKH, de Groot N et al (2018) Extensive alternative splicing of KIR transcripts. Front Immunol 9:2846. https://doi.org/10.3389/fimmu.2018.02846
Brusilovsky M, Cordoba M, Rosental B et al (2013) Genome-wide siRNA screen reveals a new cellular partner of NK cell receptor KIR2DL4: heparan sulfate directly modulates KIR2DL4-mediated Responses. J Immunol 191:5256–5267. https://doi.org/10.4049/jimmunol.1302079
Buhler S, Di Cristofaro J, Frassati C et al (2009) High levels of molecular polymorphism at the KIR2DL4 locus in French and Congolese populations: impact for anthropology and clinical studies. Hum Immunol 70:953–959. https://doi.org/10.1016/j.humimm.2009.08.002
Bukur J, Jasinski S, Seliger B (2012) The role of classical and non-classical HLA class I antigens in human tumors. Semin Cancer Biol 22:350–358. https://doi.org/10.1016/j.semcancer.2012.03.003
Castelli EC, Paz MA, Souza AS et al (2018) Hla-mapper: an application to optimize the mapping of HLA sequences produced by massively parallel sequencing procedures. Hum Immunol 79:678–684. https://doi.org/10.1016/j.humimm.2018.06.010
Castelli EC, Ramalho J, Porto IOP et al (2014) Insights into HLA-G genetics provided by worldwide haplotype diversity. Front Immunol 5 https://doi.org/10.3389/fimmu.2014.00476
Cirulli V, Zalatan J, McMaster M et al (2006) The class I HLA repertoire of pancreatic islets comprises the nonclassical class Ib antigen HLA-G. Diabetes 55:1214–1222. https://doi.org/10.2337/db05-0731
Clarke L, Fairley S, Zheng-Bradley X et al (2017) The international Genome sample resource (IGSR): a worldwide collection of genome variation incorporating the 1000 Genomes Project data. Nucleic Acids Res 45:D854–D859. https://doi.org/10.1093/nar/gkw829
Delaneau O, Zagury JF, Robinson MR et al (2019) Accurate, scalable and integrative haplotype estimation. Nat Commun 10:24–29. https://doi.org/10.1038/s41467-019-13225-y
Excoffier L, Laval G, Schneider S (2005) Arlequin (version 3.0): an integrated software package for population genetics data analysis. Evol Bioinforma 1:117693430500100. https://doi.org/10.1177/117693430500100003
Faure M, Long EO (2002) KIR2DL4 (CD158d), an NK cell-activating receptor with inhibitory potential. J Immunol 168:6208–6214. https://doi.org/10.4049/jimmunol.168.12.6208
Fondevila M, Phillips C, Santos C et al (2013) Revision of the SNPforID 34-plex forensic ancestry test: assay enhancements, standard reference sample genotypes and extended population studies. Forensic Sci Int Genet 7:63–74. https://doi.org/10.1016/j.fsigen.2012.06.007
Gedil MA, Steiner NK, Hurley CK (2005) Genomic characterization of KIR2DL4 in families and unrelated individuals reveals extensive diversity in exon and intron sequences including a common frameshift variation occurring in several alleles. Tissue Antigens 65:402–418. https://doi.org/10.1111/j.1399-0039.2005.00380.x
Gendzekhadze K, Norman PJ, Abi-Rached L et al (2009) Co-evolution of KIR2DL3 with HLA-C in a human population retaining minimal essential diversity of KIR and HLA class I ligands. Proc Natl Acad Sci U S A 106:18692–18697. https://doi.org/10.1073/pnas.0906051106
Gömez-Lozano N, de Pablo R, Puente S, Vilches C (2003) Recognition of HLA-G by the NK cell receptor KIR2DL4 is not essential for human reproduction. Eur J Immunol 33:639–644. https://doi.org/10.1002/eji.200323741
Goodridge JP, Lathbury LJ, John E et al (2009) The genotype of the NK cell receptor, KIR2DL4, influences INFγ secretion by decidual natural killer cells. Mol Hum Reprod 15:489–497. https://doi.org/10.1093/molehr/gap039
Goodridge JP, Witt CS, Christiansen FT, Warren HS (2003) KIR2DL4 (CD158d) genotype influences expression and function in NK cells. J Immunol 171:1768–1774. https://doi.org/10.4049/jimmunol.171.4.1768
Goris A, Dobosi R, Boonen S et al (2009) KIR2DL4 (CD158d) polymorphisms and susceptibility to multiple sclerosis. J Neuroimmunol 210:113–115. https://doi.org/10.1016/j.jneuroim.2009.03.001
Guethlein LA, Flodin LR, Adams EJ, Parham P (2002) NK cell receptors of the orangutan ( Pongo pygmaeus ): a pivotal species for tracking the coevolution of killer cell Ig-Like receptors with MHC-C. J Immunol 169:220–229. https://doi.org/10.4049/jimmunol.169.1.220
Hershberger KL, Shyam R, Miura A, Letvin NL (2001) Diversity of the killer cell Ig-like receptors of rhesus monkeys. J Immunol 166:4380–4390. https://doi.org/10.4049/jimmunol.166.7.4380
Hò GGT, Celik AA, Huyton T et al (2020) NKG2A/CD94 is a new immune receptor for HLA-G and distinguishes amino acid differences in the HLA-G Heavy chain. Int J Mol Sci. https://doi.org/10.3390/ijms21124362
Jiang W, Johnson C, Jayaraman J et al (2012) Copy number variation leads to considerable diversity for B but not A haplotypes of the human KIR genes encoding NK cell receptors. Genome Res 22:1845–1854. https://doi.org/10.1101/gr.137976.112
Jones DC, Edgar RS, Ahmad T et al (2006) Killer Ig-like receptor (KIR) genotype and HLA ligand combinations in ulcerative colitis susceptibility. Genes Immun 7:576–582. https://doi.org/10.1038/sj.gene.6364333
Jurisicova A, Casper RF, Maclusky NJ et al (1996) HLA-G expression during preimplantation human embryo development. Proc Natl Acad Sci U S A 93:161–165. https://doi.org/10.1073/pnas.93.1.161
Kidd JM, Sharpton TJ, Bobo D, et al (2014) Exome capture from saliva produces high quality genomic and metagenomic data BMC. Genomics 15 https://doi.org/10.1186/1471-2164-15-262
Kikuchi-Maki A, Yusa S, Catina TL, Campbell KS (2003) KIR2DL4 Is an IL-2-regulated NK cell receptor that exhibits limited expression in humans but triggers strong IFN-γ production. J Immunol 171:3415–3425. https://doi.org/10.4049/jimmunol.171.7.3415
Kumar S (2018) Natural killer cell cytotoxicity and its regulation by inhibitory receptors. Immunology 154:383–393. https://doi.org/10.1111/imm.12921
Lanier LL, Cortiss BC, Wu J et al (1998) Immunoreceptor DAP12 bearing a tyrosine-based activation motif is involved in activating NK cells. Nature. https://doi.org/10.1038/35642
Le Discorde M, Moreau P, Sabatier P et al (2003) Expression of HLA-G in human cornea, an immune-privileged tissue. Hum Immunol 64:1039–1044. https://doi.org/10.1016/j.humimm.2003.08.346
Li H, Wright PW, McCullen M, Anderson SK (2016) Characterization of KIR intermediate promoters reveals four promoter types associated with distinct expression patterns of KIR subtypes. Genes Immun. https://doi.org/10.1038/gene.2015.56
Mallet V, Blaschitz A, Crisa L et al (1999) HLA-G in the human thymus: a subpopulation of medullary epithelial but not CD83+ dendritic cells expresses HLA-G as a membrane-bound and soluble protein. Int Immunol 11:889–898. https://doi.org/10.1093/intimm/11.6.889
Martin AM, Freitas EM, Witt CS, Christiansen FT (2000) The genomic organization and evolution of the natural killer immunoglobulin-like receptor (KIR) gene cluster. Immunogenetics 51:268–280. https://doi.org/10.1007/s002510050620
Nemat-Gorgani N, Guethlein LA, Henn BM et al (2019) Diversity of KIR, HLA class I, and their interactions in seven populations of sub-Saharan Africans. J Immunol 202:2636–2647. https://doi.org/10.4049/jimmunol.1801586
Norman PJ, Abi-Rached L, Gendzekhadze K et al (2009) Meiotic recombination generates rich diversity in NK cell receptor genes, alleles, and haplotypes. Genome Res 19:757–769. https://doi.org/10.1101/gr.085738.108
Norman PJ, Hollenbach JA, Nemat-Gorgani N et al (2016) Defining KIR and HLA class I genotypes at highest resolution via high-throughput sequencing. Am J Hum Genet 99:375–391. https://doi.org/10.1016/j.ajhg.2016.06.023
Nowak I, Majorczyk E, Ploski R et al (2011) Lack of KIR2DL4 gene in a fertile Caucasian woman. Tissue Antigens 78:115–119. https://doi.org/10.1111/j.1399-0039.2011.01711.x
Nowak I, Malinowski A, Barcz E et al (2016) Possible role of HLA-G, LILRB1 and KIR2DL4 gene polymorphisms in spontaneous miscarriage. Arch Immunol Ther Exp (Warsz) 64:505–514. https://doi.org/10.1007/s00005-016-0389-7
Parham P (2005) MHC class I molecules and KIRS in human history, health and survival. Nat Rev Immunol 5:201–214. https://doi.org/10.1038/nri1570
Pende D, Falco M, Vitale M et al (2019) Killer Ig-like receptors (KIRs): their role in NK cell modulation and developments leading to their clinical exploitation. Front Immunol 10 https://doi.org/10.3389/fimmu.2019.01179
Posth C, Nakatsuka N, Lazaridis I et al (2018) Reconstructing the deep population history of Central and South America. Cell 175:1185-1197.e22. https://doi.org/10.1016/j.cell.2018.10.027
Pritchard JK, Stephens M, Donnelly P (2000) Inference of population structure using multilocus genotype data. Genetics 155:945–959
Purdy AK, Campbell KS (2009) Natural killer cells and cancer: regulation by the killer cell ig-like receptors (KIR). Cancer Biol Ther 8:2209–2218. https://doi.org/10.4161/cbt.8.23.10455
Rajagopalan S, Bryceson YT, Kuppusamy SP et al (2006) Activation of NK cells by an endocytosed receptor for soluble HLA-G. PLoS Biol 4:0070–0086. https://doi.org/10.1371/journal.pbio.0040009
Rajagopalan S, Long EO (2012) KIR2DL4 (CD158d): an activation receptor for HLA-G. Front Immunol 3:1–6. https://doi.org/10.3389/fimmu.2012.00258
Rice P, Longden L, Bleasby A (2000) EMBOSS: the European Molecular Biology Open Software Suite. Trends Genet 16:276–277. https://doi.org/10.1016/S0168-9525(00)02024-2
Rizzo R, Fainardi E, Rouas-Freiss N, Morandi F (2017) The role of HLA-Class Ib molecules in immune-related diseases, tumors, and infections 2016. J Immunol Res 2017 https://doi.org/10.1155/2017/2309574
Robinson J, Halliwell JA, Hayhurst JD et al (2015) The IPD and IMGT/HLA database: allele variant databases. Nucleic Acids Res 43:D423–D431. https://doi.org/10.1093/nar/gku1161
Sonon P, Sadissou I, Tokplonou L et al (2018) HLA-G, -E and -F regulatory and coding region variability and haplotypes in the Beninese Toffin population sample. Mol Immunol 104:108–127. https://doi.org/10.1016/j.molimm.2018.08.016
Souza AS, Sonon P, Paz MA et al (2020) Hla-C genetic diversity and evolutionary insights in two samples from Brazil and Benin. Hla 96:468–486. https://doi.org/10.1111/tan.13996
Tan CY, Chong YS, Loganath A et al (2009) Possible gene-gene interaction of KIR2DL4 with its cognate ligand HLA-G in modulating risk for preeclampsia. Reprod Sci 16:1135–1143. https://doi.org/10.1177/1933719109342280
Trompeter H-I, Gómez-Lozano N, Santourlidis S et al (2005) Three structurally and functionally divergent kinds of promoters regulate expression of clonally distributed killer cell Ig-Like Receptors ( KIR ), of KIR2DL4, and of KIR3DL3. J Immunol 174:4135–4143. https://doi.org/10.4049/jimmunol.174.7.4135
Trowsdale J, Barten R, Haude A et al (2001) The genomic context of natural killer receptor extended gene families. Immunol Rev 181:20–38. https://doi.org/10.1034/j.1600-065X.2001.1810102.x
Valiante NM, Uhrberg M, Shilling HG et al (1997) Functionally and structurally distinct NK cell receptor repertoires in the peripheral blood of two human donors. Immunity 7:739–751
Van der Auwera GA, Carneiro MO, Hartl C et al (2013) From fastQ data to high-confidence variant calls: the genome analysis toolkit best practices pipeline. Curr Protoc Bioinforma. https://doi.org/10.1002/0471250953.bi1110s43
van der Veken LT, Diez Campelo M, van der Hoorn MAWG et al (2009) Functional analysis of killer Ig-like receptor-expressing cytomegalovirus-specific CD8 + T cells. J Immunol 182:92–101. https://doi.org/10.4049/jimmunol.182.1.92
Vendelbosch S, de Boer M, Gouw RATW et al (2013) Extensive variation in gene copy number at the killer immunoglobulin-like receptor locus in humans. PLoS ONE 8:4–13. https://doi.org/10.1371/journal.pone.0067619
Vilches C, Parham P (2002) KIR: diverse, rapidly evolving receptors of innate and adaptive immunity. Annu Rev Immunol 20:217–251. https://doi.org/10.1146/annurev.immunol.20.092501.134942
Vivier E, Daëron M (1997) Immunoreceptor tyrosine-based inhibition motifs. Immunol Today 18:286–291. https://doi.org/10.1016/S0167-5699(97)80025-4
Vivier E, Tomasello E, Baratin M et al (2008) Functions of natural killer cells. Nat Immunol 9:503–510. https://doi.org/10.1038/ni1582
Wagner I, Schefzyk D, Pruschke J et al (2018) Allele-level KIR genotyping of more than a million samples: workflow, algorithm, and observations. Front Immunol 9:2843. https://doi.org/10.3389/fimmu.2018.02843
Yawata M, Yawata N, Draghi M et al (2006) Roles for HLA and KIR polymorphisms in natural killer cell repertoire selection and modulation of effector function. J Exp Med 203:633–645. https://doi.org/10.1084/jem.20051884
Zhu FM, Jiang K, Lv QF et al (2006) Investigation of killer cell immunoglobulin-like receptor KIR2DL4 diversity by sequence-based typing in Chinese population. Tissue Antigens 67:214–221. https://doi.org/10.1111/j.1399-0039.2006.00562.x
Acknowledgements
We also thank Dra. Cynthia Maria de Campos Prado Manso for language editing.
Funding
This work was supported by Fundação de Amparo à Pesquisa do Estado de São—FAPESP/Brazil (Grants 2017/19223-0 and 2017/05042-4). This study was partially funded by the Coordenação de Aperfeiçoamento de Pessoal de Nível Superior (CAPES)—Finance Code 001.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Ethics approval and consent to participate
The Human Research Ethics Committee of the School of Medicine/Unesp has approved the study protocol (Protocol 24157413.7.0000.5411). All participants signed an informed consent before blood collection.
Conflict of interest
The authors declare that they have no conflict of interest.
Supplementary Information
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Weiss, E., Andrade, H.S., Lara, J.R. et al. KIR2DL4 genetic diversity in a Brazilian population sample: implications for transcription regulation and protein diversity in samples with different ancestry backgrounds. Immunogenetics 73, 227–241 (2021). https://doi.org/10.1007/s00251-021-01206-9
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00251-021-01206-9