Abstract
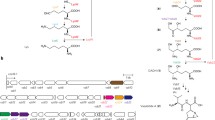
Based on the exploration of data generated by genome sequencing, a bioinformatics approach has been chosen to identify the biosynthetic pathway of the siderophores produced by Aeromonas species. The amonabactins, considered as a virulence factor, represent a family of four variants of catechol peptidic siderophores containing Dhb, Lys, Gly, and an aromatic residue either Trp or Phe in a D-configuration. The synthesis operon is constituted of seven genes named amoCEBFAGH and is iron-regulated. The cluster includes genes encoding proteins involved in the synthesis and incorporation of the Dhb monomer, and genes encoding specific nonribosomal peptide synthetases, which are responsible for the building of the peptidic moiety. The amonabactin assembly line displays a still so far not described atypical mode of synthesis that is iterative, alternative, and optional. A disruption mutant in the adenylation domain of AmoG was unable to synthesize any amonabactin and to grow in iron stress conditions while a deletion of amoH resulted in the production of only two over the four forms. The amo cluster is widespread among most of the Aeromonas species, only few species produces the enterobactin siderophore.





Similar content being viewed by others
References
Anand S, Prasad MVR, Yadav G, Kumar N, Shehara J, Ansari MZ, Mohanty D (2010) SBSPKS: structure based sequence analysis of polyketide synthases. Nucleic Acids Res 38:W487–W496. doi:10.1093/nar/gkq340
Austin B, Austin DA (2007) Bacterial fish pathogens: diseases of farmed and wild fish, 4th edn. Praxis Publishing Ltd, Chichester
Bachmann BO, Ravel J (2009) Chapter 8. Methods for in silico prediction of microbial polyketide and nonribosomal peptide biosynthetic pathways from DNA sequence data. Meth Enzymol 458:181–217
Balado M, Souto A, Vences A, Careaga VP, Valderrama K, Segade Y, Rodríguez J, Osorio CR, Jiménez C, Lemos ML (2015) Two catechol siderophores, acinetobactin and amonabactin, are simultaneously produced by Aeromonas salmonicida subsp. salmonicida sharing part of the biosynthetic pathway. ACS Chem Biol 10(12):2850–2860. doi:10.1021/acschembio.5b00624
Barghouthi S, Young R, Arceneaux JEL, Byers BR (1989a) Physiological control of amonabactin biosynthesis in Aeromonas hydrophila. Biol Met 2(3):155–160. doi:10.1007/bf01142554
Barghouthi S, Young R, Olson MO, Arceneaux JE, Clem LW, Byers BR (1989b) Amonabactin, a novel tryptophan- or phenylalanine-containing phenolate siderophore in Aeromonas hydrophila. J Bacteriol 171(4):1811–1816
Barghouthi S, Payne SM, Arceneaux JE, Byers BR (1991) Cloning, mutagenesis, and nucleotide sequence of a siderophore biosynthetic gene (amoA) from Aeromonas hydrophila. J Bacteriol 173(16):5121–5128
Beaz-Hidalgo R, Figueras MJ (2013) Aeromonas spp. whole genomes and virulence factors implicated in fish disease. J Fish Dis 36(4):371–388. doi:10.1111/jfd.12025
Caboche S, Leclère V, Pupin M, Kucherov G, Jacques P (2010) Diversity of monomers in nonribosomal peptides: towards the prediction of origin and biological activity. J Bacteriol 192(19):5143–5150. doi:10.1128/jb.00315-10
Caradec T, Pupin M, Vanvlassenbroeck A, Devignes M-D, Smaïl-Tabbone M, Jacques P, Leclère V (2014) Prediction of monomer isomery in Florine: a workflow dedicated to nonribosomal peptide discovery. PLoS One 9(1):e85667. doi:10.1371/journal.pone.0085667
Challis GL (2005) A widely distributed bacterial pathway for siderophore biosynthesis independent of nonribosomal peptide synthetases. Chembiochem 6(4):601–611. doi:10.1002/cbic.200400283
Coscelli GA, Bermúdez R, Losada AP, Faílde LD, Santos Y, Quiroga MI (2014) Acute Aeromonas salmonicida infection in turbot (Scophthalmus maximus L.). Histopathological and immunohistochemical studies. aquac 430(0):79–85. doi:10.1016/j.aquaculture.2014.04.002
Daskalov H (2006) The importance of Aeromonas hydrophila in food safety. Food Control 17(6):474–483. doi:10.1016/j.foodcont.2005.02.009
Dennis JJ, Zylstra GJ (1998) Plasposons: modular self-cloning minitransposon derivatives for rapid genetic analysis of Gram-negative bacterial genomes. Appl Environ Microbiol 64(7):2710–2715
Escolar L, Pérez-Martín J, de Lorenzo V (1999) Opening the iron box: transcriptional metalloregulation by the Fur protein. J Bacteriol 181(20):6223–6229
Esmaeel Q, Pupin M, Kieu NP, Chataigné G, Béchet M, Deravel J, Krier F, Höfte M, Jacques P, Leclère V (2016) Burkholderia genome mining for nonribosomal peptide synthetases reveals a great potential for novel siderophores and lipopeptides synthesis. MicrobiolOpen 5(3):512–526. doi:10.1002/mbo3.347
Farto R, Milton D, Bermúdez M, Nieto T (2011) Colonization of turbot tissues by virulent and avirulent Aeromonas salmonicida subsp. salmonicida strains during infection. Dis Aquat Org 95(2):167–173. doi:10.3354/dao02342
Flissi A, Dufresne Y, Michalik J, Tonon L, Janot S, Noé L, Jacques P, Leclère V, Pupin M (2015) Norine, the knowledgebase dedicated to non-ribosomal peptides, is now open to crowdsourcing. Nucleic Acids Res 44(D1):D1113–D1118. doi:10.1093/nar/gkv1143
Funahashi T, Tanabe T, Miyamoto K, Tsujibo H, Maki J, Yamamoto S (2013) Characterization of a gene encoding the outer membrane receptor for ferric enterobactin in Aeromonas hydrophila ATCC 7966 T. Biosci Biotechnol Biochem 77(2):353–360. doi:10.1271/bbb.120774
Gehring AM, Mori I, Walsh CT (1998) Reconstitution and characterization of the Escherichia coli enterobactin synthetase from EntB, EntE, and EntF. Biochemistry 37(8):2648–2659. doi:10.1021/bi9726584
Janda JM, Abbott SL (2010) The genus Aeromonas: taxonomy, pathogenicity, and infection. Clin Microbiol Rev 23(1):35–73. doi:10.1128/cmr.00039-09
Leclère V, Béchet M, Blondeau R (2004) Functional significance of a periplasmic Mn-superoxide dismutase from Aeromonas hydrophila. J Appl Microbiol 96(4):828–833. doi:10.1111/j.1365-2672.2004.02231.x
Leclère V, Beaufort S, Dessoy S, Dehottay P, Jacques P (2009) Development of a biological test to evaluate the bioavailability of iron in culture media. J Appl Microbiol 107(5):1598–1605. doi:10.1111/j.1365-2672.2009.04345.x
Massad G, Arceneaux JEL, Byers BR (1991) Acquisition of iron from host sources by mesophilic Aeromonas species. J Gen Microbiol 137(2):237–241. doi:10.1099/00221287-137-2-237
Massad G, Arceneaux JEL, Byers BR (1994) Diversity of siderophore genes encoding biosynthesis of 2,3-dihydroxybenzoic acid in Aeromonas spp. Biometals 7(3):227–236. doi:10.1007/bf00149553
May JJ, Wendrich TM, Marahiel MA (2001) The dhb operon of Bacillus subtilis encodes the biosynthetic template for the catecholic siderophore 2,3-dihydroxybenzoate-glycine-threonine trimeric ester bacillibactin. J Biol Chem 276(10):7209–7217. doi:10.1074/jbc.M009140200
Merino S, Rubires X, Knøchel S, Tomás JM (1995) Emerging pathogens: Aeromonas spp. Int J Food Microbiol 28(2):157–168. doi:10.1016/0168-1605(95)00054-2
Najimi M, Lemos M, Osorio C (2008) Identification of siderophore biosynthesis genes essential for growth of Aeromonas salmonicida under iron limitation conditions. Appl Environ Microbiol 74:2341–2348. doi:10.1128/AEM.02728-07
Payne SM (1994) Detection, isolation, and characterization of siderophores. Meth Enzymol 235:329–344
Pupin M, Esmaeel Q, Flissi A, Dufresne Y, Jacques P, Leclère V (2015) Norine: a powerful resource for novel nonribosomal peptide discovery. Synth Syst Biotechnol 1(2):89–94. doi:10.1016/j.synbio.2015.11.001
Rausch C, Hoof I, Weber T, Wohlleben W, Huson D (2007) Phylogenetic analysis of condensation domains in NRPS sheds light on their functional evolution. BMC Evolutionary Biol 7(1):78. doi:10.1186/1471-2148-7-78
Reith M, Singh R, Curtis B, Boyd J, Bouevitch A, Kimball J, Munholland J, Murphy C, Sarty D, Williams J, Nash J, Johnson S, Brown L (2008) The genome of Aeromonas salmonicida subsp. salmonicida A449: insights into the evolution of a fish pathogen. BMC Genom 9(1):427. doi:10.1186/1471-2164-9-427
Röttig M, Medema MH, Blin K, Weber T, Rausch C, Kohlbacher O (2011) NRPSpredictor 2- a web server for predicting NRPS adenylation domain specificity. Nucleic Acids Res. doi:10.1093/nar/gkr323
Saltikov CW, Newman DK (2003) Genetic identification of a respiratory arsenate reductase. Proc Natl Acad Sci 100(19):10983–10988. doi:10.1073/pnas.1834303100
Schwyn B, Neilands JB (1987) Universal chemical assay for the detection and determination of siderophores. Anal Biochem 160(1):47–56. doi:10.1016/0003-2697(87)90612-9
Seshadri R, Joseph SW, Chopra AK, Sha J, Shaw J, Graf J, Haft D, Wu M, Ren Q, Rosovitz MJ, Madupu R, Tallon L, Kim M, Jin S, Vuong H, Stine OC, Ali A, Horneman AJ, Heidelberg JF (2006) Genome sequence of Aeromonas hydrophila ATCC 7966 T. J Bacteriol 188(23):8272–8282. doi:10.1128/jb.00621-06
Shanks RMQ, Caiazza NC, Hinsa SM, Toutain CM, O’Toole GA (2006) Saccharomyces cerevisiae-based molecular tool kit for manipulation of genes from Gram-negative bacteria. Appl Environ Microbiol 72(7):5027–5036. doi:10.1128/aem.00682-06
Strieker M, Tanović A, Marahiel MA (2010) Nonribosomal peptide synthetases: structures and dynamics. Curr Opin Struct Biol 20(2):234–240. doi:10.1016/j.sbi.2010.01.009
Telford JR, Raymond KN (1997) Amonabactin: a family of novel siderophores from a pathogenic bacterium. JBIC 2(6):750–761. doi:10.1007/s007750050191
Telford JR, Leary JA, Tunstad LMG, Byers BR, Raymond KN (1994) Amonabactin: characterization of a series of siderophores from Aeromonas hydrophila. J Am Chem Soc 116(10):4499–4500. doi:10.1021/ja00089a058
Toranzo AE, Magariños B, Romalde JL (2005) A review of the main bacterial fish diseases in mariculture systems. Aquac 246(1–4):37–61. doi:10.1016/j.aquaculture.2005.01.002
Van Lanen SG, Shen B (2006) Microbial genomics for the improvement of natural product discovery. Curr Opin Microbiol 9(3):252–260. doi:10.1016/j.mib.2006.04.002
Weber T, Blin K, Duddela S, Krug D, Kim HU, Bruccoleri R, Lee SY, Fischbach MA, Müller R, Wohlleben W, Breitling R, Takano E, Medema MH (2015) antiSMASH 3.0-a comprehensive resource for the genome mining of biosynthetic gene clusters. Nucleic Acids Res. doi:10.1093/nar/gkv437
Yu HB, Zhang YL, Lau YL, Yao F, Vilches S, Merino S, Tomas JM, Howard SP, Leung KY (2005) Identification and characterization of putative virulence genes and gene clusters in Aeromonas hydrophila PPD134/91. Appl Environ Microbiol 71(8):4469–4477. doi:10.1128/aem.71.8.4469-4477.2005
Ziemert N, Podell S, Penn K, Badger JH, Allen E, Jensen PR (2012) The natural product domain seeker NaPDoS: a phylogeny based bioinformatic tool to classify secondary metabolite gene diversity. PLoS One 7(3):e34064. doi:10.1371/journal.pone.0034064
Acknowledgments
This work was supported by the University of Lille 1, the INTERREG IV program France-Wallonie-Vlaanderen (Phytobio project), the bioinformatics platform bilille and Inria. QE received financial support from Sana’a University (Yemen).
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Funding
The work was funded by the University of Lille 1, the INTERREG IV program France-Wallonie-Vlaanderen (Phytobio project), the bioinformatics platform bilille, and Inria. QE received financial support from Sana’a University (Yemen).
Conflict of interest
The authors declare that they have no conflict of interest.
Ethical approval
This article does not contain any studies with human participants or animals performed by any of the authors.
Electronic supplementary material
ESM 1
(PDF 1249 kb)
Rights and permissions
About this article
Cite this article
Esmaeel, Q., Chevalier, M., Chataigné, G. et al. Nonribosomal peptide synthetase with a unique iterative-alternative-optional mechanism catalyzes amonabactin synthesis in Aeromonas . Appl Microbiol Biotechnol 100, 8453–8463 (2016). https://doi.org/10.1007/s00253-016-7773-4
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00253-016-7773-4