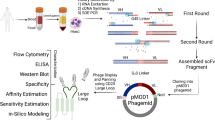
Abstract
Myasthenia gravis (MG) is an autoimmune disease caused by sensitization of the immune system to self-antigens. We have previously shown that targeting MG-susceptible alleles can significantly inhibit proliferation of disease-specific T cells. In this work, we humanized a murine monoclonal antibody (mAb) LG11, capable of blocking MG-associated DQ beta 1 (DQB1) allele and reformatted it into single-chain fragment variable (scFv). A fully functional humanized scFv was obtained by optimizing variable domain orientations and linker lengths, along with the optimization of expression conditions and codons to suit Escherichia coli expression machinery. Characterization of humanized scFv (FL8) revealed that the reformatted scFv, despite recognizing the same epitope as the parent murine LG11 mAb, exhibited superior binding affinity (0.97 nM) compared to the LG11 mAb, towards the immunizing antigen (DQB1*0601/70-90) and was able to block the proliferation of T cells cultured from PBLs of MG-patients typed DQB1*0601. The scFv was also capable of binding a variant MG-associated allele (DQB1*0502/70-90) with moderate affinity (18.7 nM), a feature that was absent in the LG11. To our knowledge, this is the first report of humanizing a MG-associated human leukocyte antigen (HLA) scFv for preclinical studies.






Similar content being viewed by others
References
Avidan N, Le Panse R, Berrih-Aknin S, Miller A (2014) Genetic basis of myasthenia gravis—a comprehensive review. J Autoimmun 52:146–153
Ayyar BV, Hearty S, O’Kennedy R (2010) Highly sensitive recombinant antibodies capable of reliably differentiating heart-type fatty acid binding protein from noncardiac isoforms. Anal Biochem 407:165–171
Ayyar BV, Aoki KR, Atassi MZ (2015a) The C-terminal heavy-chain domain of botulinum neurotoxin A is not the only site that binds neurons, as the N-terminal heavy-chain domain also plays a very active role in toxin-cell binding and interactions. Infect Immun 83:1465–1476
Ayyar BV, Hearty S, O’Kennedy R (2015b) Facile domain rearrangement abrogates expression recalcitrance in a rabbit scFv. Appl Microbiol Biotechnol 99:2693–2703
Ayyar BV, Tajhya RB, Beeton C, Zouhair Atassi M (2015c) Antigenic sites on the HN domain of botulinum neurotoxin A stimulate protective antibody responses against active toxin. Sci Rep 5:15776
Ayyar BV, Arora S, Ravi SS (2017) Optimizing antibody expression: the nuts and bolts. Methods 116:51–62
Berrih-Aknin S, Morel E, Raimond F, Safar D, Gaud C, Binet JP, Levasseur P, Bach JF (1987) The role of the thymus in myasthenia gravis: immunohistological and immunological studies in 115 cases. Ann N Y Acad Sci 505:50–70
Brochet X, Lefranc M-P, Giudicelli V (2008) IMGT/V-QUEST: the highly customized and integrated system for IG and TR standardized V-J and V-D-J sequence analysis. Nucleic Acids Res 36:W503–W508
Bujotzek A, Lipsmeier F, Harris SF, Benz J, Kuglstatter A, Georges G (2016) VH-VL orientation prediction for antibody humanization candidate selection: a case study. MAbs 8:288–305
Chamberlain N, Massad C, Oe T, Cantaert T, Herold KC, Meffre E (2016) Rituximab does not reset defective early B cell tolerance checkpoints. J Clin Invest 126:282–287
Conti-Fine BM, Diethelm-Okita B, Ostlie N, Wang W, Milani M (2009) Immunopathogenesis of myasthenia gravis. In: Kaminski H J (ed) Myasthenia gravis and related disorders. Humana Press, Totowa, pp 43–70
Deitiker PR, Oshima M, Smith RG, Mosier DR, Atassi MZ (2006) Subtle differences in HLA DQ haplotype-associated presentation of AChR α-chain peptides may suffice to mediate myasthenia gravis. Autoimmunity 39:277–288
Deitiker PR, Oshima M, Smith RG, Mosier D, Atassi MZ (2011) Association with HLA DQ of early onset myasthenia gravis in Southeast Texas region of the United States. Int J Immunogenet 38:55–62
Drachman DB (1994) Myasthenia gravis. N Engl J Med 330:1797–1810
Evans L, Hughes M, Waters J, Cameron J, Dodsworth N, Tooth D, Greenfield A, Sleep D (2010) The production, characterisation and enhanced pharmacokinetics of scFv–albumin fusions expressed in Saccharomyces cerevisiae. Protein Expr Purif 73:113–124
Fernando MMA, Stevens CR, Walsh EC, De Jager PL, Goyette P, Plenge RM, Vyse TJ, Rioux JD (2008) Defining the role of the MHC in autoimmunity: a review and pooled analysis. PLoS Genet 4:e1000024
Foote J, Winter G (1992) Antibody framework residues affecting the conformation of the hypervariable loops. J Mol Biol 224:487–499
Furrer E, Berdugo M, Stella C, Behar-Cohen F, Gurny R, Feige U, Lichtlen P, Urech DM (2009) Pharmacokinetics and posterior segment biodistribution of ESBA105, an anti–TNF-α single-chain antibody, upon topical administration to the rabbit eye. Invest Ophthalmol Vis Sci 50:771–778
Gil D, Schrum AG (2013) Strategies to stabilize compact folding and minimize aggregation of antibody-based fragments. Adv Biosci Biotechnol 4:73–84
Giraud M, Vandiedonck C, Garchon H-J (2008) Genetic factors in autoimmune myasthenia gravis. Ann N Y Acad Sci 1132:180–192
Giudicelli V, Lefranc M-P (2011) IMGT/JunctionAnalysis: IMGT standardized analysis of the V-J and V-D-J junctions of the rearranged immunoglobulins (Ig) and T cell receptors (TR). Cold Spring Harb Protoc 2011:716–725
Giudicelli V, Brochet X, Lefranc M-P (2011) IMGT/V-QUEST: IMGT standardized analysis of the immunoglobulin (Ig) and T cell receptor (TR) nucleotide sequences. Cold Spring Harb Protoc 2011:695–715
Guo J-Q, Li Q-M, Zhou J-Y, Zhang G-P, Yang Y-Y, Xing G-X, Zhao D, You S-Y, Zhang C-Y (2006) Efficient recovery of the functional IP10-scFv fusion protein from inclusion bodies with an on-column refolding system. Protein Expr Purif 45:168–174
Hohlfeld R, Kalies I, Kohleisen B, Heininger K, Conti-Tronconi B, Toyka KV (1986) Myasthenia gravis: stimulation of antireceptor autoantibodies by autoreactive T cell lines. Neurology 36:618–621
Kabat E, Wu TT, Perry HM, Gottesman KS, Foeller C (1991) Sequence of proteins of immunological interest. Publication No 91-3242. US Public Health Services, NIH, Bethesda
Kay J, Matteson EL, Dasgupta B, Nash P, Durez P, Hall S, Hsia EC, Han J, Wagner C, Xu Z, Visvanathan S, Rahman MU (2008) Golimumab in patients with active rheumatoid arthritis despite treatment with methotrexate: a randomized, double-blind, placebo-controlled, dose-ranging study. Arthritis Rheum 58:964–975
Kim Y-J, Neelamegam R, Heo M-A, Edwardraja S, Paik H-J, Lee S-G (2008) Improving the productivity of single-chain Fv antibody against c-Met by rearranging the order of its variable domains. J Microbiol Biotechnol 18:1186–1190
Koerber JT, Hornsby MJ, Wells JA (2015) An improved single-chain Fab platform for efficient display and recombinant expression. J Mol Biol 427:576–586
Kovalenko OV, Olland A, Piché-Nicholas N, Godbole A, King D, Svenson K, Calabro V, Müller MR, Barelle CJ, Somers W, Gill DS, Mosyak L, Tchistiakova L (2013) Atypical antigen recognition mode of a shark immunoglobulin new antigen receptor (IgNAR) variable domain characterized by humanization and structural analysis. J Biol Chem 288(24):17408–17419
Le Panse R, Bismuth J, Cizeron-Clairac G, Weiss JM, Cufi P, Dartevelle P, De Rosbo NK, Berrih-Aknin S (2010) Thymic remodeling associated with hyperplasia in myasthenia gravis. Autoimmunity 43:401–412
Leite MI, Jones M, Ströbel P, Marx A, Gold R, Niks E, Verschuuren JJGM, Berrih-Aknin S, Scaravilli F, Canelhas A, Morgan BP, Vincent A, Willcox N (2007) Myasthenia gravis thymus: complement vulnerability of epithelial and myoid cells, complement attack on them, and correlations with autoantibody status. Am J Pathol 171:893–905
Lu D, Jimenez X, Witte L, Zhu Z (2004) The effect of variable domain orientation and arrangement on the antigen-binding activity of a recombinant human bispecific diabody. Biochem Biophys Res Commun 318:507–513
Mamalaki A, Trakas N, Tzartos SJ (1993) Bacterial expression of a single-chain Fv fragment which efficiently protects the acetylcholine receptor against antigenic modulation caused by myasthenic antibodies. Eur J Immunol 23:1839–1845
Maniaol AH, Elsais A, Lorentzen ÅR, Owe JF, Viken MK, Sæther H, Flåm ST, Bråthen G, Kampman MT, Midgard R, Christensen M, Rognerud A, Kerty E, Gilhus NE, Tallaksen CME, Lie BA, Harbo HF (2012) Late onset myasthenia gravis is associated with HLA DRB1*15:01 in the Norwegian population. PLoS One 7:e36603
Marino M, Maiuri MT, Di Sante G, Scuderi F, La Carpia F, Trakas N, Provenzano C, Zisimopoulou P, Ria F, Tzartos SJ, Evoli A, Bartoccioni E (2014) T cell repertoire in DQ5-positive MuSK-positive myasthenia gravis patients. J Autoimmun 52:113–121
McDevitt HO (2000) Discovering the role of the major histocompatibility complex in the immune response. Annu Rev Immunol 18:1–17
Monod MY, Giudicelli V, Chaume D, Lefranc M-P (2004) IMGT/JunctionAnalysis: the first tool for the analysis of the immunoglobulin and T cell receptor complex V–J and V–D–J JUNCTIONs. Bioinformatics 20:i379–i385
Mulac-Jericevic B, Manshouri T, Yokoi T, Atassi MZ (1988) The regions of α-neurotoxin binding on the extracellular part of the α-subunit of human acetylcholine receptor. J Protein Chem 7:173–177
Nakayashiki N, Oshima M, Deitiker PR, Ashizawa T, Atassi MZ (2000) Suppression of experimental myasthenia gravis by monoclonal antibodies against MHC peptide region involved in presentation of a pathogenic T-cell epitope. J Neuroimmunol 105:131–144
Nancy P, Berrih-Aknin S (2005) Differential estrogen receptor expression in autoimmune myasthenia gravis. Endocrinology 146:2345–2353
Ofosu-Appiah W, Mokhtarian F, Shirazian D, Grob D (1994) Production of anti-acetylcholine receptor-a antibody in vitro by peripheral blood lymphocytes of patients with myasthenia gravis: role of immunoregulatory T cells and monocytes. J Lab Clin Med 124:231–241
Ojima-Kato T, Fukui K, Yamamoto H, Hashimura D, Miyake S, Hirakawa Y, Yamasaki T, Kojima T, Nakano H (2016) ‘Zipbody’ leucine zipper-fused Fab in E. coli in vitro and in vivo expression systems. Protein Eng Des Sel 29:149–157
Oshima M, Atassi MZ (1995) Effect of amino acid substitutions within the region 62-76 of I-A beta b on binding with and antigen presentation of Torpedo acetylcholine receptor alpha-chain peptide 146-162. J Immunol 154:5245–5254
Oshima M, Yokoi T, Deitiker P, Atassi MZ (1998) T cell responses in EAMG-susceptible and non-susceptible mouse strains after immunization with overlapping peptides encompassing the extracellular part of Torpedo californica acetylcholine receptor alpha chain. Implication to role in myasthenia gravis of autoimmune T-cell responses against receptor degradation products. Autoimmunity 27(2):79–90
Oshima M, Deitiker PR, Mosier DR, Smith RG, Atassi MZ (2005a) Responses in vitro of peripheral blood lymphocytes from patients with myasthenia gravis to stimulation with human acetylcholine receptor α-chain peptides: analysis in relation to age, thymic abnormality, and ethnicity. Hum Immunol 66:32–42
Oshima M, Ohtani M, Deitiker PR, Glenn Smith R, Mosier DR, Zouhair Atassi M (2005b) Suppression by mAbs against DQB1 peptides of in vitro proliferation of AChR-specific T cells from myasthenia gravis patients. Autoimmunity 38:161–169
Papanastasiou D, Mamalaki A, Eliopoulos E, Poulas K, Liolitsas C, Tzartos SJ (1999) Construction and characterization of a humanized single chain Fv antibody fragment against the main immunogenic region of the acetylcholine receptor. J Neuroimmunol 94:182–195
Protopapadakis E, Kokla A, Tzartos SJ, Mamalaki A (2005) Isolation and characterization of human anti-acetylcholine receptor monoclonal antibodies from transgenic mice expressing human immunoglobulin loci. Eur J Immunol 35:1960–1968
Robeson KR, Kumar A, Keung B, DiCapua DB, Grodinsky E, Patwa HS, Stathopoulos PA, Goldstein JM, O’Connor KC, Nowak RJ (2017) Durability of the rituximab response in acetylcholine receptor autoantibody-positive myasthenia gravis. JAMA Neurol 74:60–66
Rosenberg JS, Oshima M, Atassi MZ (1996) B-cell activation in vitro by helper T cells specific to region alpha 146-162 of Torpedo californica nicotinic acetylcholine receptor. J Immunol 157:3192–3199
Saruhan-Direskeneli G, Kiliç A, Parman Y, Serdaroğlu P, Deymeer F (2006) HLA-DQ polymorphism in Turkish patients with myasthenia gravis. Hum Immunol 67:352–358
Saruhan-Direskeneli G, Hughes T, Yilmaz V, Durmus H, Adler A, Alahgholi-Hajibehzad M, Aysal F, Yentür SP, Akalin MA, Dogan O, Marx A, Gülsen-Parman Y, Oflazer P, Deymeer F, Sawalha AH (2016) Genetic heterogeneity within the HLA region in three distinct clinical subgroups of myasthenia gravis. Clin Immunol 166–167:81–88
Shenoy M, Oshima M, Atassi MZ, Christadoss P (1993) Suppression of experimental autoimmune myasthenia gravis by epitope-specific neonatal tolerance to synthetic region alpha 146-162 of acetylcholine receptor. Clin Immunol Immunopathol 66:230–238
Sun H, Wu G, Chen Y, Tian Y, Yue Y, Zhang G (2014) Expression, production, and renaturation of a functional single-chain variable antibody fragment (scFv) against human ICAM-1. Braz J Med Biol Res 47:540–547
Teplyakov A, Obmolova G, Malia T, Gilliland G (2011) Antigen recognition by antibody C836 through adjustment of VL/VH packing. Acta Crystallogr Sect F Struct Biol Cryst Commun 67:1165–1167
Testi M, Terracciano C, Guagnano A, Testa G, Marfia GA, Pompeo E, Andreani M, Massa R (2012) Association of HLA-DQB1∗05:02 and DRB1∗16 alleles with late-onset, nonthymomatous, AChR-Ab-positive myasthenia gravis. Autoimmune Dis 2012:541760
Tiftikcioglu BI, Uludag IF, Zorlu Y, Pirim İ, Sener U, Tokucoglu F, Korucuk M (2017) Human leucocyte antigen B50 is associated with conversion to generalized myasthenia gravis in patients with pure ocular onset. Med Princ Pract 26:71–77
Tzartos SJ, Seybold ME, Lindstrom JM (1982) Specificities of antibodies to acetylcholine receptors in sera from myasthenia gravis patients measured by monoclonal antibodies. Proc Natl Acad Sci U S A 79:188–192
Tzartos S, Hochschwender S, Vasquez P, Lindstrom J (1987) Passive transfer of experimental autoimmune myasthenia gravis by monoclonal antibodies to the main immunogenic region of the acetylcholine receptor. J Neuroimmunol 15:185–194
Vander Heiden JA, Stathopoulos P, Zhou JQ, Chen L, Gilbert TJ, Bolen CR, Barohn RJ, Dimachkie MM, Ciafaloni E, Broering TJ, Vigneault F, Nowak RJ, Kleinstein SH, O’Connor KC (2017) Dysregulation of B cell repertoire formation in myasthenia gravis patients revealed through deep sequencing. J Immunol 198:1460–1473
Vandiedonck C, Beaurain G, Giraud M, Hue-Beauvais C, Eymard B, Tranchant C, Gajdos P, Dausset J, Garchon H-J (2004) Pleiotropic effects of the 8.1 HLA haplotype in patients with autoimmune myasthenia gravis and thymus hyperplasia. Proc Natl Acad Sci U S A 101:15464–15469
Vijayan N, Vijayan VK, Dreyfus PM (1977) Acetylcholinesterase activity and menstrual remissions in myasthenia gravis. J Neurol Neurosurg Psychiatry 40:1060–1065
Vrolix K, Fraussen J, Losen M, Stevens J, Lazaridis K, Molenaar PC, Somers V, Bracho MA, Le Panse R, Stinissen P, Berrih-Aknin S, Maessen JG, Van Garsse L, Buurman WA, Tzartos SJ, De Baets MH, Martinez-Martinez P (2014) Clonal heterogeneity of thymic B cells from early-onset myasthenia gravis patients with antibodies against the acetylcholine receptor. J Autoimmun 52:101–112
Wang Z, Raifu M, Howard M, Smith L, Hansen D, Goldsby R, Ratner D (2000) Universal PCR amplification of mouse immunoglobulin gene variable regions: the design of degenerate primers and an assessment of the effect of DNA polymerase 3′ to 5′ exonuclease activity. J Immunol Methods 233:167–177
Willcox N, Leite MI, Kadota Y, Jones M, Meager A, Subrahmanyam P, Dasgupta B, Morgan BP, Vincent A (2008) Autoimmunizing mechanisms in thymoma and thymus. Ann N Y Acad Sci 1132:163–173
Wolfe GI, Kaminski HJ, Aban IB, Minisman G, Kuo H-C, Marx A, Ströbel P, Mazia C, Oger J, Cea JG, Heckmann JM, Evoli A, Nix W, Ciafaloni E, Antonini G, Witoonpanich R, King JO, Beydoun SR, Chalk CH, Barboi AC, Amato AA, Shaibani AI, Katirji B, Lecky BRF, Buckley C, Vincent A, Dias-Tosta E, Yoshikawa H, Waddington-Cruz M, Pulley MT, Rivner MH, Kostera-Pruszczyk A, Pascuzzi RM, Jackson CE, Garcia Ramos GS, Verschuuren JJGM, Massey JM, Kissel JT, Werneck LC, Benatar M, Barohn RJ, Tandan R, Mozaffar T, Conwit R, Odenkirchen J, Sonett JR, Jaretzki AI, Newsom-Davis J, Cutter GR (2016) Randomized trial of thymectomy in myasthenia gravis. N Engl J Med 375:511–522
Wörn A, Plückthun A (2001) Stability engineering of antibody single-chain Fv fragments. J Mol Biol 305(5):989–1010
Yamanouchi J, Rainbow D, Serra P, Howlett S, Hunter K, Garner VES, Gonzalez-Munoz A, Clark J, Veijola R, Cubbon R, Chen S-L, Rosa R, Cumiskey AM, Serreze DV, Gregory S, Rogers J, Lyons PA, Healy B, Smink LJ, Todd JA, Peterson LB, Wicker LS, Santamaria P (2007) Interleukin-2 gene variation impairs regulatory T cell function and causes autoimmunity. Nat Genet 39:329–337
Yousefipour G-A, Salami Z, Farjadian S (2009) Association of HLA-DQA1* 0101/2 and DQB1* 0502 with myasthenia gravis in southern Iranian patients. Iran J Immunol 6:99–102
Yuan X, Gubbins MJ, Berry JD (2004) A simple and rapid protocol for the sequence determination of functional kappa light chain cDNAs from aberrant-chain-positive murine hybridomas. J Immunol Methods 294:199–207
Zagoriti Z, Kambouris ME, Patrinos GP, Tzartos SJ, Poulas K (2013) Recent advances in genetic predisposition of myasthenia gravis. Biomed Res Int 2013:404053
Zhang K, Geddie ML, Kohli N, Kornaga T, Kirpotin DB, Jiao Y, Rennard R, Drummond DC, Nielsen UB, Xu L, Lugovskoy AA (2015) Comprehensive optimization of a single-chain variable domain antibody fragment as a targeting ligand for a cytotoxic nanoparticle. MAbs 7:42–52
Acknowledgements
The authors wish to acknowledge Prof. Carlos F. Barbas III (The Scripps Research Institute, La Jolla, CA) for providing pComb3XSS vector; Dr. Minako Oshima for providing anti-DQB1*0601 mAb, synthetic peptides, and reagents for T cell assay; and Dr. Philip Deitiker for technical help with beta counter. Additionally, the authors would like to thank Dr. Timothy Palzkill and Dr. Zhizeng Sun for allowing us to use their Biacore instrument and providing useful suggestions during analysis. The authors appreciate the assistance of Dr. Sushrut Arora with the proof-reading and scientific editing.
Author information
Authors and Affiliations
Corresponding authors
Ethics declarations
Ethical statement
The authors accept the rules of good scientific practice and agree with the COPE guidelines followed by the Applied Microbiology and Biotechnology Journal.
Conflict of interest
The authors declare that they have no conflict of interest.
Ethical approval
Procedures performed in studies involving human participants were in accordance with the ethical standards of the Institutional Review Board of Baylor College of Medicine. This article does not contain any studies with animals performed by any of the authors.
Informed consent
Blood samples were obtained from patients with the clinical diagnosis of generalized myasthenia. A written informed consent was obtained from the patients, which was approved by the Institutional Review Board of Baylor College of Medicine.
Electronic supplementary material
ESM 1
(PDF 202 kb).
Rights and permissions
About this article
Cite this article
Ayyar, B., Atassi, M. Development of humanized scFv antibody fragment(s) that targets and blocks specific HLA alleles linked to myasthenia gravis. Appl Microbiol Biotechnol 101, 8165–8179 (2017). https://doi.org/10.1007/s00253-017-8557-1
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00253-017-8557-1