Abstract
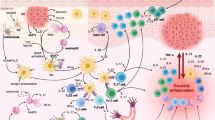
Rheumatoid arthritis (RA) is a systemic autoimmune disease that causes joint pain, inflammation, and loss of function. Disease pathogenesis involves activation and proliferation of autoreactive pro-inflammatory effector T cells. While the details of RA onset and progression remain controversial, dendritic cell (DC) numbers dramatically increase in the synovial joint tissues of RA patients. Based on their key functions as antigen-presenting cells and inducers of T cell differentiation, DCs may play an important role in the initiation of joint inflammation. Myeloid DC contributions are likely central to the development of RA, as they are more efficient at antigen presentation in comparison with their closely related cousins, plasmacytoid DCs. Synovial fluid in the joints of RA patients is enriched with pro-inflammatory cytokines and chemokines, which may stimulate or result from DC activation. Epidemiological evidence indicates that smoking and periodontal infection are major environmental risk factors for RA development. In this review, factors in the synovial environment that contribute to altered myeloid DC functions in RA and the effects of environmental risk factors on myeloid DCs are described.

Similar content being viewed by others
References
Gibofsky A (2012) Overview of epidemiology, pathophysiology, and diagnosis of rheumatoid arthritis. Am J Manag Care 18(13 Suppl):S295–S302
McInnes IB, Schett G (2011) The pathogenesis of rheumatoid arthritis. N Engl J Med 365(23):2205–2219. doi:10.1056/NEJMra1004965
Entezami P, Fox DA, Clapham PJ, Chung KC (2011) Historical perspective on the etiology of rheumatoid arthritis. Hand Clin 27(1):1–10. doi:10.1016/j.hcl.2010.09.006
Boissier MC, Semerano L, Challal S, Saidenberg-Kermanac’h N, Falgarone G (2012) Rheumatoid arthritis: from autoimmunity to synovitis and joint destruction. J Autoimmun 39(3):222–228. doi:10.1016/j.jaut.2012.05.021
Mellman I, Steinman RM (2001) Dendritic cells: specialized and regulated antigen processing machines. Cell 106(3):255–258
Jia XY, Chang Y, Sun XJ, Dai X, Wei W (2014) The role of prostaglandin E2 receptor signaling of dendritic cells in rheumatoid arthritis. Int Immunopharmacol 23(1):163–169. doi:10.1016/j.intimp.2014.08.024
Schmidt SV, Nino-Castro AC, Schultze JL (2012) Regulatory dendritic cells: there is more than just immune activation. Front Immunol 3:274. doi:10.3389/fimmu.2012.00274
Cosway E, Anderson G, Garside P, Prendergast C (2016) The thymus and rheumatology: should we care? Curr Opin Rheumatol 28(2):189–195. doi:10.1097/bor.0000000000000251
Zvaifler NJ, Steinman RM, Kaplan G, Lau LL, Rivelis M (1985) Identification of immunostimulatory dendritic cells in the synovial effusions of patients with rheumatoid arthritis. J Clin Invest 76(2):789–800. doi:10.1172/jci112036
Harding B, Knight SC (1986) The distribution of dendritic cells in the synovial fluids of patients with arthritis. Clin Exp Immunol 63(3):594–600
Miossec P (2008) Dynamic interactions between T cells and dendritic cells and their derived cytokines/chemokines in the rheumatoid synovium. Arthritis research therapy 10(Suppl 1):S2. doi:10.1186/ar2413
Leung BP, Conacher M, Hunter D, McInnes IB, Liew FY, Brewer JM (2002) A novel dendritic cell-induced model of erosive inflammatory arthritis: distinct roles for dendritic cells in T cell activation and induction of local inflammation. J Immunol (Baltimore, Md: 1950) 169 (12):7071–7077
Satpathy AT, Wu X, Albring JC, Murphy KM (2012) Re(de)fining the dendritic cell lineage. Nat Immunol 13(12):1145–1154. doi:10.1038/ni.2467
Collin M, McGovern N, Haniffa M (2013) Human dendritic cell subsets. Immunology 140(1):22–30. doi:10.1111/imm.12117
Merad M, Sathe P, Helft J, Miller J, Mortha A (2013) The dendritic cell lineage: ontogeny and function of dendritic cells and their subsets in the steady state and the inflamed setting. Annu Rev Immunol 31:563–604. doi:10.1146/annurev-immunol-020711-074950
Lebre MC, Jongbloed SL, Tas SW, Smeets TJ, McInnes IB, Tak PP (2008) Rheumatoid arthritis synovium contains two subsets of CD83-DC-LAMP- dendritic cells with distinct cytokine profiles. Am J Pathol 172(4):940–950. doi:10.2353/ajpath.2008.070703
Benson RA, Patakas A, Conigliaro P, Rush CM, Garside P, McInnes IB, Brewer JM (2010) Identifying the cells breaching self-tolerance in autoimmunity. J Immunol (Baltimore, Md: 1950) 184(11):6378–6385. doi:10.4049/jimmunol.0903951
Jongbloed SL, Benson RA, Nickdel MB, Garside P, McInnes IB, Brewer JM (2009) Plasmacytoid dendritic cells regulate breach of self-tolerance in autoimmune arthritis. J Immunol (Baltimore, Md: 1950) 182(2):963–968
Corsiero E, Bombardieri M, Manzo A, Bugatti S, Uguccioni M, Pitzalis C (2012) Role of lymphoid chemokines in the development of functional ectopic lymphoid structures in rheumatic autoimmune diseases. Immunol Lett 145(1–2):62–67. doi:10.1016/j.imlet.2012.04.013
Rovati B, Mariucci S, Manzoni M, Bencardino K, Danova M (2008) Flow cytometric detection of circulating dendritic cells in healthy subjects. EJH 52(1):45–52
Moret FM, Hack CE, van der Wurff-Jacobs KM, de Jager W, Radstake TR, Lafeber FP, van Roon JA (2013) Intra-articular CD1c-expressing myeloid dendritic cells from rheumatoid arthritis patients express a unique set of T cell-attracting chemokines and spontaneously induce Th1, Th17 and Th2 cell activity. Arthritis Res Ther 15(5):R155. doi:10.1186/ar4338
Moret FM, Hack CE, van der Wurff-Jacobs KM, Radstake TR, Lafeber FP, van Roon JA (2014) Thymic stromal lymphopoietin, a novel proinflammatory mediator in rheumatoid arthritis that potently activates CD1c + myeloid dendritic cells to attract and stimulate T cells. Arthritis Rheumatol (Hoboken, NJ) 66(5):1176–1184. doi:10.1002/art.38338
Santiago B, Izquierdo E, Rueda P, Del Rey MJ, Criado G, Usategui A, Arenzana-Seisdedos F, Pablos JL (2012) CXCL12gamma isoform is expressed on endothelial and dendritic cells in rheumatoid arthritis synovium and regulates T cell activation. Arthritis Rheum 64(2):409–417. doi:10.1002/art.33345
Segura E, Touzot M, Bohineust A, Cappuccio A, Chiocchia G, Hosmalin A, Dalod M, Soumelis V, Amigorena S (2013) Human inflammatory dendritic cells induce Th17 cell differentiation. Immunity 38(2):336–348. doi:10.1016/j.immuni.2012.10.018
Robbins SH, Walzer T, Dembele D, Thibault C, Defays A, Bessou G, Xu H, Vivier E, Sellars M, Pierre P, Sharp FR, Chan S, Kastner P, Dalod M (2008) Novel insights into the relationships between dendritic cell subsets in human and mouse revealed by genome-wide expression profiling. Genome Biol 9(1):R17. doi:10.1186/gb-2008-9-1-r17
Richez C, Schaeverbeke T, Dumoulin C, Dehais J, Moreau JF, Blanco P (2009) Myeloid dendritic cells correlate with clinical response whereas plasmacytoid dendritic cells impact autoantibody development in rheumatoid arthritis patients treated with infliximab. Arthritis Res Ther 11(3):R100. doi:10.1186/ar2746
Jongbloed SL, Lebre MC, Fraser AR, Gracie JA, Sturrock RD, Tak PP, McInnes IB (2006) Enumeration and phenotypical analysis of distinct dendritic cell subsets in psoriatic arthritis and rheumatoid arthritis. Arthritis Res Ther 8(1):R15. doi:10.1186/ar1864
Richez C, Barnetche T, Khoryati L, Duffau P, Kostine M, Contin-Bordes C, Blanco P, Schaeverbeke T (2012) Tocilizumab treatment decreases circulating myeloid dendritic cells and monocytes, 2 components of the myeloid lineage. J Rheumatol 39(6):1192–1197. doi:10.3899/jrheum.111439
Estrada-Capetillo L, Hernandez-Castro B, Monsivais-Urenda A, Alvarez-Quiroga C, Layseca-Espinosa E, Abud-Mendoza C, Baranda L, Urzainqui A, Sanchez-Madrid F, Gonzalez-Amaro R (2013) Induction of Th17 lymphocytes and Treg cells by monocyte-derived dendritic cells in patients with rheumatoid arthritis and systemic lupus erythematosus. Clin Dev Immunol 2013:584303. doi:10.1155/2013/584303
Prevosto C, Goodall JC, Gaston JSH (2012) Cytokine secretion by pathogen recognition receptor-stimulated dendritic cells in rheumatoid arthritis and ankylosing spondylitis. J Rheumatol 39(10):1918–1928. doi:10.3899/jrheum.120208
Robert C, Fuhlbrigge RC, Kieffer JD, Ayehunie S, Hynes RO, Cheng G, Grabbe S, von Andrian UH, Kupper TS (1999) Interaction of dendritic cells with skin endothelium: a new perspective on immunosurveillance. J Exp Med 189(4):627–636
Urzainqui A, Martinez del Hoyo G, Lamana A, de la Fuente H, Barreiro O, Olazabal IM, Martin P, Wild MK, Vestweber D, Gonzalez-Amaro R, Sanchez-Madrid F (2007) Functional role of P-selectin glycoprotein ligand 1/P-selectin interaction in the generation of tolerogenic dendritic cells. J Immunol (Baltimore, Md: 1950) 179 (11):7457–7465
Kuipers H, Muskens F, Willart M, Hijdra D, van Assema FB, Coyle AJ, Hoogsteden HC, Lambrecht BN (2006) Contribution of the PD-1 ligands/PD-1 signaling pathway to dendritic cell-mediated CD4 + T cell activation. Eur J Immunol 36(9):2472–2482. doi:10.1002/eji.200635978
Moret FM, van der Wurff-Jacobs KM, Bijlsma JW, Lafeber FP, van Roon JA (2014) Synovial T cell hyporesponsiveness to myeloid dendritic cells is reversed by preventing PD-1/PD-L1 interactions. Arthritis Res Ther 16(6):497. doi:10.1186/s13075-014-0497-x
Feldmann M, Brennan FM, Maini RN (1996) Role of cytokines in rheumatoid arthritis. Annu Rev Immunol 14:397–440. doi:10.1146/annurev.immunol.14.1.397
Mertens M, Singh JA (2009) Anakinra for rheumatoid arthritis. Cochrane Database Syst Rev 1:Cd005121. doi:10.1002/14651858.CD005121.pub3
Mima T, Nishimoto N (2009) Clinical value of blocking IL-6 receptor. Curr Opin Rheumatol 21(3):224–230. doi:10.1097/BOR.0b013e3283295fec
Lutzky V, Hannawi S, Thomas R (2007) Cells of the synovium in rheumatoid arthritis. Dendritic cells. Arthritis Res Ther 9(4):219. doi:10.1186/ar2200
Zhan Y, Xu Y, Lew AM (2012) The regulation of the development and function of dendritic cell subsets by GM-CSF: more than a hematopoietic growth factor. Mol Immunol 52(1):30–37. doi:10.1016/j.molimm.2012.04.009
Bhattacharya P, Budnick I, Singh M, Thiruppathi M, Alharshawi K, Elshabrawy H, Holterman MJ, Prabhakar BS (2015) Dual role of GM-CSF as a pro-inflammatory and a regulatory cytokine: implications for immune therapy. J Interfer Cytokine Res 35(8):585–599. doi:10.1089/jir.2014.0149
Reynolds G, Gibbon JR, Pratt AG, Wood MJ, Coady D, Raftery G, Lorenzi AR, Gray A, Filer A, Buckley CD, Haniffa MA, Isaacs JD, Hilkens CM (2016) Synovial CD4 + T-cell-derived GM-CSF supports the differentiation of an inflammatory dendritic cell population in rheumatoid arthritis. Ann Rheum Dis 75(5):899–907. doi:10.1136/annrheumdis-2014-206578
van Roon JA, Verweij MC, Wijk MW, Jacobs KM, Bijlsma JW, Lafeber FP (2005) Increased intraarticular interleukin-7 in rheumatoid arthritis patients stimulates cell contact-dependent activation of CD4(+) T cells and macrophages. Arthritis Rheum 52(6):1700–1710. doi:10.1002/art.21045
Dodge GR, Poole AR (1989) Immunohistochemical detection and immunochemical analysis of type II collagen degradation in human normal, rheumatoid, and osteoarthritic articular cartilages and in explants of bovine articular cartilage cultured with interleukin 1. J Clin Invest 83(2):647–661. doi:10.1172/jci113929
Martin CA, Carsons SE, Kowalewski R, Bernstein D, Valentino M, Santiago-Schwarz F 2003 Aberrant extracellular and dendritic cell (DC) surface expression of heat shock protein (hsp)70 in the rheumatoid joint: possible mechanisms of hsp/DC-mediated cross-priming. J Immunol (Baltimore, Md: 1950) 171(11):5736–5742
Spierings J, van Eden W (2016) Heat shock proteins and their immunomodulatory role in inflammatory arthritis. Rheumatology (Oxford, England). doi:10.1093/rheumatology/kew266
Egelston C, Kurko J, Besenyei T, Tryniszewska B, Rauch TA, Glant TT, Mikecz K (2012) Suppression of dendritic cell maturation and T cell proliferation by synovial fluid myeloid cells from mice with autoimmune arthritis. Arthritis Rheum 64(10):3179–3188. doi:10.1002/art.34494
Nanki T, Hayashida K, El-Gabalawy HS, Suson S, Shi K, Girschick HJ, Yavuz S, Lipsky PE (2000) Stromal cell-derived factor-1-CXC chemokine receptor 4 interactions play a central role in CD4 + T cell accumulation in rheumatoid arthritis synovium. J Immunol (Baltimore, Md: 1950) 165(11):6590–6598
Barrow AD, Raynal N, Andersen TL, Slatter DA, Bihan D, Pugh N, Cella M, Kim T, Rho J, Negishi-Koga T, Delaisse JM, Takayanagi H, Lorenzo J, Colonna M, Farndale RW, Choi Y, Trowsdale J (2011) OSCAR is a collagen receptor that costimulates osteoclastogenesis in DAP12-deficient humans and mice. J Clin Invest 121(9):3505–3516. doi:10.1172/jci45913
Schultz HS, Nitze LM, Zeuthen LH, Keller P, Gruhler A, Pass J, Chen J, Guo L, Fleetwood AJ, Hamilton JA, Berchtold MW, Panina S 2015 Collagen induces maturation of human monocyte-derived dendritic cells by signaling through osteoclast-associated receptor. J Immunol (Baltimore, Md: 1950) 194(7):3169–3179. doi:10.4049/jimmunol.1402800
Lakey RL, Morgan TG, Rowan AD, Isaacs JD, Cawston TE, Hilkens CM (2009) A novel paradigm for dendritic cells as effectors of cartilage destruction. Rheumatology (Oxford, England) 48(5):502–507. doi:10.1093/rheumatology/kep040
Hayem G, De Bandt M, Palazzo E, Roux S, Combe B, Eliaou JF, Sany J, Kahn MF, Meyer O (1999) Anti-heat shock protein 70 kDa and 90 kDa antibodies in serum of patients with rheumatoid arthritis. Ann Rheum Dis 58(5):291–296
Corrigall VM, Vittecoq O, Panayi GS (2009) Binding immunoglobulin protein-treated peripheral blood monocyte-derived dendritic cells are refractory to maturation and induce regulatory T-cell development. Immunology 128(2):218–226. doi:10.1111/j.1365-2567.2009.03103.x
Stocki P, Wang XN, Dickinson AM (2012) Inducible heat shock protein 70 reduces T cell responses and stimulatory capacity of monocyte-derived dendritic cells. J Biol Chem 287(15):12387–12394. doi:10.1074/jbc.M111.307579
Rosenthal KS, Mikecz K, Steiner HL 3rd, Glant TT, Finnegan A, Carambula RE, Zimmerman DH (2015) Rheumatoid arthritis vaccine therapies: perspectives and lessons from therapeutic ligand epitope antigen presentation system vaccines for models of rheumatoid arthritis. Expert Rev Vaccines 14(6):891–908. doi:10.1586/14760584.2015.1026330
Kazantseva MG, Highton J, Stamp LK, Hessian PA (2012) Dendritic cells provide a potential link between smoking and inflammation in rheumatoid arthritis. Arthritis Res Ther 14(5):R208. doi:10.1186/ar4046
Mastrangelo A, Colasanti T, Barbati C, Pecani A, Sabatinelli D, Pendolino M, Truglia S, Massaro L, Mancini R, Miranda F, Spinelli FR, Conti F, Alessandri C (2015) The role of posttranslational protein modifications in rheumatological diseases: focus on rheumatoid arthritis. J Immunol Res 2015:712490. doi:10.1155/2015/712490
Willemze A, Toes RE, Huizinga TW, Trouw LA (2012) New biomarkers in rheumatoid arthritis. Neth J Med 70(9):392–399
Scally SW, Petersen J, Law SC, Dudek NL, Nel HJ, Loh KL, Wijeyewickrema LC, Eckle SB, van Heemst J, Pike RN, McCluskey J, Toes RE, La Gruta NL, Purcell AW, Reid HH, Thomas R, Rossjohn J (2013) A molecular basis for the association of the HLA-DRB1 locus, citrullination, and rheumatoid arthritis. J Exp Med 210(12):2569–2582. doi:10.1084/jem.20131241
Yu H, Yang YH, Rajaiah R, Moudgil KD (2011) Nicotine-induced differential modulation of autoimmune arthritis in the Lewis rat involves changes in interleukin-17 and anti-cyclic citrullinated peptide antibodies. Arthritis Rheum 63(4):981–991. doi:10.1002/art.30219
Santegoets KC, Wenink MH, Braga FA, Cossu M, Lamers-Karnebeek FB, van Riel PL, Sturm PD, van den Berg WB, Radstake TR (2016) Impaired porphyromonas gingivalis-induced tumor necrosis factor production by dendritic cells typifies patients with rheumatoid arthritis. Arthritis Rheumatol (Hoboken, NJ) 68 (4):795–804. doi:10.1002/art.39514
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
The authors declare that they have no conflict of interest.
Ethical approval
This article does not contain any studies with human participants performed by any of the authors.
Funding
This research did not receive any specific grant from funding agencies in the public, commercial, or not-for-profit sectors.
Rights and permissions
About this article
Cite this article
Yu, M.B., Langridge, W.H.R. The function of myeloid dendritic cells in rheumatoid arthritis. Rheumatol Int 37, 1043–1051 (2017). https://doi.org/10.1007/s00296-017-3671-z
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00296-017-3671-z