Abstract
Large-scale climate oscillations may contribute to the observed dramatic fluctuations and regional synchrony in Rangifer abundance. Here, we test this hypothesis using long-term abundance and physical condition datasets to investigate the relationships between broad climate patterns, summer-range quality, and population dynamics in three barren-ground caribou herds in northern Canada. We found that positive intensities of the Arctic Oscillation (AO) in the summer were associated with warmer temperatures, improved growing conditions for vegetation, and better body condition of caribou. Over this same period, the body condition of female caribou was positively related to fecundity. We further identified that population trajectories of caribou herds followed the direction of the AO: herds increased under positive AO intensity, and decreased under negative AO intensity. Our findings suggest that the AO influences barren-ground caribou population dynamics through effects on summer-range quality, caribou physical condition, and herd productivity.
Similar content being viewed by others
Introduction
Dramatic fluctuations of barren-ground caribou (Rangifer tarandus groenlandicus) populations have been well documented, but remain poorly understood (Gunn 2003). These fluctuations have been linked to variation in summer- and winter-range quality and availability (Adamczewski et al. 1986; Manseau et al. 1996), predation (Bergerud and Ballard 1988), extreme weather events (Chan et al. 2005), parasites (Weladji et al. 2003; Hughes et al. 2009), and forage exploitation (Messier et al. 1988). A global decline in Rangifer populations has occurred across the species’ range with climate change and anthropogenic disturbance implicated as potential mechanisms (Vors and Boyce 2009). While various factors contribute to the abundance trajectory of each herd, regional synchrony in abundance has been observed across proximate caribou herds (Klein 1991; Gunn et al. 2011; Fauchald et al. 2017) although this synchrony is not consistent (Bergerud 1996). Regional variation in caribou population trends can be partly attributed to the effects of broad-scale climate patterns, such as those represented by Arctic Oscillation (AO), the North Atlantic Oscillation (NAO), and the Pacific Decadal Oscillation (PDO) (Joly et al. 2011).
The AO is a broad climate index derived from surface atmospheric pressure patterns that track the strength of the polar vortex (Thompson and Wallace 1998). The AO has positive and negative phases that influence broad weather patterns across the northern hemisphere (Thompson et al. 2000). For example, during the positive phase of the AO, atmospheric pressure over the Arctic is lower than average, which tends to result in warmer and wetter winters in northern regions as warmer air is able to move further north (Thompson et al. 2000; Aanes et al. 2002). Negative values of the AO indicate high pressure in the Arctic region, allowing greater southward penetration of cold Arctic air. However, the effects of the AO on weather patterns can vary markedly across the north (Joly et al. 2011). The AO can and does fluctuate between positive and negative phases daily, but often will remain primarily in one phase or the other for prolonged periods. Similar indices, such as the NAO and PDO, influence weather patterns in a comparable way, though in different regions of the world (Hurrell 1995; Mantua et al. 1997).
Several studies have found links between broad climate patterns, represented by climate indices, and Rangifer populations around the circumpolar world. Relationships have been identified between the intensity of the AO and reindeer population growth rates on Svalbard (Aanes et al. 2002), similarly for the AO and PDO and caribou growth rates and calf recruitment in Alaska and the Yukon Territory (Hegel et al. 2010; Joly et al. 2011), and again for the AO and Porcupine herd abundance (Griffith et al. 2002), the NAO and caribou population dynamics (Post and Stenseth 1999; Forchhammer et al. 2002; Post and Forchhammer 2002), and the NAO and caribou calf body mass (Couturier et al. 2009). In contrast, Zalatan (2006) examined a 100-year period and found an inconsistent relationship between the intensity of the AO and reported dynamics of the Bathurst population.
Here, we examine the relationship between the AO and three barren-ground caribou herds in northern Canada: the Bathurst, Beverly, and Qamanirjuaq (Nagy et al. 2011). These three herds have undergone relatively synchronous population trajectories (Fig. 1) over the past several decades, increasing through the 1980s, reaching peak abundance in the late 1980s to mid 1990s, and then declining at varying rates to present (Gunn et al. 2011). These declines, and particularly that of the Bathurst herd, have generated substantial concern among researchers, wildlife managers, and communities across the herds’ ranges. Recent studies have investigated potential mechanisms for these declines (Boulanger et al. 2011; Adamczewski et al. 2015), but have not explored whether the similar trends in abundance might be related to broad climate patterns. We hypothesize that the relative synchrony of population trajectories of the Bathurst, Beverly, and Qamanirjuaq caribou herds over the last 35 years is in part related to the effects of the intensity of the AO, and further that long-term abundance fluctuations of barren-ground caribou are linked to broad climate indices. We expect that this relationship acts through the influence of climate patterns on environmental conditions that affect forage quality, quantity, and availability on the caribou range (e.g., Aanes et al. 2002), and thus on the physical condition of individual animals. To test these hypotheses, we use long-term datasets on the physical condition and fecundity of the Beverly herd, and estimates of abundance of each herd over the past 35 years. First we describe how the AO is related to local weather patterns on the range of these herds. We then test for a relationship between the AO and physical condition of Beverly caribou from 1982 to 1987, and demonstrate that this can be linked to herd productivity. Lastly, we further investigate the relationship between the AO and barren-ground caribou by comparing the intensity of the AO to the direction of population growth for these three barren-ground caribou herds.
Materials and methods
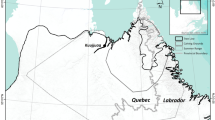
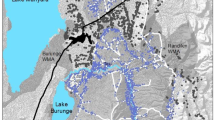
Barren-ground caribou can be classified into several ecotypes based on their level of sociality, spatial tenure, and migratory behavior (Nagy et al. 2011). The Bathurst, Beverly, and Qamanirjuaq herds are mainland migratory barren-ground caribou, an ecotype characterized by large aggregations of animals and collective long-distance migrations from winter ranges near or below the tree line to calving grounds and summer ranges on the tundra (Banfield 1954). Nagy et al. (2011) provided annual ranges for these three herds, among others, delineated by utilization distributions calculated from locations of satellite-collared female caribou collected by the Governments of Nunavut and the Northwest Territories (Fig. 2). The summer ranges that we present represent the utilization distributions of satellite-collared female caribou from June 1 to August 31.
Annual ranges (solid outline) adapted from Nagy et al. (2011), and June–August (hatched) ranges of the Bathurst, Beverly, and Qamanirjuaq barren-ground caribou (Rangifer tarandus groenlandicus) herds. Note that June–August range extents have been clipped to conform to Nagy et al. (2011) annual range extents
We used linear regression to identify associations between the intensity (positive or negative) of the AO and local climate variables. Annual, summer, and winter values of the AO index from 1979 to 2016 were calculated from data provided by the National Oceanic and Atmospheric Administration (https://www.ncdc.noaa.gov/teleconnections/ao, accessed 11 December 2016). Annual AO values were determined as the mean AO value for all months, winter values from the mean AO intensity of January, February, and March, and summer values from the AO intensities of June, July, and August. Climate data for the Beverly range were accessed from the Circum Arctic Rangifer Monitoring and Assessment (CARMA) network’s caribou range climate database (Russell et al. 2013). The CARMA database is derived from NASA’s Modern Era Retrospective Analysis for Research and Applications (Rienecker et al. 2011).
Multiple regression was used to identify associations between the body condition of male and female Beverly caribou in March and the intensity of the AO during winter, the previous summer, and throughout the previous year. Information on the physical condition of Beverly caribou from 1980 to 1987 was reported in Thomas and Kiliaan (1998a), who calculated a number of body condition indices from data that they collected from 856 female and 402 male Beverly caribou. Dissectible fat (DFAT), estimated by an equation developed for barren-ground caribou by Adamczewski et al. (1987), was found to be the best condition index among those that they calculated (Thomas and Kiliaan 1998a). We used mean values of DFAT calculated for age and sex classes to approximate body condition each year over the study period. Similar data on body condition for the other two herds were not available for our analysis.
Population estimates for the Beverly, Bathurst, and Qamanirjuaq caribou herds were taken from previous publications (Beverly: Campbell et al. 2012; Qamanirjuaq: Campbell et al. 2010, 2016; Bathurst: Heard and Williams 1991a, b; Gunn et al. 1996; Boulanger et al. 2014; Boulanger 2015). The abundance of each of these herds has been estimated periodically for over 40 years by different researchers, although over time survey methods for estimating herd abundance have changed and improved (e.g., visual calving ground surveys to photographic calving ground surveys). This creates challenges in using early abundance estimates to make direct comparisons. For these reasons, the earliest population estimate that we considered in our analysis was from 1982 when calving ground photo surveys became the standard survey method. Although this restricted the temporal scope of our study, the number of estimates across herds over 35 years provided a large enough sample for our analyses.
For each intervening period between herd population estimates, we compared the average value of the summer intensity of the AO (only summer intensity was significantly related to caribou physical condition, see “Results”) to the direction of population growth (positive, stable, or negative) for that period. We acknowledge that a linear trend over these periods, particularly the longer ones, is unlikely, and we did not think that the available data allowed for meaningful estimates of growth rates between population estimates. Herd abundance was designated as stable if no statistically significant trend was observed between consecutive population estimates. For example, the mean abundance estimate for the Qamanirjuaq herd declined from 495,665 (± 105,426 SE) in 1994 to 348,661 (± 44,861 SE) in 2008; however, due to overlapping confidence intervals, the authors were not able to decisively determine a trend (Campbell et al. 2010) and we identify the population trend over this period as “stable.” We performed an exact binomial test to determine whether the number of observed periods where the direction of the AO (positive or negative) was equivalent to the direction of population growth was greater than would be expected by chance. All statistical analyses were performed in Program R (R Core Team 2016).
Results
Arctic Oscillation and local climate
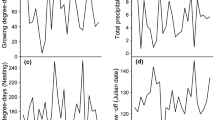
The average intensity of the AO during the summer (1 June–31 Aug) was significantly positively associated with summer temperatures on the Beverly range (Fig. 3; R2 = 0.20, F29 = 7.13, p = 0.01), meaning that summers with a positive AO value tended to be warmer. The average intensity of the AO during the summer also was positively associated with cumulative growing degrees (Russell et al. 2013) above 5 °C (Fig. 4; R2 = 0.21, F29 = 7.53, p = 0.01), an indicator of the length of a growing season. We did not find a relationship between the intensity of the AO in the summer and precipitation, although higher precipitation tended to occur in cooler years on the Beverly summer range (R2 = 0.10, F29 = 3.41, p = 0.07). There was no consistent trend in average summer temperature on the Beverly summer range over our study period (Fig. 5).
Significant (R2 = 0.20, F29 = 7.13, p = 0.01) relationship between the annual mean summer temperature on the Beverly summer range (data from CARMA caribou range climate database) and the summer (June, July, August) intensity of the Arctic Oscillation (AO), 1981–2011 (National Oceanic and Atmospheric Administration)
Significant relationship between the cumulative growing degrees above 5 °C (R2 = 0.21, F29 = 7.53, p = 0.01) on the Beverly summer range (data from CARMA caribou range climate database) and the summer (June, July, August) intensity of the Arctic Oscillation (AO), 1981–2011 (National Oceanic and Atmospheric Administration)
Beverly herd body condition
We constructed linear models of DFAT for each sex by age with the intensity of the AO (either in the winter, previous summer, or previous year) as predictor variables. The intensities of the AO in the winter and previous year were not significantly related to DFAT. However, DFAT increased with age in years with larger, positive intensities of the previous summer AO in both male (Table 1; R2 = 0.65, F25 = 23.04, p < 0.0001) and female (Table 1; R2 = 0.43, F36 = 13.50, p < 0.0001) caribou. To test the independent effect of the previous summer intensity of the AO, we also constructed a linear model of DFAT as predicted by age only for both male (Table 1; R2 = 0.42, F26 = 18.83, p < 0.001) and female (Table 1; R2 = 0.23, F37 = 11.17, p = 0.002) caribou. For both male and female caribou, models that included the previous summer intensity of the AO explained approximately 20% more variation in DFAT than models considering age of the animals alone. To test whether the relationship between climate and DFAT was not more parsimoniously explained by summer temperature rather than intensity of the AO, we constructed a linear model of DFAT for each sex with age and the mean temperature in the previous summer as predictor variables. For both sexes, models including intensity of AO rather than mean summer temperature explained greater variation in DFAT (Table 1). The intensity of the AO explained a greater amount of the variance in DFAT in male caribou than female caribou.
Arctic Oscillation and barren-ground caribou abundance
We identified a significant, positive association between the average summer intensity of the AO and the direction of population growth of the Bathurst, Beverly, and Qamanirjuaq herds (Table 2; exact binomial test, p = 0.0074). We demonstrate this relationship during both positive and negative phases of the AO, as well as through periods of low and high caribou abundance. Warmer temperatures and longer growing seasons corresponded to stable or increasing population growth across years and herds. Note that our analysis considered only the direction of population growth between abundance estimates and the signature (positive or negative) of the summer AO and did not take into account possible variation in growth rates during the period between survey estimates. We did not attempt to estimate specific growth rates between abundance estimates.
Discussion
We found a significant relationship between broad climate patterns and the population trajectories of three barren-ground caribou herds, consistent with previous studies of climate patterns and Rangifer population dynamics (Post and Stenseth 1999; Aanes et al. 2002; Forchhammer et al. 2002; Post and Forchhammer 2002; Joly et al. 2011). From 1988 to 1996, the summer intensity of the AO was largely in the positive phase, with a mean value of 0.207 (± 0.135 SE), and this was a period of population stability or growth for each of the three herds that we examined here. In contrast, from 1997 to 2016 the summer AO has remained largely in the negative phase, with a mean value of − 0.154 (± 0.077 SE), and over this period the Bathurst, Beverly, and Qamanirjuaq herds declined in abundance. We found that a greater proportion of the variation in body condition was explained for male caribou compared to females. This is likely because our model did not account for pregnant cows that tend to have higher body fat than non-pregnant cows (Thomas and Kiliaan 1998a).
Our results suggest that during periods of positive AO intensity, warmer temperatures on the summer range result in improved growing conditions for vascular plants that benefits foraging caribou. Conversely, negative summer AO intensity is associated with cooler temperatures with associated shorter growing seasons and increased precipitation on the Beverly summer range. Though we do not have direct measures of forage quality or quantity, previous studies have found that cooler and shorter summers tend to result in reduced vascular plant growth (Chapin and Shaver 1985; Rachlow and Bowyer 1998; Lenart et al. 2002; Van der Wal and Stien 2014), and therefore reduced forage quantity and availability (although climate can also affect forage species quality, e.g., Turunen et al. 2009; Mallory and Boyce 2018; Zamin et al. 2017). We did not investigate relationships between the AO and climate variables on the Bathurst or Qamanirjuaq ranges and have made the assumption that similar trends exist as on the Beverly range. We justify this assumption with the evidence of some range overlap (Fig. 2) between the three herds during the winter, spring, summer, and late summer periods (Campbell et al. in prep.).
From 1982 to 1987, differences in the physical condition of Beverly caribou corresponded with changes in the intensity and signature of the summer AO. We again reason that this can be explained by the effect of climate on the availability of forage (Chapin and Shaver 1985; Lenart et al. 2002; Van der Wal and Stien 2014) because caribou body condition is necessarily influenced by the availability of nutritious forage in summer (Reimers 1983; Crête and Huot 1993; Gerhart et al. 1996; Couturier et al. 2009; Tveraa et al. 2013; Albon et al. 2017). Caribou spend 7 months of the year with a negative protein balance, stemming from the low protein content of lichen, the primary constituent of their late-winter diet (Gerhart et al. 1996). Protein stores developed during the summer are not only necessary for caribou to maintain body condition over winter, but also for fetal growth and development in pregnant females. Fetal protein is synthesized from maternal tissues (Gerhart et al. 1996), and up to 80% of fetal mass is deposited in late winter, when caribou diet provides little protein (Côté et al. 2012).
In addition to the relationships between female nutrition and fetal development, the physical condition of pregnant cows also has been shown to affect calf survival (Bergerud 1996), and reduced body condition of individuals can lead to changes in population demography through effects on survival and reproduction (Crête and Huot 1993; Mahoney and Schaefer 2002; Albon et al. 2017). The physical condition of female caribou is positively linked to their reproductive success (Cameron et al. 1993; Gerhart et al. 1997; Tveraa et al. 2013), a relationship that has been documented at the population level in Beverly caribou (Thomas and Kiliaan 1998b). Between 1980 and 1987, years with fatter female caribou corresponded to higher fecundity across the herd (Thomas and Kiliaan 1998b). These findings allow us to propose a mechanism where positive AO intensity in the summer corresponds to warmer average temperatures and a longer growing season and improved foraging conditions for caribou. This leads to caribou in better physical condition going into winter, perhaps reducing adult winter mortality, and increasing rates of fecundity and late-winter calf survival. Conditions during periods of positive AO intensity then contribute to stable or increasing population growth in these caribou herds. During periods of negative AO intensity, summers are cooler and growing seasons shorter, which then contributes to reduced quality, quantity, and availability of preferred forage species, poorer condition of caribou, lower rates of fecundity, and negative population growth. We note that this proposed mechanism assumes that a similar relationship exists between the AO and condition of Bathurst and Qamanirjuaq caribou to the one observed with Beverly caribou between 1982 and 1987. We support this assumption with the relative proximity of the herd summer ranges (i.e., in terms of broad climate patterns), and the well-documented relationships between climate, growing season length, availability of summer forage, and caribou condition (Reimers 1983; Chapin and Shaver 1985; Crête and Huot 1993; Lenart et al. 2002; Cebrian et al. 2008; Couturier et al. 2009; Van der Wal and Stien 2014; Tveraa et al. 2013; Albon et al. 2017).
The dynamics of barren-ground caribou populations are the result of many factors, both density-dependent and stochastic (Bergerud 1996; Sæther 1997; Gunn 2003). Due to the limited data that are available for the Qamanirjuaq, Beverly, and Bathurst herds, we were unable to incorporate factors such as predation and insect harassment in our analysis. Although no substitute for these and other important factors exist, broad climate indices provide useful proxies that can be used to approximate a variety of environmental conditions that are important to caribou, such as forage quality and availability, temperature, snow depth, and precipitation. Caribou population fluctuations have now been linked to large-scale climate patterns across their circumpolar range, from the PDO in Alaska (Joly et al. 2011), the AO in the central Canadian Arctic (Zalatan 2006; this study), and the NAO in the Greenland and Norway (Post and Stenseth 1999; Aanes et al. 2002). This collective evidence suggests that the effects of broad climate patterns could play an important role as a driver of cyclical fluctuations in caribou populations. Other mechanisms, such as density-dependent forage exploitation, have been identified as important contributors to these fluctuations for some herds (Messier et al. 1988), but could be less important for other caribou populations (Rickbeil et al. 2015). The growing number of studies identifying a link between climate patterns and fluctuations in caribou abundance provide compelling support for climate as an important driver of barren-ground caribou cycling, a link that helps to explain global patterns (Vors and Boyce 2009), and also allows for local and regional differences in caribou population trends (Joly et al. 2011). The observed synchrony among adjacent herds is consistent with the well-documented influence of climate-caused perturbations on population synchrony (e.g., Moran 1953; Ranta et al. 1997; Post and Forchhammer 2002; Liebhold et al. 2004). Although we have insufficient data to rule out alternative hypotheses, if we can assume comparable mechanisms of population regulation among herds, we speculate that a Moran effect could contribute a theoretical underpinning to our results.
We suggest that the effects on forage of these climate patterns contribute to long-term fluctuations in caribou abundance and are of crucial importance to caribou population dynamics because they likely increase the vulnerability or resilience of caribou to other mechanisms affecting population decline and growth such as predation, insect harassment, and disease (Bergerud 1996). However, we acknowledge the potential significance of other mechanisms, including anthropogenic disturbance, which through modifications of caribou behavior and range can overwhelm other drivers and lead to less-predictable effects on caribou condition, productivity, and ultimately abundance. Although the intensity of the summer AO has been predominantly negative over the past 20 years, long-term fluctuations of the AO are such that it will again return to a predominantly positive phase, and this could be beneficial to barren-ground caribou by affording conditions that support improved forage productivity, and in turn, reproductive productivity. However, we caution that the effects of a changing climate and warming Arctic on the relationships that we identify here are unclear. Climate change is likely to significantly alter the baseline environmental conditions to which this species is adapted, and the oscillations of broad-scale climate patterns around this baseline might further disrupt ecosystems. For example, increasingly warm summers are predicted to increase the prevalence of parasitic insects in Arctic regions and allow other parasitic species to expand the northern extent of their range (Callaghan et al. 2004; Kutz et al. 2013). Harassment by insects is a major energetic cost to caribou (Helle and Tarvainen 1984; Hagemoen and Reimers 2002; Witter et al. 2012; Mallory and Boyce 2018), and one that might outweigh the benefits of potential increases in forage productivity associated with warmer summers. Further, improved forage associated with positive intensities of the summer AO might shift should climate change alter caribou summer range quality through changing plant community composition (e.g., increased abundance of shrubs; Myers-Smith et al. 2011; Sturm et al. 2001; Frost and Epstein 2014) and reduced forage quality (Turunen et al. 2009; Thompson and Barboza 2014; Zamin et al. 2017). In fact, under a changing environmental baseline we speculate that the relationships we identified here might be altered, even reversed, and summers with negative AO intensity that are cooler than average could potentially correlate positively to caribou life-history traits. In light of this, while we suggest that wildlife managers could consider using the AO as an index of the vulnerability of the Bathurst, Beverly, and Qamanirjuaq herds, this must be done carefully and in consideration of the implications of a changing climate and other potential mechanisms affecting caribou and their habitats.
References
Aanes R, Sæther B-E, Smith FM, Cooper EJ, Wookey PA, Øritslandet NA (2002) The Arctic Oscillation predicts effects of climate change in two trophic levels in a high-arctic ecosystem. Ecol Lett 5:445–453
Adamczewski JZ, Gates CC, Soutar BM, Hudson RJ (1986) Limiting effects of snow on seasonal habitat use and diets of caribou (Rangifer tarandus groenlandicus) on Coats Island, Northwest Territories, Canada. Can J Zool 66:1986–1996
Adamczewski JZ, Gates CC, Hudson RJ (1987) Fat distribution and indices of carcass composition in Coats Island caribou (Rangifer tarandus groenlandicus). Can J Zool 65:368–374
Adamczewski JZ, Gunn A, Poole KG, Hall A, Nishi J, Boulanger J (2015) What happened to the Beverly caribou herd after 1994? Arctic 68:407–421
Albon SD et al (2017) Contrasting effects of summer and winter warming on body mass explain population dynamics in a food-limited Arctic herbivore. Glob Change Biol 23:1374–1389
Banfield AWF (1954) Preliminary investigations of the barren-ground caribou. Part I. Former and present distribution, migrations, and status. Can Wildl Serv Wildlife Management Bulletin Series 1, No. 10A
Bergerud AT (1996) Evolving perspectives on caribou population dynamics, have we got it right yet? Rangifer 16:95–116
Bergerud AT, Ballard WB (1988) Wolf predation on caribou: the Nelchina herd case history, a different interpretation. J Wildl Manage 52:344–357
Boulanger J (2015) Interim report: estimates of breeding females and herd size from the 2015 Bathurst calving ground survey
Boulanger J, Gunn A, Azamczewski JZ, Croft B (2011) A data-driven demographic model to explore the decline of the Bathurst caribou herd. J Wildl Manage 75:883–896
Boulanger J, Croft B, Adamczewski JZ (2014) An estimate of breeding females and analyses of demographics for the Bathurst Herd of barren-ground caribou: 2012 calving ground photographic survey. File report no. 142. Dept Env and Nat Res, Gov of NWT. Yellowknife
Callaghan TV et al (2004) Key findings and extended summaries. Ambio 33:386–392
Cameron RD, Smith WT, Fancy SF, Gerhart KL, White RG (1993) Calving success of female caribou in relation to body weight. Can J Zool 71:480–486
Campbell MW, Nishi J, Boulanger J (2010) A calving ground photo survey of the Qamanirjuaq migratory barren-ground caribou (Rangifer tarandus groenlandicus) population—June 2008. Gov of Nunavut Dept of Env technical report series no. 01-2010
Campbell MW, Boulanger J, Lee DS, Dumond M, McPherson J (2012) Calving ground abundance estimates of the Beverly and Ahiak Subpopulations of Barren-Ground Caribou (Rangifer tarandus groenlandicus)—June 2011. Gov of Nunavut Dept of Env technical report series no. 03-2012
Campbell MW, Boulanger J, Lee DS (2016) Estimating abundance of the Qamanirjuaq Mainland Migratory Barren-Ground Caribou Subpopulation—June 2014. Gov of Nunavut Dept of Env technical report series no. 01-2016
Cebrian MR, Kielland K, Finstad G (2008) Forage quality and reindeer productivity: multiplier effects amplified by climate change. Arct Antarct Alp Res 40:48–54
Chan K-S, Mysterud A, Øritsland NA, Severinsen T, Stenseth NC (2005) Continuous and discrete extreme climatic events affecting the dynamics of a high-arctic reindeer population. Oecologia 145:556–563
Chapin FS III, Shaver GR (1985) Individualistic growth response of tundra plant species to environmental manipulations in the field. Ecology 66:564–576
Côté SD et al (2012) Caribou herd dynamics: impacts of climate change on traditional and sport harvesting. In: Allard M, Lemay M (eds) Nunavik and Nunatsiavut: From science to policy. An Integrated Regional Impact Study (IRIS) of climate change and modernization. ArcticNet Inc., Quebec, pp 249–269
Couturier S, Côté SD, Otto RD, Weladji RB, Huot J (2009) Variation in calf body mass in migratory caribou: the role of habitat, climate, and movements. J Mammal 90:442–452
Crête M, Huot J (1993) Regulation of a large herd of migratory caribou: summer nutrition affects calf growth and body reserves of dams. Can J Zool 71:2291–2296
Fauchald P, Park T, Tømmervik H, Myneni R, Hausner VH (2017) Arctic greening from warming promotes declines in caribou populations. Sci Adv 3:e1601365
Forchhammer MC, Post E, Stenseth NC, Boertmann DM (2002) Long-term responses in arctic ungulate dynamics to changes in climatic and trophic processes. Popul Ecol 44:113–120
Frost GV, Epstein HE (2014) Tall shrub and tree expansion in Siberian tundra ecotones since the 1960s. Glob Change Biol 20:1264–1277
Gerhart KL, White RG, Cameron RD, Russell DE (1996) Body composition and nutrient reserves of arctic caribou. Can J Zool 74:136–146
Gerhart KL, Russell DE, Van DeWetering D, White RG, Cameron RD (1997) Pregnancy of adult caribou (Rangifer tarandus): evidence for lactational infertility. J Zool 242:17–30
Griffith B et al. (2002) Section 3: the porcupine caribou herd. Biological Science Report USGS/BRD/BSR-2002-0001 837. US Geological Survey, Biological Resources Division
Gunn A (2003) Voles, lemmings and caribou—population cycles revisited? Rangifer 23:105–111
Gunn A, Dragon J, Nishi J (1996) Bathurst calving ground survey 1996. Dept of Res, Wildl and Econ Dev, Gov of NWT, Yellowknife, pp 30–46
Gunn A, Russell D, Eamer J (2011) Northern caribou population trends in Canada. Canadian biodiversity: ecosystem status and trends 2010, technical thematic report no. 10. Canadian Councils of Resource Ministers, Ottawa
Hagemoen RIM, Reimers E (2002) Reindeer summer activity pattern in relation to weather and insect harassment. J Anim Ecol 71:883–892
Heard DC, Williams TM (1991a) Appendix C. Bathurst calving ground survey, June 1986. In: Gunn A et al. (eds) Bathurst calving ground survey 1996. Dept of Res, Wildl and Econ Dev, Gov of NWT, Yellowknife, pp 30–46
Heard DC, Williams TM (1991b) Appendix D. Bathurst calving ground survey, June 1990. In: Gunn A et al. (eds) Bathurst calving ground survey 1996. Dept of Res, Wildl and Econ Dev, Gov of NWT, Yellowknife, pp 47–62
Hegel TM, Mysterud A, Ergon T, Loe LE, Huettmann F, Stenseth NC (2010) Seasonal effects of Pacific-based climate on recruitment in a predator-limited large herbivore. J Anim Ecol 79:471–482
Helle T, Tarvainen L (1984) Effects of insect harassment on weight gain and survival in reindeer calves. Rangifer 4:24–27
Hughes J, Albon SD, Irvine RJ, Woodin S (2009) Is there a cost of parasites to caribou? Parasitology 136:253–265
Hurrell JW (1995) Decadal trends in the North Atlantic Oscillation: regional temperatures and precipitation. Science 269:676–679
Joly K, Klein DR, Verbyla DL, Rupp TS, Chapin FS III (2011) Linkages between large-scale climate patterns and the dynamics of Arctic caribou populations. Ecography 34:345–352
Klein DR (1991) Limiting factors in caribou population ecology. Rangifer 7:30–35
Kutz SJ et al (2013) Invasion, establishment, and range expansion of two parasitic nematodes in the Canadian Arctic. Glob Change Biol 19:3254–3262
Lenart EA, Bowyer RT, Ver Hoef J, Ruess RW (2002) Climate change and caribou: effects of summer weather on forage. Can J Zool 80:664–678
Liebhold A, Koenig WD, Bjørnstad ON (2004) Spatial synchrony in population dynamics. Annu Rev Ecol Evol Syst 35:467–490
Mahoney SP, Schaefer JA (2002) Changes in demography and migration of Newfoundland caribou. J Mammal 83:957–963
Mallory CD, Boyce MS (2018) Observed and predicted effects of climate change on Arctic caribou and reindeer. Environ Rev. https://doi.org/10.1139/er-2017-0032
Manseau M, Huot J, Crete M (1996) Effects of summer grazing by caribou on composition and productivity of vegetation: community and landscape level. J Ecol 84:503–513
Mantua NJ, Hare SR, Zhang Y, Wallace JM, Francis RC (1997) A Pacific interdecadal climate oscillation with impacts on salmon production. Bull Am Meteorol Soc 78:1069–1079
Messier F, Huot J, Le Henaff D, Luttich S (1988) Demography of the George River Caribou Herd: evidence of population regulation by forage exploitation and range expansion. Arctic 41:279–287
Moran PAP (1953) The statistical analysis of the Canadian lynx cycle. II. Synchronization and meteorology. Aust J Zool 1:291–298
Myers-Smith IH et al (2011) Shrub expansion in tundra ecosystems: dynamics, impacts and research priorities. Environ Res Lett 6:045509
Nagy JA, Johnson DL, Larter NC, Campbell MW, Derocher AE, Kelly A, Dumond M, Allaire D, Croft B (2011) Subpopulation structure of caribou (Rangifer tarandus L.) in arctic and subarctic Canada. Ecol Appl 21:2334–2348
Post E, Forchhammer MC (2002) Synchronization of animal population dynamics by large-scale climate. Nature 420:168–171
Post E, Stenseth NC (1999) Climatic variability, plant phenology, and northern ungulates. Ecology 80:1322–1339
Rachlow JL, Bowyer RT (1998) Habitat selection by Dall’s sheep (Ovis dalli): maternal trade-offs. J Zool 245:457–465
Ranta E, Kaitala V, Lindstrӧm J, Helle E (1997) The Moran effect and synchrony in population dynamics. Oikos 78:136–142
Reimers E (1983) Growth rate and body size differences in Rangifer: a study of cause and effect. Rangifer 3:3–15
Rickbeil GJM, Coops NC, Adamczewski JZ (2015) The grazing impacts of four barren ground caribou herds (Rangifer tarandus groenlandicus) on their summer ranges: an application of archived remotely sensed vegetation productivity data. Remote Sens Environ 164:314–323
Rienecker MM et al (2011) MERRA: NASA’s modern-era retrospective analysis for research and applications. J Clim 24:3624–3648
Russell DE, Whitfield PH, Cai J, Gunn A, White RG, Poole K (2013) CARMA’s MERRA-based caribou range climate database. Rangifer 33:145–152
Sæther B-E (1997) Environmental stochasticity and population dynamics of large herbivores: a search for mechanisms. Trends Ecol Evol 12:143–149
Sturm M, Racine C, Tape K (2001) Climate change: increasing shrub abundance in the Arctic. Nature 411:546–547
R Core Team (2016) R: a language and environment for statistical computing. R Foundation for Statistical Computing, Vienna. https://www.R-project.org/
Thomas DC, Kiliaan HPL (1998a) Fire-caribou relationships: (I) physical characteristics of the Beverly herd, 1980–1987. Tech rep series no. 309. Can Wildl Serv, Prairie and Northern Reg
Thomas DC, Kiliaan HPL (1998b) Fire-caribou relationships: (II) fecundity and physical condition of the Beverly herd, 1980–1987. Tech rep series no. 310. Can Wildl Serv, Prairie and Northern Reg
Thompson DP, Barboza PS (2014) Nutritional implications of increased shrub cover for caribou (Rangifer tarandus) in the Arctic. Can J Zool 92:339–351
Thompson DWJ, Wallace JM (1998) The Arctic Oscillation signature in the wintertime geopotential height and temperature fields. Geophys Res Lett 25:1297–1300
Thompson DWJ, Wallace JM, Hegerl GC (2000) Annular modes in the extratropical circulation. Part II: trends*. J Clim 13:1018–1036
Turunen M, Soppela P, Kinnunen H, Sutinen M-L, Martz M (2009) Does climate change influence the availability and quality of reindeer forage plants? Polar Biol 32:813–832
Tveraa T, Stien A, Bårdsen B-J, Fauchald P (2013) Population densities, vegetation green-up, and plant productivity: impacts on reproductive success and juvenile body mass in reindeer. PLoS ONE 8(2):e56450
Van der Wal R, Stien A (2014) High-arctic plants like it hot: a long-term investigation of between-year variability in plant biomass. Ecology 95:3414–3427
Vors LS, Boyce MS (2009) Global declines of caribou and reindeer. Glob Change Biol 15:2626–2633
Weladji RB, Holand Ø, Almøy T (2003) Use of climatic data to assess the effect of insect harassment on the autumn weight of reindeer (Rangifer tarandus) calves. J Zool 260:79–85
Witter LA, Johnson CJ, Croft B, Gunn A, Gillingham MP (2012) Behavioural trade-offs in response to external stimuli: time allocation of an Arctic ungulate during varying intensities of harassment by parasitic flies. J Anim Ecol 81:284–295
Zalatan R (2006) The relation between climate and abundance cycles in barren-ground caribou herds of the Northwest Territories, Canada. Thesis, University of British Columbia
Zamin TJ, Côté SD, Tremblay J-P, Grogan P (2017) Experimental warming alters migratory caribou forage quality. Ecol Appl. https://doi.org/10.1002/eap.1590
Acknowledgements
The Natural Sciences and Engineering Research Council of Canada and the Government of Nunavut provided funding for this study. We thank D. Russell and the CARMA network for producing and providing the caribou range climate database. We acknowledge and thank the many researchers whose work made our study possible. We thank M. Scrafford, M. Mallory, and the anonymous reviewers who provided comments on earlier versions of this manuscript.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Mallory, C.D., Campbell, M.W. & Boyce, M.S. Climate influences body condition and synchrony of barren-ground caribou abundance in Northern Canada. Polar Biol 41, 855–864 (2018). https://doi.org/10.1007/s00300-017-2248-3
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00300-017-2248-3