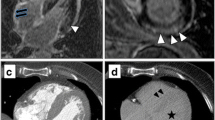
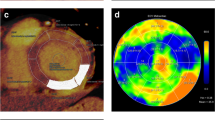
Abstract
Objective
To measure myocardium iodine concentration (MIC) in patients with cardiac amyloidosis (CA) using dual-energy computed tomography (DECT).
Methods
Twenty-two patients with CA, 13 with non-amyloid hypertrophic cardiomyopathies (CH) and 10 control patients were explored with pre-contrast, arterial and 5-minute DECT acquisition (Iomeprol; 1.5 mL/kg). Inter-ventricular septum (IVS) thickness, blood pool iodine concentration (BPIC), MIC (mg/mL), iodine ratio and extra-cellular volume (ECV) were calculated.
Results
IVS thickness was significantly (p < 0.001) higher in CA (17 ± 4 mm) and CH (15 ± 3 mm) patients than in control patients (10 ± 1 mm). CA patients exhibited significantly (p < 0.001) higher 5-minute MIC [2.6 (2.3–3.1) mg/mL], 5-minute iodine ratio (0.88 ± 0.12) and ECV (0.56 ± 0.07) than CH [1.7 (1.4–2.2) mg/mL, 0.57 ± 0.07 and 0.36 ± 0.05, respectively] and control patients [1.9 (1.7–2.4) mg/mL, 0.58 ± 0.07 and 0.35 ± 0.04, respectively). CH and control patients exhibited similar values (p = 0.9). The area under the curve of 5-minute iodine ratio for the differential diagnosis of CA from CH patients was 0.99 (0.73–1.0; p = 0.001). With a threshold of 0.65, the sensitivity and specificity of 5-minute iodine ratio were 100% and 92%, respectively.
Conclusion
Five-minute MIC and iodine ratio were increased in CA patients and exhibited best diagnosis performance to diagnose CA in comparison to other parameters.
Key point
• Dual-energy computed tomography can be used to detect cardiac amyloidosis
• Five-minute myocardial iodine concentration and iodine ratio increase in cardiac amyloidosis
• Among iodine parameters, 5-minute iodine ratio has the best diagnosis performance


Similar content being viewed by others
Abbreviations
- BPIC:
-
Blood pool iodine concentration
- CA:
-
Cardiac amyloidosis
- CH:
-
Cardiac hypertrophy
- DECT:
-
Dual-energy computed tomography
- EMB:
-
Endo-myocardial biopsy
- ECV:
-
Extra-cellular volume
- GSI:
-
Gemstone spectral imaging
- HCM:
-
Hypertrophic cardiomyopathy
- IVS:
-
Inter-ventricular septum
- AL:
-
Light-chain amyloidosis
- MIC:
-
Myocardium iodine concentration
- ATTR:
-
Transthyretin amyloidosis
References
Gertz MA, Benson MD, Dyck PJ et al (2015) Diagnosis, Prognosis, and Therapy of Transthyretin Amyloidosis. J Am Coll Cardiol 66:2451–2466
Wechalekar AD, Gillmore JD, Hawkins PN (2016) Systemic amyloidosis. Lancet Lond Engl 387:2641–2654
Quarta CC, Kruger JL, Falk RH (2012) Cardiac amyloidosis. Circulation 126:e178–e182
White JA, Fine NM (2016) Recent Advances in Cardiovascular Imaging Relevant to the Management of Patients with Suspected Cardiac Amyloidosis. Curr Cardiol Rep 18:77
Quintana-Quezada RA, Yusuf SW, Banchs J (2016) Use of Noninvasive Imaging in Cardiac Amyloidosis. Curr Treat Options Cardiovasc Med 18:46
Deux J-F, Damy T, Rahmouni A et al (2014) Noninvasive detection of cardiac involvement in patients with hereditary transthyretin associated amyloidosis using cardiac magnetic resonance imaging: a prospective study. Amyloid Int J Exp Clin Investig Off J Int Soc Amyloidosis 21:246–255
Gillmore JD, Maurer MS, Falk RH et al (2016) Nonbiopsy Diagnosis of Cardiac Transthyretin Amyloidosis. Circulation 133:2404–2412
Falk RH, Quarta CC (2015) Echocardiography in cardiac amyloidosis. Heart Fail Rev 20:125–131
Bandula S, White SK, Flett AS et al (2013) Measurement of myocardial extracellular volume fraction by using equilibrium contrast-enhanced CT: validation against histologic findings. Radiology 269:396–403
Kammerlander AA, Marzluf BA, Zotter-Tufaro C et al (2016) T1 Mapping by CMR Imaging: From Histological Validation to Clinical Implication. JACC Cardiovasc Imaging 9:14–23
Banypersad SM, Sado DM, Flett AS et al (2013) Quantification of myocardial extracellular volume fraction in systemic AL amyloidosis: an equilibrium contrast cardiovascular magnetic resonance study. Circ Cardiovasc Imaging 6:34–39
Boynton SJ, Geske JB, Dispenzieri A et al (2016) LGE Provides Incremental Prognostic Information Over Serum Biomarkers in AL Cardiac Amyloidosis. JACC Cardiovasc Imaging 9:680–686
Neilan TG, Coelho-Filho OR, Shah RV et al (2013) Myocardial extracellular volume fraction from T1 measurements in healthy volunteers and mice: relationship to aging and cardiac dimensions. JACC Cardiovasc Imaging 6:672–683
Karamitsos TD, Piechnik SK, Banypersad SM et al (2013) Noncontrast T1 mapping for the diagnosis of cardiac amyloidosis. JACC Cardiovasc Imaging 6:488–497
den Harder AM, Willemink MJ, de Jong PA et al (2016) New horizons in cardiac CT. Clin Radiol 71:758–767
Langer C, Both M, Harders H et al (2015) Late enhanced computed tomography in Hypertrophic Cardiomyopathy enables accurate left-ventricular volumetry. Eur Radiol 25:575–584
Delgado Sánchez-Gracián C, Oca Pernas R, Trinidad López C et al (2016) Quantitative myocardial perfusion with stress dual-energy CT: iodine concentration differences between normal and ischemic or necrotic myocardium. Initial experience. Eur Radiol 26:3199–3207
Patino M, Prochowski A, Agrawal MD et al (2016) Material Separation Using Dual-Energy CT: Current and Emerging Applications. Radiogr Rev Publ Radiol Soc N Am Inc 36:1087–1105
Lee H-J, Im DJ, Youn J-C et al (2016) Myocardial Extracellular Volume Fraction with Dual-Energy Equilibrium Contrast-enhanced Cardiac CT in Nonischemic Cardiomyopathy: A Prospective Comparison with Cardiac MR Imaging. Radiology 280:49–57
Schwarz F, Ruzsics B, Schoepf UJ et al (2008) Dual-energy CT of the heart--principles and protocols. Eur J Radiol 68:423–433
Treibel TA, Bandula S, Fontana M et al (2015) Extracellular volume quantification by dynamic equilibrium cardiac computed tomography in cardiac amyloidosis. J Cardiovasc Comput Tomogr 9:585–592
Deux J-F, Mihalache C-I, Legou F et al (2015) Noninvasive detection of cardiac amyloidosis using delayed enhanced MDCT: a pilot study. Eur Radiol 25:2291–2297
Nacif MS, Kawel N, Lee JJ et al (2012) Interstitial myocardial fibrosis assessed as extracellular volume fraction with low-radiation-dose cardiac CT. Radiology 264:876–883
Kristen AV, Brokbals E, Aus dem Siepen F et al (2016) Cardiac Amyloid Load: A Prognostic and Predictive Biomarker in Patients With Light-Chain Amyloidosis. J Am Coll Cardiol 68:13–24
Nacif MS, Liu Y, Yao J et al (2013) 3D left ventricular extracellular volume fraction by low-radiation dose cardiac CT: assessment of interstitial myocardial fibrosis. J Cardiovasc Comput Tomogr 7:51–57
Wong TC, Piehler K, Meier CG et al (2012) Association between extracellular matrix expansion quantified by cardiovascular magnetic resonance and short-term mortality. Circulation 126:1206–1216
Fontana M (2016) Prognosis in Cardiac Amyloidosis by LGE: Ready for Prime Time? JACC Cardiovasc Imaging 9:687–689
Gallego-Delgado M, González-López E, Muñoz-Beamud F et al (2016) Extracellular volume detects amyloidotic cardiomyopathy and correlates with neurological impairment in transthyretin-familial amyloidosis. Rev Esp Cardiol (Engl Ed). doi:10.1016/j.rec.2016.02.027
Fontana M, Banypersad SM, Treibel TA et al (2015) Differential Myocyte Responses in Patients with Cardiac Transthyretin Amyloidosis and Light-Chain Amyloidosis: A Cardiac MR Imaging Study. Radiology 277:388–397
Fontana M, Pica S, Reant P et al (2016) Response to Letters Regarding Article, “Prognostic Value of Late Gadolinium Enhancement Cardiovascular Magnetic Resonance in Cardiac Amyloidosis.”. Circulation 133:e450–e451
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Guarantor
The scientific guarantor of this publication is Jean-François Deux.
Conflict of interest
The authors of this manuscript declare no relationships with any companies whose products or services may be related to the subject matter of the article.
Funding
The authors state that this work has not received any funding.
Statistics and biometry
One of the authors has significant statistical expertise.
Ethical approval
Institutional review board approval was obtained.
Informed consent
Written informed consent was obtained from all subjects (patients) in this study.
Methodology
• prospective
• experimental
• performed at one institution
Rights and permissions
About this article
Cite this article
Chevance, V., Damy, T., Tacher, V. et al. Myocardial iodine concentration measurement using dual-energy computed tomography for the diagnosis of cardiac amyloidosis: a pilot study. Eur Radiol 28, 816–823 (2018). https://doi.org/10.1007/s00330-017-4984-8
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00330-017-4984-8