Abstract
Objectives
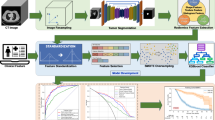
It is of high clinical importance to identify the primary lesion and its pathological types for patients with brain metastases (BM). The purpose of this study is to investigate the feasibility and accuracy of differentiating the primary adenocarcinoma (AD) and squamous cell carcinoma (SCC) of non-small-cell lung cancer (NSCLC) for patients with BM based on radiomics from brain contrast-enhanced computer tomography (CECT) images.
Methods
A total of 144 BM patients (94 male, 50 female) were enrolled in this study with 102 with primary lung AD and 42 with SCC, respectively. Radiomics features from manually contoured tumors were extracted using python. Mann–Whitney U test and the least absolute shrinkage and selection operator (LASSO) logistic regression were applied to select relative radiomics features. Binary logistic regression and support vector machines (SVM) were applied to build models with radiomics features alone and with radiomics features plus age and sex.
Results
Fourteen features were selected from a total of 105 radiomics features for the final model building. The area under the curves (AUCs) and accuracy of SVM and binary logistic regression models were 0.765 vs. 0.769, 0.795 vs.0.828, and 0.716 vs. 0.726, 0.768 vs. 0.758, respectively, for models with radiomics features alone and models with radiomics features plus sex and age.
Conclusions
Brain CECT radiomics are promising in differentiating primary AD and SCC to achieve optimal therapeutic management in patients with BM from NSCLC.
Key Points
• It is of high clinical importance to identify the primary lesion and its pathological types for patients with brain metastases (BM) to define the prognosis and treatment.
• Few studies had investigated the feasibility and accuracy of differentiating the pathological subtypes of primary non-small-cell lung cancer between adenocarcinoma (AD) and squamous cell carcinoma (SCC) for patients with BM based on radiomics from brain contrast-enhanced CT (CECT) images, although CECT images are often the initial imaging modality to screen for metastases and are recommended on equal footing with MRI for the detection of cerebral metastases.
• Brain CECT radiomics are promising in differentiating primary AD and SCC to achieve optimal therapeutic management in patients with BM from NSCLC with a highest area under the curve (AUC) of 0.828 and an accuracy of 0.758, respectively.




Similar content being viewed by others
Abbreviations
- AD:
-
Adenocarcinoma
- AUC:
-
Area under the curve
- BM:
-
Brain metastases
- CECT:
-
Contrast-enhanced computer tomography
- ECCR:
-
Ethics Committee in Clinical Research
- GLCM:
-
Gray-level co-occurrence matrix
- GLRLM:
-
Gray-level run-length matrix
- GLZLM:
-
Gray-level zone length matrix
- LASSO:
-
Least absolute shrinkage and selection operator
- MRI:
-
Magnetic resonance imaging
- NGLDM:
-
Neighborhood gray-level different matrix
- NSCLC:
-
Non-small-cell lung cancer
- ROC:
-
Receiver operating characteristic
- SCC:
-
Squamous cell carcinoma
- SCLC:
-
Small cell lung cancer
- SVM:
-
Support vector machines
- TTF-1:
-
Thyroid transcription factor-1
References
Norden AD, Wen PY, Kesari S (2005) Brain metastases. Curr Opin Neurol 18(6):654–661
Wen PY, Black PM, Loeffler JS (2001) Metastatic brain cancer. In: DeVita V, Hellman S, Rosenberg SA (eds) Cancer: principles and practice of oncology, 6th edn. Lippincott, WIlliams, Philadelphia, pp 2655–2670
Greenberg H, Chandler WF, Sandler HM (1999) Brain metastases. In: Brain Tumors. Oxford University Press, New York, pp 299–317
Zimm S, Wampler GL, Stablein D, Hazra T, Young HF (1981) Intracerebral metastases in solid-tumor patients: natural history and results of treatment. Cancer 48(2):384–394
Sundström JT, Minn H, Lertola KK, Nordman E (1998) Prognosis of patients treated for intracranial metastases with whole-brain irradiation. Ann Med 30(3):296–299
Gore ME, Szczylik C, Porta C et al (2009) Safety and efficacy of sunitinib for metastatic renal-cell carcinoma: an expanded-access trial. Lancet Oncol 10(8):757e63
Robinson SD, O’Shaughnessy JA, Lance Cowey C, Konduri K (2014) BRAFV600E-mutated lung adenocarcinoma with metastases to the brain responding to treatment with vemurafenib. Lung Cancer 85(2):326e30
Bachelot T, Romieu G, Campone M et al (2013) Lapatinib plus capecitabine in patients with previously untreated brain metastases from HER2-positive metastatic breast cancer (LANDSCAPE): a single-group phase 2 study. Lancet Oncol 14(1):64e71
Margolin K, Ernstoff MS, Hamid O et al (2012) Ipilimumab in patients with melanoma and brain metastases: an open-label, phase 2 trial. Lancet Oncol 13(5):459e65
Drlicek M, Bodenteich A, Urbanits S, Grisold W (2004) Immunohistochemical panel of antibodies in the diagnosis of brain metastases of the unknown primary. Pathol Res Pract 200(10):727–734
Soffietti R, Cornu P, Delattre JY et al (2006) EFNS Guidelines on diagnosis and treatment of brain metastases: report of an EFNS Task Force. Eur J Neurol 13:674–681
Molina JR, Yang P, Cassivi SD, Schild SE, Adjei AA (2008) Non-small cell lung cancer: epidemiology, risk factors, treatment, and survivorship. Mayo Clin Proc 83(5):584–594
Kim HS, Mitsudomi T, Soo RA, Cho BC (2013) Personalized therapy on the horizon for squamous cell carcinoma of the lung. Lung Cancer 80(3):249–255
Sperduto PW, Yang TJ, Beal K et al (2017) Estimating survival in patients with lung cancer and brain metastases: an update of the graded prognostic assessment for lung cancer using molecular markers (LungmolGPA). JAMA Oncol 3(6):827e31
Wu CC, Maher MM, Shepard JA (2011) Complications of CT-guided percutaneous needle biopsy of the chest: prevention and management. AJR Am J Roentgenol 196(6):W678–W682
Gillies RJ, Kinahan PE, Hricak H (2016) Radiomics: images are more than pictures, they are data. Radiology 278:563–577
Kniep HC, Madesta F, Schneider T et al (2019) Radiomics of brain MRI: utility in prediction of metastatic tumor type. Radiology 290(2):479–487
Li Z, Mao Y, Li H, Yu G, Wan H, Li B (2016) Differentiating brain metastases from different pathological types of lung cancers using texture analysis of T1 postcontrast MR. Magn Reson Med 76(5):1410–1419
Silvestri GA, Gould MK, Margolis ML et al (2007) Noninvasive staging of non-small cell lung cancer: ACCP evidenced-based clinical practice guidelines (2nd edition). Chest 132(3 suppl):178S–1201S
Barajas RF, Cha S (2012) Imaging diagnosis of brain metastasis. Prog Neurol Surg 25:55–73
Van Griethuysen JJM, Fedorov A, Parmar C et al (2017) Computational radiomics system to decode the radiographic phenotype. Cancer Res 77(21):e104–e107
Friedman J, Hastie T, Tibshirani R (2010) Regularization paths for generalized linear models via coordinate descent. J Stat Softw 33:1
Burges CJC (1998) A tutorial on support vector machines for pattern recognition. Data Min Knowl Discov 2:121–167
Agazzi S, Pampallona S, Pica A et al (2004) The origin of brain metastases in patients with an undiagnosed primary tumour. Acta Neurochir (Wien) 146(2):153–157
Jin J, Zhou X, Liang X et al (2013) Brain metastases as the first symptom of lung cancer: a clinical study from an Asian medical center. J Cancer Res Clin Oncol 139(3):403–408
Giese A, Westphal M (2001) Treatment of malignant glioma: A problem beyond the margins of resection. J Cancer Res Clin Oncol 127:217–225
Kawaguchi KR, Lu FI, Kaplan R et al (2014) In search of the ideal immunopanel to distinguish metastatic mammary carcinoma from primary lung carcinoma: a tissue microarray study of 207 cases. Appl Immunohistochem Mol Morphol 22(4):266–274
Bekaert L, Emery E, Levallet G, Lechapt-Zalcman E (2017) Histopathologic diagnosis of brain metastases: current trends in management and future considerations. Brain Tumor Pathol 34(1):8–19
Malone H, Yang J, Hershman DL, Wright JD, Bruce JN, Neugut AI (2015) Complications following stereotactic needle biopsy of intracranial tumors. World Neurosurg 84:1084–1089
Chand P, Amit S, Gupta R, Agarwal A (2016) Errors, limitations, and pitfalls in the diagnosis of central and peripheral nervous system lesions in intraoperative cytology and frozen sections. J Cytol 33:93
Funding
This work was partially funded by Wenzhou Municipal Science and Technology Bureau (Nos. 2018ZY016 and H20180003) and National Natural Science Foundation of China under Grant (No. 11675122).
Author information
Authors and Affiliations
Corresponding authors
Ethics declarations
Guarantor
The scientific guarantor of this publication is Xiance Jin.
Conflict of interest
The authors of this manuscript declare no relationships with any companies whose products or services may be related to the subject matter of the article.
Statistics and biometry
One of the authors has significant statistical expertise.
Informed consent
Written informed consent was waived by the Institutional Review Board.
Ethical approval
Institutional Review Board approval was obtained.
Methodology
• retrospective
Additional information
Publisher’s note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Ji Zhang and Juebin Jin are equal contributors.
Rights and permissions
About this article
Cite this article
Zhang, J., Jin, J., Ai, Y. et al. Differentiating the pathological subtypes of primary lung cancer for patients with brain metastases based on radiomics features from brain CT images. Eur Radiol 31, 1022–1028 (2021). https://doi.org/10.1007/s00330-020-07183-z
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00330-020-07183-z