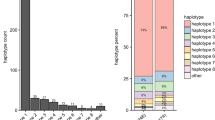
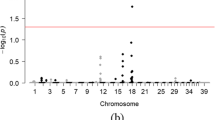
Abstract
Primary hypoadrenocorticism, also known as Addison’s disease, is an autoimmune disorder leading to the destruction of the adrenal cortex and subsequent loss of glucocorticoid and mineralocorticoid hormones. The disease is prevalent in Standard Poodles and is believed to be highly heritable in the breed. Using genotypes derived from the Illumina Canine HD SNP array, we performed a genome-wide association study of 133 carefully phenotyped Standard Poodles (61 affected, 72 unaffected) and found no markers significantly associated with the disease. We also sequenced the entire genomes of 20 Standard Poodles (13 affected, 7 unaffected) and analyzed the data to identify common variants (including SNPs, indels, structural variants, and copy number variants) across affected dogs and variants segregating within a single pedigree of highly affected dogs. We identified several candidate genes that may be fixed in both Standard Poodles and a small population of dogs of related breeds. Further studies are required to confirm these findings more broadly, as well as additional gene-mapping efforts aimed at fully understanding the genetic basis of what is likely a complex inherited disorder.



Similar content being viewed by others
Notes
We are aware of one unpublished report of a genome-wide association study of 120 affected and 120 unaffected Standard Poodles performed at the Broad Institute that failed to identify any genetic differences between these two groups (The Broad Institute).
References
Ashburner M, Ball CA, Blake JA et al (2000) Gene ontology: tool for the unification of biology. The Gene Ontology Consortium. Nat Genet 25:25–29. doi:10.1038/75556
Blomhoff A, Lie BA, Myhre AG et al (2004) Polymorphisms in the cytotoxic T lymphocyte antigen-4 gene region confer susceptibility to Addison’s disease. J Clin Endocrinol Metab 89:3474–3476. doi:10.1210/jc.2003-031854
Boag AM, Catchpole B (2014) A review of the genetics of hypoadrenocorticism. Top Companion Anim Med 29:96–101. doi:10.1053/j.tcam.2015.01.001
Boag AM, Christie MR, McLaughlin KA et al (2015) Autoantibodies against cytochrome P450 side-chain cleavage enzyme in dogs (Canis lupus familiaris) affected with hypoadrenocorticism (Addison’s Disease). PLoS ONE 10:e0143458. doi:10.1371/journal.pone.0143458
Bogdanos DP, Smyk DS, Invernizzi P et al (2013) Tracing environmental markers of autoimmunity: introducing the infectome. Immunol Res 56:220–240. doi:10.1007/s12026-013-8399-6
Cartwright JA, Stone J, Rick M, Dunning MD (2016) Polyglandular endocrinopathy type II (Schmidt’s syndrome) in a Dobermann pinscher. J Small Anim Pract 57:491–494. doi:10.1111/jsap.12535
Colafrancesco S, Agmon-Levin N, Perricone C, Shoenfeld Y (2013) Unraveling the soul of autoimmune diseases: pathogenesis, diagnosis and treatment adding dowels to the puzzle. Immunol Res 56:200–205. doi:10.1007/s12026-013-8429-4
Davydov EV, Goode DL, Sirota M et al (2010) Identifying a high fraction of the human genome to be under selective constraint using GERP++. PLoS Comp Biol 6:e1001025. doi:10.1371/journal.pcbi.1001025
Delaneau O, Marchini J, Zagury J-F (2012) A linear complexity phasing method for thousands of genomes. Nat Methods 9:179–181. doi:10.1038/nmeth.1785
Downs LM, Wallin-Håkansson B, Boursnell M et al (2011) A frameshift mutation in golden retriever dogs with progressive retinal atrophy endorses SLC4A3 as a candidate gene for human retinal degenerations. PLoS ONE 6:e21452. doi:10.1371/journal.pone.0021452
Drögemüller M, Jagannathan V, Becker D et al (2014) A Mutation in the FAM83G gene in dogs with Hereditary footpad hyperkeratosis (HFH). PLoS Genet 10:e1004370. doi:10.1371/journal.pgen.1004370
Famula TR, Belanger JM, Oberbauer AM (2003) Heritability and complex segregation analysis of hypoadrenocorticism in the standard poodle. J Small Anim Pract 44:8–12
Frank CB, Valentin SY, Scott-Moncrieff JCR, Miller MA (2013) Correlation of inflammation with adrenocortical atrophy in canine adrenalitis. J Comp Pathol 149:268–279. doi:10.1016/j.jcpa.2012.11.242
Friedenberg SG, Meurs KM (2016) Genotype imputation in the domestic dog. Mamm Genome. doi:10.1007/s00335-016-9636-9
Friedenberg SG, Meurs KM, Mackay TFC (2016) Evaluation of artificial selection in Standard Poodles using whole-genome sequencing. Mamm Genome. doi:10.1007/s00335-016-9660-9
Hadlow WJ (1953) Adrenal cortical atrophy in the dog; report of three cases. Am J Path 29:353–361
Hanson JM, Tengvall K, Bonnett BN, Hedhammar A (2015) Naturally occurring adrenocortical insufficiency—an epidemiological study based on a Swedish-insured dog population of 525,028 dogs. J Vet Intern Med. doi:10.1111/jvim.13815
Hayward JJ, Castelhano MG, Oliveira KC et al (2016) Complex disease and phenotype mapping in the domestic dog. Nat Commun 7:10460. doi:10.1038/ncomms10460
Howie BN, Donnelly P, Marchini J (2009) A flexible and accurate genotype imputation method for the next generation of genome-wide association studies. PLoS Genet 5:e1000529. doi:10.1371/journal.pgen.1000529
Hughes AM, Nelson RW, Famula TR, Bannasch DL (2007) Clinical features and heritability of hypoadrenocorticism in Nova Scotia Duck Tolling Retrievers: 25 cases (1994–2006). J Am Vet Med Assoc 231:407–412. doi:10.2460/javma.231.3.407
Jagannathan V, Bannoehr J, Plattet P et al (2013) A mutation in the SUV39H2 gene in Labrador Retrievers with hereditary nasal parakeratosis (HNPK) provides insights into the epigenetics of keratinocyte differentiation. PLoS Genet 9:e1003848. doi:10.1371/journal.pgen.1003848
Kang HM, Sul JH, Service SK et al (2010) Variance component model to account for sample structure in genome-wide association studies. Nat Genet 42:348–354
Klambauer G, Schwarzbauer K, Mayr A et al (2012) cn.MOPS: mixture of poissons for discovering copy number variations in next-generation sequencing data with a low false discovery rate. Nucleic Acids Res 40:e69–e69. doi:10.1093/nar/gks003
Kyöstilä K, Cizinauskas S, Seppälä EH et al (2012) A SEL1L mutation links a canine progressive early-onset cerebellar ataxia to the endoplasmic reticulum-associated protein degradation (ERAD) machinery. PLoS Genet 8:e1002759. doi:10.1371/journal.pgen.1002759
Lawrence M, Huber W, Pagès H et al (2013) Software for computing and annotating genomic ranges. PLoS Comp Biol 9:e1003118. doi:10.1371/journal.pcbi.1003118
Lennon EM, Boyle TE, Hutchins RG et al (2007) Use of basal serum or plasma cortisol concentrations to rule out a diagnosis of hypoadrenocorticism in dogs: 123 cases (2000-2005). J Am Vet Med Assoc 231:413–416. doi:10.2460/javma.231.3.413
Lequarré A-S, Andersson L, André C et al (2011) LUPA: a European initiative taking advantage of the canine genome architecture for unravelling complex disorders in both human and dogs. Vet J 189:155–159. doi:10.1016/j.tvjl.2011.06.013
Marchini J, Howie B, Myers S et al (2007) A new multipoint method for genome-wide association studies by imputation of genotypes. Nat Genet 39:906–913. doi:10.1038/ng2088
McLaren W, Pritchard B, Rios D et al (2010) Deriving the consequences of genomic variants with the ensembl API and SNP effect predictor. Bioinformatics 26:2069–2070. doi:10.1093/bioinformatics/btq330
Meurs KM, Mauceli E, Lahmers S et al (2010) Genome-wide association identifies a deletion in the 3′ untranslated region of striatin in a canine model of arrhythmogenic right ventricular cardiomyopathy. Hum Genet 128:315–324. doi:10.1007/s00439-010-0855-y
Mitchell AL, Pearce SHS (2012) Autoimmune Addison disease: pathophysiology and genetic complexity. Nat Rev Endocrinol 8:306–316. doi:10.1038/nrendo.2011.245
Myhre AG, Undlien DE, Løvås K et al (2002) Autoimmune adrenocortical failure in Norway autoantibodies and human leukocyte antigen class II associations related to clinical features. J Clin Endocrinol Metab 87:618–623. doi:10.1210/jcem.87.2.8192
Oberbauer AM, Bell JS, Belanger JM, Famula TR (2006) Genetic evaluation of Addison’s disease in the Portuguese water dog. BMC Vet Res 2:15. doi:10.1186/1746-6148-2-15
Pedersen NC, Brucker L, Tessier NG et al (2015) The effect of genetic bottlenecks and inbreeding on the incidence of two major autoimmune diseases in standard poodles, sebaceous adenitis and Addison’s disease. Canine Genet Epidemiol 2:14. doi:10.1186/s40575-015-0026-5
Petryszak R, Keays M, Tang YA et al (2016) Expression Atlas update–an integrated database of gene and protein expression in humans, animals and plants. Nucleic Acids Res 44:D746–D752. doi:10.1093/nar/gkv1045
Safra N, Bassuk AG, Ferguson PJ et al (2013) Genome-wide association mapping in dogs enables identification of the homeobox gene, NKX2-8, as a genetic component of neural tube defects in humans. PLoS Genet 9:e1003646. doi:10.1371/journal.pgen.1003646
Schaer M, Riley WJ, Buergelt CD et al (1986) Autoimmunity and Addison’s disease in the dog. J Am Anim Hosp Assoc 22:786–794
Skinningsrud B, Husebye ES, Gervin K et al (2008a) Mutation screening of PTPN22: association of the 1858T-allele with Addison’s disease. Eur J Hum Genet 16:977–982. doi:10.1038/ejhg.2008.33
Skinningsrud B, Husebye ES, Pearce SH et al (2008b) Polymorphisms in CLEC16A and CIITA at 16p13 are associated with primary adrenal insufficiency. J Clin Endocrinol Metab 93:3310–3317. doi:10.1210/jc.2008-0821
Tengvall K, Kierczak M, Bergvall K et al (2013) Genome-wide analysis in German shepherd dogs reveals association of a locus on CFA 27 with atopic dermatitis. PLoS Genet 9:e1003475. doi:10.1371/journal.pgen.1003475
The Broad Institute Addison’s disease. https://www.broadinstitute.org/scientific-community/science/projects/mammals-models/dog/disease-research/addison%E2%80%99s-disease. Accessed 26 May 2016
Treangen TJ, Salzberg SL (2012) Repetitive DNA and next-generation sequencing: computational challenges and solutions. Nat Rev Genet 13:36–46. doi:10.1038/nrg3117
Van Lanen K, Sande A (2014) Canine hypoadrenocorticism: pathogenesis, diagnosis, and treatment. Top Companion Anim Med 29:88–95. doi:10.1053/j.tcam.2014.10.001
vonHoldt BM, Pollinger JP, Lohmueller KE et al (2010) Genome-wide SNP and haplotype analyses reveal a rich history underlying dog domestication. Nature 464:898–902. doi:10.1038/nature08837
Wiik AC, Wade C, Biagi T et al (2008) A deletion in nephronophthisis 4 (NPHP4) is associated with recessive cone-rod dystrophy in standard wire-haired dachshund. Genome Res 18:1415–1421. doi:10.1101/gr.074302.107
Wolf ZT, Leslie EJ, Arzi B et al (2014) A LINE-1 insertion in DLX6 is responsible for cleft palate and mandibular abnormalities in a canine model of Pierre Robin sequence. PLoS Genet 10:e1004257. doi:10.1371/journal.pgen.1004257
Ye K, Schulz MH, Long Q et al (2009) Pindel: a pattern growth approach to detect break points of large deletions and medium sized insertions from paired-end short reads. Bioinformatics 25:2865–2871. doi:10.1093/bioinformatics/btp394
Acknowledgements
SGF is supported by a National Institutes of Health T32 training award (5T32OD011130-07). Funding for whole-genome sequencing was provided in part by the Poodle Club of America Foundation and the Morris Animal Foundation. Seed funding for this project was provided by the North Carolina State University Comparative Medicine Institute. Some whole-genome sequencing data were graciously contributed by Drs. Leigh Anne-Clark (13 dogs), Natasha J. Olby and Theirry Olivry (11 dogs), and Joshua A. Stern (two dogs).
Author contributions
SGF collected samples, designed the study, analyzed the data, and wrote the manuscript. KFL collected samples and provided guidance regarding phenotyping of dogs. KMM collected samples and supervised the study. All authors have read and edited the manuscript.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
The authors declare no conflict of interest.
Electronic supplementary material
Below is the link to the electronic supplementary material.
335_2016_9671_MOESM1_ESM.tiff
Supplemental figure 1—Scoring algorithm and follow-up criterial used to evaluate genetic variants for Addison’s disease. Each variant was allocated one point for meeting criteria in each of three categories: variant consequence, GERP++ conservation score, or biological function (top). Scoring was carried out using custom scripting/filtering in R. A maximum of 3 points was allocated to each variant. After each variant was scored, specific follow-up criteria were applied by manual curation (bottom). Variants flagged for follow-up were evaluated in additional population of dogs as described in the Methods section of the text. See http://useast.ensembl.org/info/genome/variation/predicted_data.html#consequences for a complete listing of VEP variant consequences. (TIFF 1848 kb)
335_2016_9671_MOESM2_ESM.tiff
Supplemental figure 2—Principal components plots of the first and second eigenvalues for 175 Standard Poodles passing initial filtering criteria. Individual dogs are colored to indicate (A) MDS outliers, (B) phenotype, and (C) gender. In (A), gray squares represent the 42 dogs that were removed from from downstream GWAS analysis based upon MDS outlier detection. (TIFF 36918 kb)
Rights and permissions
About this article
Cite this article
Friedenberg, S.G., Lunn, K.F. & Meurs, K.M. Evaluation of the genetic basis of primary hypoadrenocorticism in Standard Poodles using SNP array genotyping and whole-genome sequencing. Mamm Genome 28, 56–65 (2017). https://doi.org/10.1007/s00335-016-9671-6
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00335-016-9671-6