Abstract
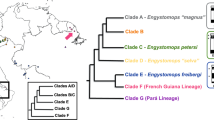
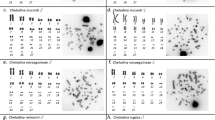
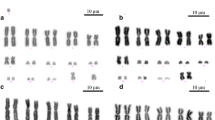
Heteromorphic sex chromosomes are common in eukaryotes and largely ubiquitous in birds and mammals. The largest number of multiple sex chromosomes in vertebrates known today is found in the monotreme platypus (Ornithorhynchus anatinus, 2n = 52) which exhibits precisely 10 sex chromosomes. Interestingly, fish, amphibians, and reptiles have sex determination mechanisms that do or do not involve morphologically differentiated sex chromosomes. Relatively few amphibian species carry heteromorphic sex chromosomes, and when present, they are frequently represented by only one pair, either XX:XY or ZZ:ZW types. Here, in contrast, with several evidences, from classical and molecular cytogenetic analyses, we found 12 sex chromosomes in a Brazilian population of the smoky jungle frog, designated as Leptodactylus pentadactylus Laurenti, 1768 (Leptodactylinae), which has a karyotype with 2n = 22 chromosomes. Males exhibited an astonishing stable ring-shaped meiotic chain composed of six X and six Y chromosomes. The number of sex chromosomes is larger than the number of autosomes found, and these data represent the largest number of multiple sex chromosomes ever found among vertebrate species. Additionally, sequence and karyotype variation data suggest that this species may represent a complex of species, in which the chromosomal rearrangements may possibly have played an important role in the evolution process.




Similar content being viewed by others
References
Abramyan J, Ezaz T, Graves JAM, Koopmanand P (2009) W sex chromosomes in the cane toad (Bufo marinus). Chromosom Res 17(8):1015–1024. https://doi.org/10.1007/s10577-009-9095-1
Amaro-Ghilardi RC, Rodrigues MT, Yonenaga-Yassuda YY (2004) Chromosomal studies after differential staining and fluorescence in situ hybridization using telomeric probe in three Leptodactylus species (Leptodactylidae, Anura). Caryologia 57(1):53–65. https://doi.org/10.1080/00087114.2004.10589372
Bachtrog D, Mank JE, Peichel CL, Kirkpatrick M, Otto SP, Ashman T-L, Hahn MW, Kitano J, Mayrose I, Ming R, Perrin N, Ross L, Valenzuela N, Vamosi JC (2014) Sex determination: why so many ways of doing it? PLoS Biol 12(7):e1001899. https://doi.org/10.1371/journal.pbio.1001899
Baldissera FA Jr, Oliveira PSL, Kasahara S (1993) Cytogenetics of four Brazilian Hyla species (Amphibia-Anura) and description of a case with a supernumerary chromosome. Rev Bras Genet 16:335–345
Bogart JP (1974) A karyosystematic study of frogs in the genus Leptodactylus (Anura: Leptodactylidae). Copeia 3:728–737
Brum-Zorilla N, Saez FA (1968) Chromosomes of Leptodactylidae (Amphibia Anura). Experientia 24(9):969. https://doi.org/10.1007/BF02138689
Campos JRC, Ananias F, Haddad CFB, Kasahara S (2008) Karyotypic similarity among Barycholos ternetzi and five species of the genus Eleutherodactylus from southeastern Brazil (Anura, Brachycephalidae). Micron 39(2):151–159. https://doi.org/10.1016/j.micron.2006.11.001
Clement M, Posada D, Crandall KA (2000) TCS: a computer program to estimate gene genealogies. Mol Ecol 9(10):1657–1659. https://doi.org/10.1046/j.1365-294x.2000.01020.x
Cnaani A, Lee BY, Zilberman N, Ozouf-Costaz C, Hulata G, Ron M, D’Hont A, Baroiller JF, D’Cotta H, Penman DJ, Tomasino E, Coutanceau JP, Pepey E, Shirak A, Kocher TD (2008) Genetics of sex determination in Tilapiine species. Sex Dev 2(1):43–54. https://doi.org/10.1159/000117718
Coelho AC, de Mattos TL, Viana P, Terencio ML, Schneider CH, Menin M, Gross MC (2016) Intra-generic and interspecific karyotype patterns of Leptodactylus and Adenomera (Anura, Leptodactylidae) with inclusion of five species from Central Amazonia. Genetica 144(1):37–46. https://doi.org/10.1007/s10709-015-9876-8
Crawford AJ (2003) Huge populations and old species of costa Rican and Panamanian dirt frogs inferred from mitochondrial and nuclear gene sequences. Mol Ecol 12(10):2525–2540. https://doi.org/10.1046/j.1365-294X.2003.01910.x
Ellegren H (2011) Sex-chromosome evolution: recent progress and the influence of male and female heterogamety. Nat Rev Genet 12(3):157–166. https://doi.org/10.1038/nrg2948
Frost DR (2017) Amphibian species of the world: an online reference (Version 6.0) http://research.amnh.org/vz/herpetology/amphibia/Amphibia/Anura/Leptodactylidae/Leptodactylinae/Leptodactylus/Leptodactylus-pentadactylus. Accessed 10 March 2017
Gamble T, Geneva AJ, Glor RE, Zarkower D (2014) Anolis sex chromosomes are derived from a single ancestral pair. Evolution 68(4):1027–1041. https://doi.org/10.1111/evo.12328
Gatto KP, Busin CS, Lourenço LB (2016) Unraveling the sex chromosome heteromorphism of the paradoxical frog Pseudis tocantins. PLoS One 11(5):e0156176. https://doi.org/10.1371/journal.pone.0156176
Gazoni T, Gruber SL, Silva APZ, Araújo OGS, Narimatsu H, Strüssmann C, Haddad CFB, Kasahara S (2012) Cytogenetic analyses of eight species in the genus Leptodactylus Fitzinger, 1843 (Amphibia, Anura, Leptodactylidae), including a new diploid number and a karyotype with multiple translocations. BMC Genet 13(1):109. https://doi.org/10.1186/1471-2156-13-109
Graves JAM (2016) Evolution of vertebrate sex chromosomes and dosage compensation. Nat Rev Genet 17(1):33–46. https://doi.org/10.1038/nrg.2015.2
Gross MC, Feldberg E, Cella DM, Schneider MC, Schneider CH, Porto JI, Martins C (2009) Intriguing evidence of translocations in discus fish (Symphysodon, Cichlidae) and a report of the largest meiotic chromosomal chain observed in vertebrates. Heredity 102(5):435–441. https://doi.org/10.1038/hdy.2009.3
Gruetzner F, Ashley T, Rowell DM, Graves JAM (2006) How did the platypus get its sex chromosome chain? A comparison of meiotic multiples and sex chromosomes in plants and animals. Chromosoma 115(2):75–88. https://doi.org/10.1007/s00412-005-0034-4
Grützner F, Rens W, Tsend-Ayush E, El-Mogharbel N, O’Brien PCM, Jones RC, Ferguson-Smith MA, Graves JAM (2004) In the platypus a meiotic chain of ten sex chromosomes shares genes with the bird Z and mammal X chromosomes. Nature 432(7019):913–917. https://doi.org/10.1038/nature03021
Guerrero R, Kirkpatrick M, Perrin N (2012) Cryptic recombination in the ever-young sex chromosomes of hylid frogs. J Evol Biol 25(10):1947–1954. https://doi.org/10.1111/j.1420-9101.2012.02591.x
Guindon S, Dufayard JF, Lefort V, Anisimova M, Hordijk W, Gascuel O (2010) New algorithms and methods to estimate maximum-likelihood phylogenies: assessing the performance of PhyML 3.0. Syst Biol 59(3):307–321. https://doi.org/10.1093/sysbio/syq010
Heyer WR, Diment MJ (1974) The karyotype of Vanzolinius discodactylus and comments on usefulness of karyotypes in determining relationships in the Leptodactylus-Complex (Amphibia, Leptodactylidae). Proc Biol Soc Wash 87:327–336
Hotz H, Uzzellt T, Berger L (1997) Linkage groups of protein-coding genes in western Palearctic water frogs reveal extensive evolutionary conservation. Genetics 147(1):255–270
Katoh S (2013) MAFFT multiple sequence alignment software version 7: improvements in performance and usability. Mol Biol Evol 30(4):772–780. https://doi.org/10.1093/molbev/mst010
Lefort V, Longueville JM, Gascuel O (2017) SMS: smart model selection in PhyML. Mol Biol and Evol 34(9):2422–2424. https://doi.org/10.1093/molbev/msx149
Lourenço LB, Recco-Pimentel SM, Cardoso AJ (2000) A second case of multivalent meiotic configurations in diploid species of Anura. Genet Mol Biol 23(1):131–133. https://doi.org/10.1590/S1415-47572000000100024
Lyra ML, Haddad CFBH, Azeredo-Espin AML (2016) Meeting the challenge of DNA barcoding amphibians from Neotropics: polymerase chain reaction optimization and new COI primers. Mol Ecol Resour 17(5):966–980. https://doi.org/10.1111/1755-0998.12648
Malone JH, Fontenot BE (2008) Patterns of reproductive isolation in toads. PLoS One 3(12):e3900. https://doi.org/10.1371/journal.pone.0003900
Maniatis T, Fritsch EF, Sambrook J (1982) Molecular cloning: a laboratory manual. Cold Spring Harbor Laboratory, New York
Matsubara K, Knopp T, Sarre SD, Georges A, Ezaz T (2013) Karyotypic analysis and FISH mapping of microsatellite motifs reveal highly differentiated XX/XY sex chromosomes in the pink-tailed wormlizard (Aprasia parapulchella, Pygopodidae, Squamata). Mol Cytogenet 6(1):60. https://doi.org/10.1186/1755-8166-6-60
Matsubara K, O’Meally D, Azad B, Georges A, Sarre SD, Graves JA, Matsuda Y, Ezaz T (2016) Amplification of microsatellite repeat motifs is associated with the evolutionary differentiation and heterochromatinization of sex chromosomes in Sauropsida. Chromosoma 125(1):111–123. https://doi.org/10.1007/s00412-015-0531-z
Meunier-Rotival M, Cortadas J, Macaya G, Bernardi G (1979) Isolation and organization of calf ribosomal DNA. Nucleic Acids Res 6(6):2109–2123. https://doi.org/10.1093/nar/6.6.2109
Miura I (2007) An evolutionary witness: the frog Rana rugosa, underwent change of heterogametic sex from XY male to ZW female. Sex Dev 1(6):323–331. https://doi.org/10.1159/000111764
Murtagh CE (1977) Unique cytogenetic system in monotremes. Chromosoma 65(1):37–57. https://doi.org/10.1007/BF00293129
O’Meally D, Patel HR, Stiglec R, Sarre SD, Georges A, Graves JAM, Ezaz T (2010) Non-homologous sex chromosomes of birds and snakes share repetitive sequences. Chromosom Res 18(7):787–800. https://doi.org/10.1007/s10577-010-9152-9
Palumbi SR, Martin AP, Romano SL, Mcmillan WO, Stice L, Grabowski G (1991) The simple fool’s guide to PCR. Honolulu, Dept. of Zoology, University of Hawaii
Perrin N (2009) Sex reversal: a fountain of youth for sex chromosomes? Evolution 63(12):3043–3049. https://doi.org/10.1111/j.1558-5646.2009.00837.x
Pinkel D, Straume T, Gray JW (1986) Cytogenetic analysis using quantitative, high-sensitivity, fluorescence hybridization. Proc Natl Acad Sci U S A 83(9):2934–2938. https://doi.org/10.1073/pnas.83.9.2934
Pokorná M, Altmanová M, Kratochvíl L (2014) Multiple sex chromosomes in the light of female meiotic drive in amniote vertebrates. Chromosom Res 22(1):35–44. https://doi.org/10.1007/s10577-014-9403-2
Rens W, O’Brien PCM, Grützner F, Clarke O, Graphodatskaya D, Tsend-Ayush E, Trifonov VA, Skelton H, Wallis MC, Johnston S, Veyrunes F, Graves JAM, Ferguson-Smith MA (2007) The multiple sex chromosomes of platypus and echidna are not completely identical and several share homology with the avian Z. Genome Biol 8(11):R243. https://doi.org/10.1186/gb-2007-8-11-r243
Rodrigues N, Vueille Y, Brelsford BA, Merilä J, Perrin N (2016) The genetic contribution to sex determination and number of sex chromosomes vary among populations of common frogs (Rana temporaria). Heredity 117(1):25–32. https://doi.org/10.1038/hdy.2016.22
Ross JA, Urton JR, Boland J, Shapiro MD, Peichel CL (2009) Turnover of sex chromosomes in the stickleback fishes (Gasterosteidae). PLoS Genet 5(2):e1000391. https://doi.org/10.1371/journal.pgen.1000391
Rovatsos M, Pokorná MJ, Kratochvíl L (2015) Differentiation of sex chromosomes and karyotype characterisation in the Dragonsnake Xenodermus javanicus (Squamata: Xenodermatidae). Cytogenet Genome Res 147(1):48–54. https://doi.org/10.1159/000441646
Sarre SD, Ezaz T, Georges A (2011) Transitions between sex-determining systems in reptiles and amphibians. Annu Rev Genom Human Genet 12(1):391–406. https://doi.org/10.1146/annurev-genom-082410-101518
Schartl M (2004) Sex chromosome evolution in non-mammalian vertebrates. Curr Opin Genet Dev 14(6):634–641. https://doi.org/10.1016/j.gde.2004.09.005
Schmid M (1978) Chromosome banding in Amphibia. I. Constitutive heterochromatin and nucleolus organizer regions in Bufo and Hyla. Chromosoma 66(4):361–388. https://doi.org/10.1007/BF00328536
Schmid M, Steinlein C, Feichtinger W (1992) Chromosome banding in Amphibia. XVII. First demonstration of multiple sex chromosomes in amphibians: Eleutherodactylus maussi (Anura, Leptodactylidae). Chromosoma 101(5-6):284–292. https://doi.org/10.1007/BF00346007
Schmid M, Feichtinger W, Steilein C, Visbal García R, Fernandez Badillo A (2003) Chromosome banding in Amphibia XXVIII. Homomorphic XY sex chromosomes and derived Y-autosome translocation in Eleutherodactylus riveroi (Anura, Leptodactylidae). Cytogenet Genome Res 101(1):62–73. https://doi.org/10.1159/000073420
Schmid M, Steinlein C, Bogart JP, Feichtinger W, León P, La Marca E, Diaz LM, Sanz A, Chen S-H, Hedges SB (2010) The chromosomes of Terraranan frogs: insights into vertebrate cytogenetics. Cytogenet Genome Res 130-131(1-8):1–568. https://doi.org/10.1159/000301339
Šíchová J, Voleníková A, Dincă V, Nguyen P, Vila R, Sahara K, Marec F (2015) Dynamic karyotype evolution and unique sex determination systems in Leptidea wood white butterflies. BMC Evol Biol 15(1):89. https://doi.org/10.1186/s12862-015-0375-4
Šíchová J, Ohno M, Dincă V, Watanabe M, Sahara K, Marec F (2016) Fissions, fusions, and translocations shaped the karyotype and multiple sex chromosome constitution in the northeast-Asian wood white butterfly, Leptidea amurensis. Biol J Linnean Soc 118(3):457–471. https://doi.org/10.1111/bij.12756
Singh L, Panicker SG, Nagaraj R, Majumdar KC (1994) Banded krait minor-satellite (Bkm)-associated Y chromosome-specific repetitive DNA in mouse. Nucleic Acids Res 22(12):2289–2295. https://doi.org/10.1093/nar/22.12.2289
Siqueira S, Ananias F, Recco-Pimentel SM (2004) Cytogenetics of three Brazilian species of Eleutherodactylus (Anura, Leptodactylidae) with 22 chromosomes and re-analysis of multiple translocations in E. binotatus. Genet Mol Biol 27(3):363–372. https://doi.org/10.1590/S1415-47572004000300010
Stöck M, Croll D, Dumas Z, Biollay S, Wang J, Perrin N (2011a) A cryptic heterogametic transition revealed by sex-linked DNA markers in Palearctic green toads. J Evol Biol 24(5):1064–1107. https://doi.org/10.1111/j.1420-9101.2011.02239.x
Stöck M, Horn A, Grossen C, Lindtke D, Sermier R, Betto-Colliard C, Dufresnes C, Bonjour E, Dumas Z, Luquet E, Maddalena T, Sousa HC, Martinez-Solano I, Perrin N (2011b) Ever-young sex-chromosomes in European tree frogs. PLoS Biol 9(5):e1001062. https://doi.org/10.1371/journal.pbio.1001062
Stöck M, Savary R, Betto-Colliard C, Biollay S, Jourdan-Pineau H, Perrin N (2013a) Low rates of X-Y recombination, not turnovers, account for homomorphic sex chromosomes in several diploid species of Palearctic green toads (Bufo viridis subgroup). J Evol Biol 26(3):674–682. https://doi.org/10.1111/jeb.12086
Stöck M, Savary R, Zaborowska A, Górecki G, Brelsford A, Rozenblut-Kościsty B, Ogielska M, Perrin N (2013b) Maintenance of ancestral sex chromosomes in Palearctic tree frogs: direct evidence from Hyla orientalis. Sex Dev 7(5):261–266. https://doi.org/10.1159/000351089
Tamura K, Stecher G, Peterson D, Filipski A, Kumar S (2013) MEGA6: Molecular Evolutionary Genetics Analysis Version 6.0. Molecular Biology and Evolution 30 (12):2725–2729
Tanaka K, Takehana Y, Naruse K, Hamaguchi S, Sakaizumi M (2007) Evidence for different origins of sex chromosomes in closely related Oryzias fishes: substitution of the master sex-determining gene. Genetics 177(4):2075–2081. https://doi.org/10.1534/genetics.107.075598
Uno Y, Nishida C, Yoshimoto S, Ito M, Oshima Y (2008) Diversity in the origins of sex chromosomes in anurans inferred from comparative mapping of sexual differentiation genes for three species of the Raninae and Xenopodinae. Chromosom Res 16(7):999–1011. https://doi.org/10.1007/s10577-008-1257-z
Van der Meijden A, Vences M, Hoegg S, Boistel R, Channing A, Meyer A (2007) Nuclear gene phylogeny of narrow-mouthed toads (family: Microhylidae) and a discussion of competing hypotheses concerning their biogeographical origins. Mol Phylogenet Evol 44(3):1017–1030. https://doi.org/10.1016/j.ympev.2007.02.008
Vieites DR, Wollenberg KC, Andreone F, Köhler J, Glaw F, Vences M (2009) Vast underestimation of Madagascar’s biodiversity evidenced by an integrative amphibian inventory. Proc Natl Acad Sci U S A 106(20):8267–8272. https://doi.org/10.1073/pnas.0810821106
Villesen P (2007) FaBox: an online toolbox for FASTA sequences. Mol Ecol Notes 7(6):965–968. https://doi.org/10.1111/j.1471-8286.2007.01821.x
Volff JN, Nanda I, Schmid M, Schartl M (2007) Governing sex determination in fish: regulatory putsches and ephemeral dictators. Sex Dev 2:85–99
Wiens JJ, Fetzner JW Jr, Parkinson CL, Reeder TW (2005) Hylid frog phylogeny and sampling strategies for speciose clades. Syst Biol 54(5):778–807. https://doi.org/10.1080/10635150500234625
Wright AE, Dean R, Zimmer F, Mank JE (2016) How to make a sex chromosome. Nat Commun 7:12087. https://doi.org/10.1038/ncomms12087
Acknowledgements
We thank Natalia Maria Espíndola Salles, Juliano Mafra Neves, and Karll Cavalcante Pinto for help in field work, and to João Rodrigo Cabeza as well as Companhia Hidrelétrica Teles Pires for allowing collecting activities in their study areas. We are grateful to Heidi Horn, for a revision in the final version of this manuscript. TG thanks Instituto Chico Mendes de Conservação da Biodiversidade (ICMBio – SISBIO) for providing a collection permit (no. 30202-2). The authors are also grateful to the Centro de Estudos de Insetos Sociais, UNESP – Univ. Estadual Paulista, Rio Claro, Brazil, for allowing the utilization of facilities for molecular analyses.
Funding
This work was supported by Coordenação de Aperfeiçoamento de Pessoal de Nível Superior – CAPES, which provided a PhD fellowship to TG, by Conselho Nacional de Desenvolvimento Científico e Tecnológico - CNPq for research funding (no. 473264/212-9) to PPPM, and by São Paulo Research Foundation (FAPESP) for a research grant (no. 2013/50741–7) to CFBH. CNPq provided a research fellowship to CFBH.
Author information
Authors and Affiliations
Contributions
T. G., P. P. P. M., and C. F. B. H. designed the study. T. G. performed the cytogenetic analyses. D. C. C. M. assisted on microsatellite and CGH experiments. M. L. L. performed DNA sequencing and similarity analyses. T. G. and H. N. collected the specimens. T. G. and P. P. P. M. wrote the main manuscript, and all authors contributed to improve it. All authors approved the final version of the manuscript.
Corresponding author
Ethics declarations
Conflict of interest
The authors declare that they have no conflict of interest.
Ethical approval
All applicable international, national, and/or institutional guidelines for the care and use of animals were followed. All procedures performed in studies involving animals were in accordance with the ethical standards of the institution or practice at which the studies were conducted.
Electronic supplementary material
ESM 1
(DOCX 69 kb)
Rights and permissions
About this article
Cite this article
Gazoni, T., Haddad, C.F.B., Narimatsu, H. et al. More sex chromosomes than autosomes in the Amazonian frog Leptodactylus pentadactylus. Chromosoma 127, 269–278 (2018). https://doi.org/10.1007/s00412-018-0663-z
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00412-018-0663-z