Abstract
Main conclusion
Riboside type cytokinins are key components in cytokinin metabolism, transport, and sensitivity, making them important functional signals in plant growth and development and environmental stress responses.
Abstract
Cytokinin (CKs) are phytohormones that regulate multiple processes in plants and are critical for agronomy, as they are involved in seed filling and plant responses to biotic and abiotic stress. Among the over 30 identified CKs, there is uncertainty about the roles of many of the individual CK structural forms. Cytokinin free bases (CKFBs), have been studied in great detail, but, by comparison, roles of riboside-type CKs (CKRs) in CK metabolism and associated signaling pathways and their distal impacts on plant physiology remain largely unknown. Here, recent findings on CKR abundance, transport and localization, are summarized, and their importance in planta is discussed. The history of CKR analyses is reviewed, in the context of the determination of CK metabolic pathways, and research on CKR affinity for CK receptors, all of which yield essential insights into their functions. Recent studies suggest that CKR forms are a lot more than a group of transport CKs and, beyond this, they play important roles in plant development and responses to environmental stress. In this context, this review discusses the involvement of CKRs in plant development, and highlight the less anticipated functions of CKRs in abiotic stress tolerance. Based on this, possible mechanisms for CKR modes of action are proposed and experimental approaches to further uncover their roles and future biotechnological applications are suggested.



Similar content being viewed by others
References
Abusamhadneh E, McDonald NE, Kline PC (2000) Isolation and characterization of adenosine nucleosidase from yellow lupin (Lupinus luteus). Plant Sci 153(1):25–32. https://doi.org/10.1016/S0168-9452(99)00240-X
Akiyoshi DE, Regier DA, Gordon MP (1987) Cytokinin production by Agrobacterium and Pseudomonas spp. J Bacteriol 169(9):4242–4248. https://doi.org/10.1128/jb.169.9.4242-4248.1987
Alvarez S, Marsh EL, Schroeder SG, Schachtman DP (2008) Metabolomic and proteomic changes in the xylem sap of maize under drought. Plant Cell Environ 31(3):325–340. https://doi.org/10.1111/j.1365-3040.2007.01770.x
Andrabi SBA, Tahara M, Matsubara R, Toyama T, Aonuma H, Sakakibara H, Suematsu M, Tanabe K, Nozaki T, Nagamune K (2018) Plant hormone cytokinins control cell cycle progression and plastid replication in apicomplexan parasites. Parasitol Int 67(1):47–58. https://doi.org/10.1016/j.parint.2017.03.003
Andreas P, Kisiala A, Emery RJN, Clerck-Floate D, Tooker JF, Price PW, Miller DG III, Chen MS, Connor EF (2020) Cytokinins are abundant and widespread among insect species. Plants 9(2):208–231. https://doi.org/10.3390/plants9020208
Aoki MM, Kisiala A, Li S, Stock NL, Brunetti CR, Huber RJ, Emery RJN (2019a) Cytokinin detection during the Dictyostelium discoideum life cycle: Profiles are dynamic and affect cell growth and spore germination. Biomolecules 9(11):702–718. https://doi.org/10.3390/biom9110702
Aoki MM, Seegobin M, Kisiala A, Noble A, Brunetti C, Emery RJN (2019b) Phytohormone metabolism in human cells: Cytokinins are taken up and interconverted in HeLa cell culture. FASEB BioAdvances 1(5):320–331. https://doi.org/10.1096/fba.2018-00032
Arkhipova TN, Veselov SU, Melentiev AI, Martynenko EV, Kudoyarova GR (2005) Ability of bacterium Bacillus subtilis to produce cytokinins and to influence the growth and endogenous hormone content of lettuce plants. Plant Soil 272(1):201–209. https://doi.org/10.1007/s11104-004-5047-x
Ashihara H, Stasolla C, Fujimura T, Crozier A (2018) Purine salvage in plants. Phytochemistry 147:89–124. https://doi.org/10.1016/j.phytochem.2017.12.008
Atkins CA (2013) Mechanism of long-distance solute transport in phloem elements. Symplasmic Transport in Vascular Plants. Springer, New York, pp 165–181
Bedada LT, Seth MS, Runo SM, Teffera W, Mugoya C, Masiga CW, Oduor RO, Blumewald E, Wachira F (2016) Drought tolerant tropical maize (Zea mays L.) developed through genetic transformation with isopentenyltransferase gene. Afr J Biotechnol 15(43):2447–2464. https://doi.org/10.5897/ajb2016.15228
Bromley JR, Warnes BJ, Newell CA, Thomson JC, James CM, Turnbull CG, Hanke DE (2014) A purine nucleoside phosphorylase in Solanum tuberosum L. (potato) with specificity for cytokinins contributes to the duration of tuber endodormancy. Biochemical J 458(2):225–237. https://doi.org/10.1042/BJ20130792
Bruce SA, Saville BJ, Emery RJN (2011) Ustilago maydis produces cytokinins and abscisic acid for potential regulation of tumor formation in maize. J Plant Growth Regul 30(1):51–63. https://doi.org/10.1007/s00344-010-9166-8
Brugière N, Humbert S, Rizzo N, Bohn J, Habben JE (2008) A member of the maize isopentenyl transferase gene family, Zea mays isopentenyl transferase 2 (ZmIPT2), encodes a cytokinin biosynthetic enzyme expressed during kernel development: Cytokinin biosynthesis in maize. Plant Mol Biol 67(3):215–229. https://doi.org/10.1007/s11103-008-9312-x
Bürkle L, Cedzich A, Döpke C, Stransky H, Okumoto S, Gillissen B, Kühn C, Frommer WB (2003) Transport of cytokinins mediated by purine transporters of the PUP family expressed in phloem, hydathodes, and pollen of Arabidopsis. Plant J 34(1):13–26. https://doi.org/10.1046/j.1365-313X.2003.01700.x
Chanclud E, Kisiala A, Emery RJN, Chalvon V, Ducasse A, Romiti-Michel C, Gravot A, Kroj T, Morel JB (2016) Cytokinin production by the rice blast fungus is a pivotal requirement for full virulence. PLoS Pathog 12(2):e1005457–e1005482. https://doi.org/10.1371/journal.ppat.1005457
Chang C (2016) Q and A: How do plants respond to ethylene and what is its importance? BMC Biol 14(1):7–14. https://doi.org/10.1186/s12915-016-0230-0
Chen CM, Kristopeit SM (1981) Metabolism of cytokinin: deribosylation of cytokinin ribonucleoside by adenosine nucleosidase from wheat germ cells. Plant Physiol 68(5):1020–1023. https://doi.org/10.1104/pp.68.5.1020
Chen CM, Melitz DK, Clough FW (1982) Metabolism of cytokinin: Phosphoribosylation of cytokinin bases by adenine phosphoribosyltransferase from wheat germ. Arch Biochem Biophys 214(2):634–641. https://doi.org/10.1016/0003-9861(82)90069-8
Chen L, Zhao J, Song J, Jameson PE (2020) Cytokinin dehydrogenase: a genetic target for yield improvement in wheat. Plant Biotechnol J 18(3):614–630. https://doi.org/10.1111/pbi.13305
Daudu D, Allion E, Liesecke F, Papon N, Courdavault V, Dugé de Bernonville T, Mélin C, Oudin A, Clastre M, Lanoue A, Courtois M (2017) CHASE-containing histidine kinase receptors in apple tree: From a common receptor structure to divergent cytokinin binding properties and specific functions. Front Plant Sci 8:1614–1629. https://doi.org/10.3389/fpls.2017.01614
Daudu D, Kisiala A, Ribeiro CW, Mélin C, Perrot L, Clastre M, Courdavault V, Papon N, Oudin A, Courtois M, de Bernonville TD (2019) Setting-up a fast and reliable cytokinin biosensor based on a plant histidine kinase receptor expressed in Saccharomyces cerevisiae. J Biotechnol 289:103–111. https://doi.org/10.1016/j.jbiotec.2018.11.013
De Meutter J, Tytgat T, Witters E, Gheysen G, Van Onckelen H, Gheysen G (2003) Identification of cytokinins produced by the plant parasitic nematodes Heterodera schachtii and Meloidogyne incognita. Mol Plant Pathol 4(4):271–277. https://doi.org/10.1046/j.1364-3703.2003.00176.x
Dhara A, Raichaudhuri A (2021) ABCG transporter proteins with beneficial activity on plants. Phytochemistry 184:112663. https://doi.org/10.1016/j.phytochem.2021.112663
Dobra J, Motyka V, Dobrev P, Malbeck J, Prasil IT, Haisel D, Gaudinova A, Havlova M, Gubis J, Vankova R (2010) Comparison of hormonal responses to heat, drought and combined stress in tobacco plants with elevated proline content. J Plant Physiol 167(16):1360–1370. https://doi.org/10.1016/j.jplph.2010.05.013
Dolgikh EA, Shaposhnikov AI, Dolgikh AV, Gribchenko ES, Bodyagina KB, Yuzhikhin OS, Tikhonovich IA (2017) Identification of Pisum sativum L. cytokinin and auxin metabolic and signaling genes, and an analysis of their role in symbiotic nodule development. Int J Plant Physiol Biochem 9(3):22–35. https://doi.org/10.5897/ijppb2017.0266
Durán-Medina Y, Díaz-Ramírez D, Marsch-Martínez N (2017) Cytokinins on the move. Front Plant Sci 8:146–153. https://doi.org/10.3389/fpls.2017.00146
Eguchi T, Yoshida S (2008) Effects of application of sucrose and cytokinin to roots on the formation of tuberous roots in sweetpotato (Ipomoea batatas (L.) Lam.). Plant Root 2:7–13. https://doi.org/10.3117/plantroot.2.7
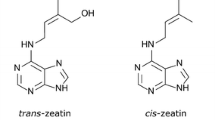
Emery RJN, Leport L, Barton JE, Turner NC, Atkins CA (1998) Cis-isomers of cytokinins predominate in chickpea seeds throughout their development. Plant Physiol 117(4):1515–1523. https://doi.org/10.1104/pp.117.4.1515
Emery RJN, Ma Q, Atkins CA (2000) The forms and sources of cytokinins in developing white lupine seeds and fruits. Plant Physiol 123(4):1593–1604. https://doi.org/10.1104/pp.123.4.1593
Frébort I, Kowalska M, Hluska T, Frébortová J, Galuszka P (2011) Evolution of cytokinin biosynthesis and degradation. J Exp Bot 62(8):2431–2452. https://doi.org/10.1093/jxb/err004
Gillissen B, Bürkle L, André B, Kühn C, Rentsch D, Brandl B, Frommer WB (2000) A new family of high-affinity transporters for adenine, cytosine, and purine derivatives in Arabidopsis. Plant Cell 12(2):291–300. https://doi.org/10.1105/tpc.12.2.291
Giron D, Glevarec G (2014) Cytokinin-induced phenotypes in plant-insect interactions: Learning from the bacterial world. J Chem Ecol 40(7):826–835. https://doi.org/10.1007/s10886-014-0466-5
Glanz-Idan N, Tarkowski P, Turečková V, Wolf S (2020) Root–shoot communication in tomato plants: cytokinin as a signal molecule modulating leaf photosynthetic activity. J Exp Bot 71(1):247–257. https://doi.org/10.1093/jxb/erz399
Goggin DE, Emery RJN, Powles SB, Steadman KJ (2010) Initial characterisation of low and high seed dormancy populations of Lolium rigidum produced by repeated selection. J Plant Physiol 167(15):1282–1288. https://doi.org/10.1016/j.jplph.2010.04.004
Goggin DE, Emery RJN, Kurepin LV, Powles SB (2015) A potential role for endogenous microflora in dormancy release, cytokinin metabolism and the response to fluridone in Lolium rigidum seeds. Ann Bot 115(2):293–301. https://doi.org/10.1093/aob/mcu231
Held MA, Quesnelle PE, Emery RJN (2005) Seasonal changes of cytokinins in upper and lower leaves of a sugar maple crown. Biol Plant 49(3):455–458. https://doi.org/10.1007/s10535-005-0028-3
Hirose N, Makita N, Yamaya T, Sakakibara H (2005) Functional characterization and expression analysis of a gene, OsENT2, encoding an equilibrative nucleoside transporter in rice suggest a function in cytokinin transport. Plant Physiol 138(1):196–206. https://doi.org/10.1104/pp.105.060137
Hirose N, Makita N, Kojima M, Kamada-Nobusada T, Sakakibara H (2007) Overexpression of a type-A response regulator alters rice morphology and cytokinin metabolism. Plant Cell Physiol 48(3):523–539. https://doi.org/10.1093/pcp/pcm022
Hirose N, Takei K, Kuroha T, Kamada-Nobusada T, Hayashi H, Sakakibara H (2008) Regulation of cytokinin biosynthesis, compartmentalization and translocation. J Exp Bot 59(1):75–83. https://doi.org/10.1093/jxb/erm157
Hluska T, Hluskova L, Emery RJN (2021) The Hulks and the Deadpools of the cytokinin universe: a dual strategy for cytokinin production, translocation, and signal transduction. Plants 11(2):209. https://doi.org/10.3390/biom11020209
Hošek P, Hoyerová K, Kiran NS, Dobrev PI, Zahajská L, Filepová R, Motyka V, Müller K, Kamínek M (2020) Distinct metabolism of N-glucosides of isopentenyladenine and trans-zeatin determines cytokinin metabolic spectrum in Arabidopsis. New Phytol 225(6):2423–2438. https://doi.org/10.1111/nph.16310
Hoyerová K, Hošek P (2020) New insights into the metabolism and role of cytokinin n-glucosides in plants. Front Plant Sci 11:741–748. https://doi.org/10.3389/fpls.2020.00741
Hu Z, Lan S, Zhao N, Su N, Xue Q, Liu J, Deng Q, Yang J, Zhang M (2019) Soft-X-irradiated pollens induce parthenocarpy in watermelon via rapid changes of hormone-signaling and hormonal regulation. Sci Hortic 250:317–328. https://doi.org/10.1016/j.scienta.2019.02.036
Hwang JU, Song WY, Hong D, Ko D, Yamaoka Y, Jang S, Yim S, Lee E, Khare D, Kim K, Palmgren M (2016) Plant ABC transporters enable many unique aspects of a terrestrial plant’s lifestyle. Mol Plant 9(3):338–355. https://doi.org/10.1016/j.molp.2016.02.003
Jain M, Tyagi AK, Khurana JP (2006) Molecular characterization and differential expression of cytokinin-responsive type-A response regulators in rice (Oryza sativa). BMC Plant Biol 6(1):1–12. https://doi.org/10.1186/1471-2229-6-1
Jameson PE, Song J (2016) Cytokinin: A key driver of seed yield. J Exp Bot 67(3):593–606. https://doi.org/10.1093/jxb/erv461
Jorge GL, Kisiala A, Morrison E, Aoki M, Nogueira AP, Emery RJN (2019) Endosymbiotic Methylobacterium oryzae mitigates the impact of limited water availability in lentil (Lens culinaris Medik.) by increasing plant cytokinin levels. Environ Exp Bot 162:525–540. https://doi.org/10.1016/j.envexpbot.2019.03.028
Joshi S, Choukimath A, Isenegger D, Panozzo J, Spangenberg G, Kant S (2019) Improved wheat growth and yield by delayed leaf senescence using developmentally regulated expression of a cytokinin biosynthesis gene. Front Plant Sci 10:1285–1296. https://doi.org/10.3389/FPLS.2019.01285
Jung B, Flörchinger M, Kunz HH, Traub M, Wartenberg R, Jeblick W, Neuhaus HE, Möhlmann T (2009) Uridine-Ribohydrolase is a key regulator in the uridine degradation pathway of Arabidopsis. Plant Cell 21(3):876–891. https://doi.org/10.1105/tpc.108.062612
Kamada-Nobusada T, Sakakibara H (2009) Molecular basis for cytokinin biosynthesis. Phytochemistry 70:444–449. https://doi.org/10.1016/j.phytochem.2009.02.007
Kambhampati S, Kurepin LV, Kisiala A, Bruce KE, Cober ER, Morrison MJ, Emery RJN (2017) Yield associated traits correlate with cytokinin profiles in developing pods and seeds of field-grown soybean cultivars. Field Crops Res 214:175–184. https://doi.org/10.1016/j.fcr.2017.09.009
Kang J, Lee Y, Sakakibara H, Martinoia E (2017) Cytokinin transporters: Go and stop in signaling. Trends Plant Sci 22(6):455–461. https://doi.org/10.1016/j.tplants.2017.03.003
Karnwal A, Kaushik P (2011) Cytokinin production by fluorescent Pseudomonas in the presence of rice root exudates. Arch Phytopathol 44(17):1728–1735. https://doi.org/10.1080/03235408.2010.526768
Kiba T, Takei K, Kojima M, Sakakibara H (2013) Side-chain modification of cytokinins controls shoot growth in Arabidopsis. Dev Cell 27(4):452–461. https://doi.org/10.1016/j.devcel.2013.10.004
Kieber JJ, Schaller GE (2014) Cytokinins. The Arabidopsis Book/american Society of Plant Biologists. https://doi.org/10.1199/tab.0168
Kieber JJ, Schaller GE (2018) Cytokinin Signaling in Plant Development. Development 145:4–11. https://doi.org/10.1242/dev.149344
Kisiala A, Laffont C, Emery RJN, Frugier F (2013) Bioactive cytokinins are selectively secreted by sinorhizobium meliloti nodulating and nonnodulating strains. Mol Plant Microbe Interact 26(10):1225–1231. https://doi.org/10.1094/MPMI-02-13-0054-R
Kisiala A, Kambhampati S, Stock NL, Aoki M, Emery RN (2019) Quantification of cytokinins using high-resolution accurate-mass Orbitrap mass spectrometry and parallel reaction monitoring (PRM). Anal Chem 91(23):15049–15056. https://doi.org/10.1021/acs.analchem.9b03728
Kojima M, Kamada-Nobusada T, Komatsu H, Takei K, Kuroha T, Mizutani M, Ashikari M, Ueguchi-Tanaka M, Matsuoka M, Suzuki K, Sakakibara H (2009) Highly sensitive and high-throughput analysis of plant hormones using ms-probe modification and liquid chromatography tandem mass spectrometry: An application for hormone profiling in Oryza sativa. Plant Cell Physiol 50(7):1201–1214. https://doi.org/10.1093/pcp/pcp057
Kraigher H, Grayling A, Wang TL, Hanke DE (1991) Cytokinin production by two ectomycorrhizal fungi in liquid culture. Phytochemistry 30(7):2249–2254. https://doi.org/10.1016/0031-9422(91)83623-S
Kubiasová K, Montesinos JC, Šamajová O, Nisler J, Mik V, Semerádová H, Plíhalová L, Novák O, Marhavý P, Cavallari N, Zalabák D (2020) Cytokinin fluoroprobe reveals multiple sites of cytokinin perception at plasma membrane and endoplasmic reticulum. Nat Commun 11(1):1–11. https://doi.org/10.1038/s41467-020-17949-0
Kuderová A, Gallová L, Kuricová K, Nejedlá E, Čurdová A, Micenková L, Plíhal O, Šmajs D, Spíchal L, Hejátko J (2015) Identification of AHK2- and AHK3-like cytokinin receptors in Brassica napus reveals two subfamilies of AHK2 orthologues. J Exp Bot 66(1):339–353. https://doi.org/10.1093/jxb/eru422
Kudo T, Kiba T, Sakakibara H (2010) Metabolism and long-distance translocation of cytokinins. J Integr Plant Biol 52(1):53–60. https://doi.org/10.1111/j.1744-7909.2010.00898.x
Kumar MN, Verslues PE (2015) Stress physiology functions of the Arabidopsis histidine kinase cytokinin receptors. Physiol Plant 154(3):369–380. https://doi.org/10.1111/ppl.12290
Kurepin LV, Emery RJN, Pharis RP, Reid DM (2007) The interaction of light quality and irradiance with gibberellins, cytokinins and auxin in regulating growth of Helianthus annuus hypocotyls. Plant Cell Environ 30(2):147–155. https://doi.org/10.1111/j.1365-3040.2006.01612.x
Kuroha T (2002) A trans-zeatin riboside in root xylem sap negatively regulates adventitious root formation on cucumber hypocotyls. J Exp Bot 53(378):2193–2200. https://doi.org/10.1093/jxb/erf077
Kuroha T, Tokunaga H, Kojima M, Ueda N, Ishida T, Nagawa S, Fukuda H, Sugimoto K, Sakakibara H (2009) Functional analyses of LONELY GUY cytokinin-activating enzymes reveal the importance of the direct activation pathway in Arabidopsis. Plant Cell 21(10):3152–3169. https://doi.org/10.1105/tpc.109.068676
Le DT, Nishiyama R, Watanabe Y, Vankova R, Tanaka M, Seki M, Yamaguchi-Shinozaki K, Shinozaki K, Tran LS (2012) Identification and expression analysis of cytokinin metabolic genes in soybean under normal and drought conditions in relation to cytokinin levels. PLoS ONE 7(8):e42411–e42426. https://doi.org/10.1371/journal.pone.0042411
Li G, Liu K, Baldwin SA, Wang D (2003) Equilibrative nucleoside transporters of Arabidopsis thaliana: cDNA cloning, expression pattern, and analysis of transport activities. J of Biol Chem 278(37):35732–35742. https://doi.org/10.1074/jbc.M304768200
Li Y, Yang L, Wang H, Xu R, Chang S, Hou F, Jia Q (2019) Nutrient and planting modes strategies improves water use efficiency, grain-filling and hormonal changes of maize in semi-arid regions of China. Agric Water Manag 223:105723–105731. https://doi.org/10.1016/j.agwat.2019.105723
Lindner AC, Lang D, Seifert M, Podlešáková K, Novák O, Strnad M, Reski R, von Schwartzenberg K (2014) Isopentenyltransferase-1 (IPT1) knockout in Physcomitrella together with phylogenetic analyses of IPTs provide insights into evolution of plant cytokinin biosynthesis. J Exp Bot 65(9):2533–2543. https://doi.org/10.1093/jxb/eru142
Liu Z, Lv Y, Zhang M, Liu Y, Kong L, Zou M, Lu G, Cao J, Yu X (2013) Identification, expression, and comparative genomic analysis of the IPT and CKX gene families in Chinese cabbage (Brassica rapa ssp. pekinensis). BMC Genom 14:594. https://doi.org/10.1186/1471-2164-14-594
Liu CJ, Zhao Y, Zhang K (2019a) Cytokinin transporters: multisite players in cytokinin homeostasis and signal distribution. Front Plant Sci 10:693. https://doi.org/10.3389/fpls.2019.00693
Liu X, Liang W, Li YX, Li MJ, Ma BQ, Liu CH, Ma FW, Li CY (2019b) Transcriptome analysis reveals the effects of alkali stress on root system architecture and endogenous hormones in apple rootstocks. J Integr Agric 18(10):2264–2271. https://doi.org/10.1016/S2095-3119(19)62706-1
Lomin SN, Yonekura-Sakakibara K, Romanov GA, Sakakibara H (2011) Ligand-binding properties and subcellular localization of maize cytokinin receptors. J Exp Bot 62(14):5149–5159. https://doi.org/10.1093/jxb/err220
Mader JC, Emery RJN, Turnbull CGN (2003) Spatial and temporal changes in multiple hormone groups during lateral bud release shortly following apex decapitation of chickpea (Cicer arietinum) seedlings. Physiol Plant 119(2):295–308. https://doi.org/10.1034/j.1399-3054.2003.00179.x
Mangieri MA, Hall AJ, Striker GG, Chimenti CA (2017) Cytokinins: A key player in determining differences in patterns of canopy senescence in Stay-Green and Fast Dry-Down sunflower (Helianthus annuus L.) hybrids. Eur J Agron 86:60–70. https://doi.org/10.1016/j.eja.2017.03.007
Mapes CC, Davies PJ (2001) Cytokinins in the ball gall of Solidago altissima and in the gall forming larvae of Eurosta solidaginis. New Phytol 151(1):203–212. https://doi.org/10.1046/j.1469-8137.2001.00158.x
Mi X, Wang X, Wu H, Gan L, Ding J, Li Y (2017) Characterization and expression analysis of cytokinin biosynthesis genes in Fragaria vesca. Plant Growth Regul 82(1):139–149. https://doi.org/10.1007/s10725-016-0246-z
Miller CO, Skoog F, Okumura FS, Von Saltza MH, Strong FM (1956) Isolation, structure and synthesis of kinetin, a substance promoting cell division. J Am Chem Soc 78(7):1375–1380. https://doi.org/10.1021/ja01588a032
Morrison EN, Emery RJN, Saville BJ (2015a) Phytohormone involvement in the Ustilago maydis- Zea mays pathosystem: Relationships between abscisic acid and cytokinin levels and strain virulence in infected cob tissue. PLoS ONE 10(6):e0130945–e0130969. https://doi.org/10.1371/journal.pone.0130945
Morrison EN, Knowles S, Hayward A, Thorn RG, Saville BJ, Emery RJ (2015b) Detection of phytohormones in temperate forest fungi predicts consistent abscisic acid production and a common pathway for cytokinin biosynthesis. Mycologia 107(2):245–257. https://doi.org/10.3852/14-157
Müller B, Sheen J (2008) Cytokinin and auxin interaction in root stem-cell specification during early embryogenesis. Nature 453(7198):1094–1097. https://doi.org/10.1038/nature06943
Nawiri S, Oduor R, Mbinda W (2018) Isopentenyletransferase gene enhances drought tolerance in genetically engineered sweetpotato (Ipomoea batatas (L.) Lam). J Plant Biochem. Physiol 6(3):221. https://doi.org/10.4172/2329-9029.1000221
Nguyen HN, Lai N, Kisiala AB, Emery RJN (2021) Isopentenyltransferases (IPT) as master regulators of crop performance: their function, manipulation and genetic potential for stress adaptation and yield improvement. Plant Biotechnol J. https://doi.org/10.1111/1467-7652
Noble A, Kisiala A, Galer A, Clysdale D, Emery RJN (2014) Euglena gracilis (Euglenophyceae) produces abscisic acid and cytokinins and responds to their exogenous application singly and in combination with other growth regulators. Eur J Phycol 49(2):244–254. https://doi.org/10.1080/09670262.2014.911353
Novák O, Tarkowski P, Tarkowská D, Doležal K, Lenobel R, Strnad M (2003) Quantitative analysis of cytokinins in plants by liquid chromatography-single-quadrupole mass spectrometry. Anal Chim Acta 480(2):207–218. https://doi.org/10.1016/S0003-2670(03)00025-4
Ogawa J, Takeda S, Xie SX, Hatanaka H, Ashikari T, Amachi T, Shimizu S (2001) Purification, characterization, and gene cloning of purine nucleosidase from Ochrobactrum anthropi. Appl Environ Microbiol 67(4):1783–1787. https://doi.org/10.1128/AEM.67.4.1783-1787.2001
Oslovsky VE, Savelieva EM, Drenichev MS, Romanov GA, Mikhailov SN (2020) Distinct peculiarities of in planta synthesis of isoprenoid and aromatic cytokinins. Biomolecules 10(1):86–100. https://doi.org/10.3390/biom10010086
Osugi A, Sakakibara H (2015) Q and A: How do plants respond to cytokinins and what is their importance? BMC Biol 13(1):1–10. https://doi.org/10.1186/s12915-015-0214-5
Osugi A, Kojima M, Takebayashi Y, Ueda N, Kiba T, Sakakibara H (2017) Systemic transport of trans-zeatin and its precursor have differing roles in Arabidopsis shoots. Nat Plants 3(8):1–6. https://doi.org/10.1038/nplants.2017.112
Pallai R, Hynes RK, Verma B, Nelson LM (2012) Phytohormone production and colonization of canola (Brassica napus L) roots by Pseudomonas fluorescens 6–8 under gnotobiotic conditions. Can J Microbiol 58(2):170–178. https://doi.org/10.1139/w11-120
Palni LM, Summons R, Letham D (1983) Mass spectrometric analysis of cytokinins in plant tissues: V. Identification of the cytokinin complex of Datura innoxia crown gall tissue. Plant Physiol 72(3):858–863. https://doi.org/10.1104/pp.72.3.858
Panda BB, Sekhar S, Dash SK, Behera L, Shaw BP (2018) Biochemical and molecular characterisation of exogenous cytokinin application on grain filling in rice. BMC Plant Biol 18(1):89–108. https://doi.org/10.1186/s12870-018-1279-4
Pate JS (1980) Transport and partitioning of nitrogenous solutes. Annu Rev Plant Physiol 31(1):313–340. https://doi.org/10.1146/annurev.pp.31.060180.001525
Pate JS, Atkins CA, Hamel K, McNeil DL, Layzell DB (1979) Transport of organic solutes in phloem and xylem of a nodulated legume. Plant Physiol 63(6):1082–1088. https://doi.org/10.1104/pp.63.6.1082
Pertry I, Václavíková K, Depuydt S, Galuszka P, Spíchal L, Temmerman W, Stes E, Schmülling T, Kakimoto T (2009) Identification of Rhodococcus fascians cytokinins and their modus operandi to reshape the plant. PNAS 106(3):929–934. https://doi.org/10.1073/pnas.0811683106
Polya GM (1974) Regulation of a plant 5’(3’) ribonucleotide phosphohydrolase by cyclic nucleotides and pyrimidine, purine, and cytokinin ribosides. PNAS 71(4):1299–1303. https://doi.org/10.1073/pnas.71.4.1299
Powell AF, Paleczny AR, Olechowski H, Emery RJN (2013) Changes in cytokinin form and concentration in developing kernels correspond with variation in yield among field-grown barley cultivars. Plant Physiol Biochem 64:33–40. https://doi.org/10.1016/j.plaphy.2012.12.010
Prerostova S, Dobrev PI, Kramna B, Gaudinova A, Knirsch V, Spichal L, Zatloukal M, Vankova R (2020) Heat acclimation and inhibition of cytokinin degradation positively affect heat stress tolerance of Arabidopsis. Front Plant Sci 11:87–101. https://doi.org/10.3389/fpls.2020.00087
Prudent M, Salon C, Smith DL, Emery RJN (2016) Nod factor supply under water stress conditions modulates cytokinin biosynthesis and enhances nodule formation and N nutrition in soybean. Plant Signal Behav 11(9):e1212799–e1212805. https://doi.org/10.1080/15592324.2016.1212799
Průšová A (2016) Light on phloem transport (an MRI approach). Dissertation, Wageningen University
Qi Z, Xiong L (2013) Characterization of a purine permease family gene OsPUP7 involved in growth and development control in rice. J Integr Plant Biol 55(11):1119–1135. https://doi.org/10.1111/jipb.12101
Qin S, Chen X, Jiang C, Li M, Yuan Y, Yang J, Wu Q (2019) Pruning induced yield and quality variations and the correlated gene expression and phytohormone changes in Lonicera japonica. Ind Crops Prod 132:386–395. https://doi.org/10.1016/j.indcrop.2019.02.048
Quesnelle PE, Emery RJN (2007) cis-cytokinins that predominate in Pisum sativum during early embryogenesis will accelerate embryo growth in vitro. Can J Bot 85(1):91–103. https://doi.org/10.1139/B06-149
Ralph RK, Wojcik SJ (1977) Effects of N6-benzyladenine on nucleotides in Chinese cabbage leaf discs. Plant Sci Lett 10(1):41–44. https://doi.org/10.1016/0304-4211(77)90047-5
Raspor M, Motyka V, Ninković S, Dobrev PI, Malbeck J, Ćosić T, Cingel A, Savić J, Tadić V, Dragićević IČ (2020) Endogenous levels of cytokinins, indole-3-acetic acid and abscisic acid in in vitro grown potato: A contribution to potato hormonomics. Sci Rep 10(1):1–13. https://doi.org/10.1038/s41598-020-60412-9
Reid D, Nadzieja M, Novák O, Heckmann AB, Sandal N, Stougaard J (2017) Cytokinin biosynthesis promotes cortical cell responses during nodule development. Plant Physiol 175(1):361–375. https://doi.org/10.1104/pp.17.00832
Riegler H, Geserick C, Zrenner R (2011) Arabidopsis thaliana nucleosidase mutants provide new insights into nucleoside degradation. New Phytol 191(2):349–359. https://doi.org/10.1111/j.1469-8137.2011.03711.x
Rolle RS, Chism GW (1989) Kinetic comparison of cytokinin nucleosidase activity isolated from normally ripening and mutant tomato varieties. Plant Physiol 91(1):148–150. https://doi.org/10.1104/pp.91.1.148
Romanov GA, Schmülling T (2021) Opening doors for cytokinin trafficking at the ER membrane. Trends Plant Sci 26(4):305–308. https://doi.org/10.1016/j.tplants.2021.02.006
Romanov GA, Lomin SN, Schmülling T (2006) Biochemical characteristics and ligand-binding properties of Arabidopsis cytokinin receptor AHK3 compared to CRE1/AHK4 as revealed by a direct binding assay. J Exp Bot 57(15):4051–4058. https://doi.org/10.1093/jxb/erl179
Sakakibara H (2006) Cytokinins: Activity, biosynthesis, and translocation. Annu Rev Plant Biol 57:431–449. https://doi.org/10.1146/annurev.arplant.57.032905.105231
Sakakibara H (2010) Cytokinin biosynthesis and metabolism. Springer, Dordrecht 2010:95–114
Samuelson ME, Eliasson L, Larsson CM (1992) Nitrate-regulated growth and cytokinin responses in seminal roots of barley. Plant Physiol 98(1):309–315. https://doi.org/10.1104/pp.98.1.309
Schäfer M, Brütting C, Meza-Canales ID, Großkinsky DK, Vankova R, Baldwin IT, Meldau S (2015) The role of cis-zeatin-type cytokinins in plant growth regulation and mediating responses to environmental interactions. J Exp Bot 66(16):4873–4884. https://doi.org/10.1093/jxb/erv214
Schaller GE, Street IH, Kieber JJ (2014) Cytokinin and the cell cycle. Curr Op Plant Biol 21:7–15. https://doi.org/10.1016/j.pbi.2014.05.015
Seegobin M, Kisiala A, Noble A, Kaplan D, Brunetti C, Emery RJN (2018) Canis familiaris tissues are characterized by different profiles of cytokinins typical of the tRNA degradation pathway. FASEB J 32(12):6575–6581. https://doi.org/10.1096/fj.201800347
Shoaib M, Yang W, Shan Q, Sajjad M, Zhang A (2019) Genome-wide identification and expression analysis of new cytokinin metabolic genes in bread wheat (Triticum aestivum L.). PeerJ 7:e6300–e6323. https://doi.org/10.7717/peerj.6300
Sokołowska K (2013) Symplasmic transport in wood: the importance of living xylem cells. Symplasmic transport in vascular plants. Springer, New York, pp 101–132
Stolz A, Riefler M, Lomin SN, Achazi K, Romanov GA, Schmülling T (2011) The specificity of cytokinin signalling in Arabidopsis thaliana is mediated by differing ligand affinities and expression profiles of the receptors. Plant J 67(1):157–168. https://doi.org/10.1111/j.1365-313X.2011.04584.x
Straka JR, Hayward AR, Emery RJN (2010) Gall-inducing Pachypsylla celtidis (Psyllidae) infiltrate hackberry trees with high concentrations of phytohormones. J Plant Interact 5(3):197–203. https://doi.org/10.1080/17429145.2010.484552
Sun J, Hirose N, Wang X, Wen P, Xue L, Sakakibara H, Zuo J (2005) Arabidopsis SOI33/AtENT8 gene encodes a putative equilibrative nucleoside transporter that is involved in cytokinin transport in planta. J Integr Plant Biol 47(5):588–603. https://doi.org/10.1111/j.1744-7909.2005.00104.x
Svačinová J, Novák O, Plačková L, Lenobel R, Holík J, Strnad M, Doležal K (2012) A new approach for cytokinin isolation from Arabidopsis tissues using miniaturized purification: pipette tip solid-phase extraction. Plant Methods 8(1):17–31. https://doi.org/10.1186/1746-4811-8-17
Takagi M, Yokota T, Murofushi N, Saka H, Takahashi N (1989) Quantitative changes of free-base, riboside, ribotide and glucoside cytokinins in developing rice grains. Plant Growth Regul 8(4):349–364. https://doi.org/10.1007/BF00024665
Takei K, Sakakibara H, Taniguchi M, Sugiyama T (2001) Nitrogen-dependent accumulation of cytokinins in root and the translocation to leaf: Implication of cytokinin species that induces gene expression of maize response regulator. Plant Cell Physiol 42(1):85–93. https://doi.org/10.1093/pcp/pce009
Takei K, Yamaya T, Sakakibara H (2004) Arabidopsis CYP735A1 and CYP735A2 encode cytokinin hydroxylases that catalyze the biosynthesis of trans-Zeatin. J Biol Chem 279(40):41866–41872. https://doi.org/10.1074/jbc.M406337200
Talarek-Karwel M, Bajguz A, Piotrowska-Niczyporuk A (2020) Hormonal response of Acutodesmus obliquus exposed to combined treatment with 24-epibrassinolide and lead. J Appl Phycol 32(5):2903–2914. https://doi.org/10.1007/s10811-020-02191-4
Tan M, Li G, Qi S, Liu X, Chen X, Ma J, Zhang D, Han M (2018) Identification and expression analysis of the IPT and CKX gene families during axillary bud outgrowth in apple (Malus domestica Borkh.). Gene 651:106–117. https://doi.org/10.1016/j.gene.2018.01.101
Tang YJ, Qian W, Xue JY, Yan L, Li RM, Van Nocker S, Wang YJ, Zhang CH (2018) Gene cloning and expression analyses of WBC genes in the developing grapevine seeds. J Integr Agric 17(6):1348–1359. https://doi.org/10.1016/S2095-3119(17)61827-6
Tarkowská D, Filek M, Biesaga-Kościelniak J, Marcińska I, Macháčková I, Krekule J, Strnad M (2012) Cytokinins in shoot apices of Brassica napus plants during vernalization. Plant Sci 187:105–112. https://doi.org/10.1016/j.plantsci.2012.02.003
Tarkowski P, Ge L, Yong JWH, Tan SN (2009) Analytical methods for cytokinins. Trends Anal Chem 28(3):323–335. https://doi.org/10.1016/j.trac.2008.11.010
Tessi TM, Brumm S, Winklbauer E, Schumacher B, Pettinari G, Lescano I, González CA, Wanke D, Maurino VG, Harter K, Desimone M (2021) Arabidopsis AZG2 transports cytokinins in vivo and regulates lateral root emergence. New Phytol 229(2):979–993. https://doi.org/10.1111/nph.16943
To JPC, Kieber JJ (2008) Cytokinin signaling: two-components and more. Trends Plant Sci 13:85–92. https://doi.org/10.1016/j.tplants.2007.11.005
To JPC, Haberer G, Ferreira FJ, Deruere J, Mason MG, Schaller GE, Alonso JM, Ecker JR, Kieber JJ (2004) Type-A Arabidopsis response regulators are partially redundant negative regulators of cytokinin signaling. Plant Cell 16(3):658–671. https://doi.org/10.1105/tpc.018978
Tokunaga H, Kojima M, Kuroha T, Ishida T, Sugimoto K, Kiba T, Sakakibara H (2012) Arabidopsis lonely guy (LOG) multiple mutants reveal a central role of the LOG-dependent pathway in cytokinin activation. Plant J 69(2):355–365. https://doi.org/10.1111/j.1365-313X.2011.04795.x
Tran LSP, Urao T, Qin F, Maruyama K, Kakimoto T, Shinozaki K, Yamaguchi-Shinozaki K (2007) Functional analysis of AHK1/ATHK1 and cytokinin receptor histidine kinases in response to abscisic acid, drought, and salt stress in Arabidopsis. PNAS 104(51):20623–20628. https://doi.org/10.1073/pnas.0706547105
Tuan PA, Yamasaki Y, Kanno Y, Seo M, Ayele BT (2019) Transcriptomics of cytokinin and auxin metabolism and signaling genes during seed maturation in dormant and non-dormant wheat genotypes. Sci Rep 9(1):1–16. https://doi.org/10.1038/s41598-019-40657-9
Vanková R, Kosová K, Dobrev P, Vítámvás P, Trávníčková A, Cvikrová M, Pešek B, Gaudinová A, Prerostová S, Musilová J, Galiba G (2014) Dynamics of cold acclimation and complex phytohormone responses in Triticum monococcum lines G3116 and DV92 differing in vernalization and frost tolerance level. Env Exp Bot 101:12–25. https://doi.org/10.1016/j.envexpbot.2014.01.002
Wang W, Hao Q, Wang W, Li Q, Chen F, Ni F, Wang Y, Fu D, Wu J, Wang W (2019) The involvement of cytokinin and nitrogen metabolism in delayed flag leaf senescence in a wheat stay-green mutant, tasg1. Plant Sci 278:70–79. https://doi.org/10.1016/j.plantsci.2018.10.024
Wang L, Xue J, Yan J, Liu M, Tang Y, Wang Y, Zhang C (2020) Expression and functional analysis of VviABCG14 from Vitis vinifera suggest the role in cytokinin transport and the interaction with VviABCG7. Plant Physiol Biochem 153:1–10. https://doi.org/10.1016/j.plaphy.2020.05.011
Wheeldon CD, Bennett T (2020) There and back again: An evolutionary perspective on long-distance coordination of plant growth and development. Academic Press, Semin Cell Dev Biol. https://doi.org/10.1016/j.semcdb.2020.06.011
Wulfetange K, Lomin SN, Romanov GA, Stolz A, Heyl A, Schmülling T (2011) The cytokinin receptors of Arabidopsis are located mainly to the endoplasmic reticulum. Plant Physiol 156(4):1808–1818. https://doi.org/10.1104/pp.111.180539
Wybouw B, De Rybel B (2019) Cytokinin – A developing story. Trends Plant Sci 24(2):177–185. https://doi.org/10.1016/j.tplants.2018.10.012
Xiao Y, Zhang J, Yu G, Lu X, Mei W, Deng H, Zhang G, Chen G, Chu C, Tong H, Tang W (2020) Endoplasmic reticulum-localized PURINE PERMEASE1 regulates plant height and grain weight by modulating cytokinin distribution in rice. Front Plant Sci 11:618560. https://doi.org/10.3389/fpls.2020.618560
Yang J, Peng S, Visperas RM, Sanico AL, Zhu Q, Gu S (2000) Grain filling pattern and cytokinin content in the grains and roots of rice plants. Plant Growth Regul 30(3):261–270. https://doi.org/10.1023/A:1006356125418
Yang W, Cortijo S, Korsbo N, Roszak P, Schiessl K, Gurzadyan A, Wightman R, Jönsson H, Meyerowitz E (2021) Molecular mechanism of cytokinin-activated cell division in Arabidopsis. Science 371.6536:1350–1355. https://doi.org/10.1126/science.abe2305
Zalabák D, Pospíšilová H, Šmehilová M, Mrízová K, Frébort I, Galuszka P (2013) Genetic engineering of cytokinin metabolism: Prospective way to improve agricultural traits of crop plants. Biotechnol Adv 31(1):97–117. https://doi.org/10.1016/j.biotechadv.2011.12.003
Zalabák D, Galuszka P, Mrízová K, Podlešáková K, Gu R, Frébortová J (2014) Biochemical characterization of the maize cytokinin dehydrogenase family and cytokinin profiling in developing maize plantlets in relation to the expression of cytokinin dehydrogenase genes. Plant Physiol Biochem 74:283–293. https://doi.org/10.1016/j.plaphy.2013.11.020
Zemanová V, Pavlíková D, Dobrev PI, Motyka V, Pavlik M (2019) Endogenous phytohormone profiles in Pteris fern species differing in arsenic accumulating ability. Environ Exp Bot 166:103822–103832. https://doi.org/10.1016/j.envexpbot.2019.103822
Zhang K, Novak O, Wei Z, Gou M, Zhang X, Yu Y, Yang H, Cai Y, Strnad M, Liu CJ (2014) Arabidopsis ABCG14 protein controls the acropetal translocation of root-synthesized cytokinins. Nat Commun 5(1):1–12. https://doi.org/10.1038/ncomms4274
Zhang YJ, Li A, Liu XQ, Sun JX, Guo WJ, Zhang JW, Lyu YM (2019) Changes in the morphology of the bud meristem and the levels of endogenous hormones after low temperature treatment of different Phalaenopsis cultivars. S Afr J Bot 125:499–504. https://doi.org/10.1016/j.sajb.2019.08.016
Zhao J, Yu N, Ju M, Fan B, Zhang Y, Zhu E, Zhang M, Zhang K (2019) ABC transporter OsABCG18 controls the shootward transport of cytokinins and grain yield in rice. J Exp Bot 70(21):6277–6291. https://doi.org/10.1093/jxb/erz382
Zhao J, Ding B, Zhu E, Deng X, Zhang M, Zhang P, Wang L, Dai Y, Xiao S, Zhang C, Liu CJ (2021) Phloem 25 unloading via the apoplastic pathway is essential for shoot distribution of root-synthesized cytokinins. Plant Physiol. https://doi.org/10.1093/plphys/kiab188
Zhou M, Ghnaya T, Dailly H, Cui G, Vanpee B, Han R, Lutts S (2019) The cytokinin trans-zeatine riboside increased resistance to heavy metals in the halophyte plant species Kosteletzkya pentacarpos in the absence but not in the presence of NaCl. Chemosphere 233:954–965. https://doi.org/10.1016/j.chemosphere.2019.06.023
Žižková E, Kubeš M, Dobrev PI, Přibyl P, Šimura J, Zahajská L, Záveská Drábková L, Novák O, Motyka V (2017) Control of cytokinin and auxin homeostasis in cyanobacteria and algae. Ann Bot 119(1):151–166. https://doi.org/10.1093/aob/mcw194
Acknowledgements
Financial support from the Natural Sciences and Engineering Council of Canada (RGPIN-05436 and STPGP 521417 to RJNE) is gratefully acknowledged. HNN is supported by International Graduate Scholarship (IGS) from Environmental Life Sciences Graduate (EnLS) Program, Trent University.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
The authors report no commercial or proprietary interest in any product or concept discussed in this article.
Additional information
Communicated by Gerhard Leubner.
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Nguyen, H.N., Nguyen, T.Q., Kisiala, A.B. et al. Beyond transport: cytokinin ribosides are translocated and active in regulating the development and environmental responses of plants. Planta 254, 45 (2021). https://doi.org/10.1007/s00425-021-03693-2
Received:
Accepted:
Published:
DOI: https://doi.org/10.1007/s00425-021-03693-2