Abstract
During memory formation, structural rearrangements of dendritic spines provide a mean to durably modulate synaptic connectivity within neuronal networks. New neurons generated throughout the adult life in the dentate gyrus of the hippocampus contribute to learning and memory. As these neurons become incorporated into the network, they generate huge numbers of new connections that modify hippocampal circuitry and functioning. However, it is yet unclear as to how the dynamic process of memory formation influences their synaptic integration into neuronal circuits. New memories are established according to a multistep process during which new information is first acquired and then consolidated to form a stable memory trace. Upon recall, memory is transiently destabilized and vulnerable to modification. Using contextual fear conditioning, we found that learning was associated with an acceleration of dendritic spines formation of adult-born neurons, and that spine connectivity becomes strengthened after memory consolidation. Moreover, we observed that afferent connectivity onto adult-born neurons is enhanced after memory retrieval, while extinction training induces a change of spine shapes. Together, these findings reveal that the neuronal activity supporting memory processes strongly influences the structural dendritic integration of adult-born neurons into pre-existing neuronal circuits. Such change of afferent connectivity is likely to impact the overall wiring of hippocampal network, and consequently, to regulate hippocampal function.





Similar content being viewed by others
References
Aimone JB, Deng W, Gage FH (2011) Resolving new memories: a critical look at the dentate gyrus, adult neurogenesis, and pattern separation. Neuron 70:589–596
Alberini CM (2005) Mechanisms of memory stabilization: are consolidation and reconsolidation similar or distinct processes? Trends Neurosci 28:51–56
Alberini CM, Ledoux JE (2013) Memory reconsolidation. Curr Biol 23:R746–R750
Andreadis ST, Brott D, Fuller AO, Palsson BO (1997) Moloney murine leukemia virus-derived retroviral vectors decay intracellularly with a half-life in the range of 5.5 to 7.5 hours. J Virol 71:7541–7548
Arellano JI, Benavides-Piccione R, Defelipe J, Yuste R (2007) Ultrastructure of dendritic spines: correlation between synaptic and spine morphologies. Front Neurosci 1:131–143
Arruda-Carvalho M, Sakaguchi M, Akers KG, Josselyn SA, Frankland PW (2011) Posttraining ablation of adult-generated neurons degrades previously acquired memories. J Neurosci 31:15113–15127
Bast T, da Silva BM, Morris RG (2005) Distinct contributions of hippocampal NMDA and AMPA receptors to encoding and retrieval of one-trial place memory. J Neurosci 25:5845–5856
Bergami M et al (2015) A critical period for experience-dependent remodeling of adult-born neuron connectivity. Neuron 85:710–717
Bouton ME (2004) Context and behavioral processes in extinction. Learn Mem 11:485–494
Clelland CD et al (2009) A functional role for adult hippocampal neurogenesis in spatial pattern separation. Science 325:210–213
De Roo M, Klauser P, Mendez P, Poglia L, Muller D (2008) Activity-dependent PSD formation and stabilization of newly formed spines in hippocampal slice cultures. Cereb Cortex 18:151–161
Debiec J, LeDoux JE, Nader K (2002) Cellular and systems reconsolidation in the hippocampus. Neuron 36:527–538
Deng W, Saxe MD, Gallina IS, Gage FH (2009) Adult-born hippocampal dentate granule cells undergoing maturation modulate learning and memory in the brain. J Neurosci 29:13532–13542
Denny CA, Burghardt NS, Schachter DM, Hen R, Drew MR (2012) 4- to 6-week-old adult-born hippocampal neurons influence novelty-evoked exploration and contextual fear conditioning. Hippocampus 22:1188–1201
Ding XF, Gao X, Ding XC, Fan M, Chen J (2016) Postnatal dysregulation of Notch signal disrupts dendrite development of adult-born neurons in the hippocampus and contributes to memory impairment. Sci Rep 6:25780
Dudai Y (2012) The restless engram: consolidations never end. Annu Rev Neurosci 35:227–247
Eichenbaum H (2000) A cortical–hippocampal system for declarative memory. Nat Rev Neurosci 1:41–50
Epp JR, Silva Mera R, Köhler S, Josselyn SA, Frankland PW (2016) Neurogenesis-mediated forgetting minimizes proactive interference. Nat Commun 7:10838
Feng R et al (2001) Deficient neurogenesis in forebrain-specific presenilin-1 knockout mice is associated with reduced clearance of hippocampal memory traces. Neuron 32:911–926
Frankland PW, Bontempi B (2005) The organization of recent and remote memories. Nat Rev Neurosci 6:119–130
Frankland PW, Bontempi B, Talton LE, Kaczmarek L, Silva AJ (2004) The involvement of the anterior cingulate cortex in remote contextual fear memory. Science 304:881–883
Gafford GM, Parsons RG, Helmstetter FJ (2013) Memory accuracy predicts hippocampal mTOR pathway activation following retrieval of contextual fear memory. Hippocampus 23:842–847
Garthe A, Behr J, Kempermann G (2009) Adult-generated hippocampal neurons allow the flexible use of spatially precise learning strategies. PLoS One 4:e5464
Ge S, Yang CH, Hsu KS, Ming GL, Song H (2007) A critical period for enhanced synaptic plasticity in newly generated neurons of the adult brain. Neuron 54:559–566
Goncalves JT et al (2016) In vivo imaging of dendritic pruning in dentate granule cells. Nat Neurosci 19:788–791
Gould E, Gross CG (2002) Neurogenesis in adult mammals: some progress and problems. J Neurosci 22:619–623
Gu Y, Arruda-Carvalho M, Wang J, Janoschka SR, Josselyn SA, Frankland PW, Ge S (2012) Optical controlling reveals time-dependent roles for adult-born dentate granule cells. Nat Neurosci 15:1700–1706
Harris KM, Jensen FE, Tsao B (1992) Three-dimensional structure of dendritic spines and synapses in rat hippocampus (CA1) at postnatal day 15 and adult ages: implications for the maturation of synaptic physiology and long-term potentiation. J Neurosci 12:2685–2705
Hebb DO (1949) The organization of behavior: a neuropsychological theory. Wiley, New York
Holtmaat AJ, Trachtenberg JT, Wilbrecht L, Shepherd GM, Zhang X, Knott GW, Svoboda K (2005) Transient and persistent dendritic spines in the neocortex in vivo. Neuron 45:279–291
Huang YB, Hu CR, Zhang L, Yin W, Hu B (2015) In vivo study of dynamics and stability of dendritic spines on olfactory bulb interneurons in Xenopus laevis tadpoles. PLoS One 10:e0140752
Izquierdo I, Medina JH (1997) Memory formation: the sequence of biochemical events in the hippocampus and its connection to activity in other brain structures. Neurobiol Learn Mem 68:285–316
Izquierdo I, Schroder N, Netto CA, Medina JH (1999) Novelty causes time-dependent retrograde amnesia for one-trial avoidance in rats through NMDA receptor- and CaMKII-dependent mechanisms in the hippocampus. Eur J Neurosci 11:3323–3328
Kandel ER (2001) The molecular biology of memory storage: a dialogue between genes and synapses. Science 294:1030–1038
Kasai H, Fukuda M, Watanabe S, Hayashi-Takagi A, Noguchi J (2010) Structural dynamics of dendritic spines in memory and cognition. Trends Neurosci 33:121–129
Kee N, Teixeira CM, Wang AH, Frankland PW (2007) Preferential incorporation of adult-generated granule cells into spatial memory networks in the dentate gyrus. Nat Neurosci 10:355–362
Kheirbek MA, Tannenholz L, Hen R (2012) NR2B-dependent plasticity of adult-born granule cells is necessary for context discrimination. J Neurosci 32:8696–8702
Kitamura T, Inokuchi K (2014) Role of adult neurogenesis in hippocampal–cortical memory consolidation. Mol Brain 7:13
Kitamura T et al (2012) Hippocampal function is not required for the precision of remote place memory. Mol Brain 5:5
Ko HG et al (2009) Effect of ablated hippocampal neurogenesis on the formation and extinction of contextual fear memory. Mol Brain 2:1
Kopec CD, Li B, Wei W, Boehm J, Malinow R (2006) Glutamate receptor exocytosis and spine enlargement during chemically induced long-term potentiation. J Neurosci 26:2000–2009
Krezymon A et al (2013) Modifications of hippocampal circuits and early disruption of adult neurogenesis in the Tg2576 mouse model of Alzheimer’s disease. PLoS One 8:e76497
Kropff E, Yang SM, Schinder AF (2015) Dynamic role of adult-born dentate granule cells in memory processing. Curr Opin Neurobiol 35:21–26
Lai CS, Franke TF, Gan WB (2012) Opposite effects of fear conditioning and extinction on dendritic spine remodelling. Nature 483:87–91
Lamprecht R, LeDoux J (2004) Structural plasticity and memory. Nat Rev Neurosci 5:45–54
Majewska AK, Newton JR, Sur M (2006) Remodeling of synaptic structure in sensory cortical areas in vivo. J Neurosci 26:3021–3029
Marin-Burgin A, Mongiat LA, Pardi MB, Schinder AF (2012) Unique processing during a period of high excitation/inhibition balance in adult-born neurons. Science 335:1238–1242
Matsuzaki M (2007) Factors critical for the plasticity of dendritic spines and memory storage. Neurosci Res 57:1–9
Matsuzaki M, Honkura N, Ellis-Davies GC, Kasai H (2004) Structural basis of long-term potentiation in single dendritic spines. Nature 429:761–766
Maviel T, Durkin TP, Menzaghi F, Bontempi B (2004) Sites of neocortical reorganization critical for remote spatial memory. Science 305:96–99
McGaugh JL (2000) Memory–a century of consolidation. Science 287:248–251
Ming GL, Song H (2005) Adult neurogenesis in the mammalian central nervous system. Annu Rev Neurosci 28:223–250
Myers KM, Ressler KJ, Davis M (2006) Different mechanisms of fear extinction dependent on length of time since fear acquisition. Learn Mem 13:216–223
Nader K, Hardt O (2009) A single standard for memory: The case for reconsolidation. Nat Rev Neurosci 10:224–234
Nader K, Schafe GE, Le Doux JE (2000a) Fear memories require protein synthesis in the amygdala for reconsolidation after retrieval. Nature 406:722–726
Nader K, Schafe GE, LeDoux JE (2000b) Reply—Reconsolidation: The labile nature of consolidation theory. Nat Rev Neurosci 1:216–219
Nusser Z, Lujan R, Laube G, Roberts JD, Molnar E, Somogyi P (1998) Cell type and pathway dependence of synaptic AMPA receptor number and variability in the hippocampus. Neuron 21:545–559
Pan YW, Storm DR, Xia Z (2013) Role of adult neurogenesis in hippocampus-dependent memory, contextual fear extinction and remote contextual memory: New insights from ERK5 MAP kinase. Neurobiol Learn Mem 105:81–92
Quirk GJ, Pare D, Richardson R, Herry C, Monfils MH, Schiller D, Vicentic A (2010) Erasing fear memories with extinction training. J Neurosci 30:14993–14997
Rampon C, Tang YP, Goodhouse J, Shimizu E, Kyin M, Tsien JZ (2000) Enrichment induces structural changes and recovery from nonspatial memory deficits in CA1 NMDAR1-knockout mice. Nat Neurosci 3:238–244
Restivo L, Vetere G, Bontempi B, Ammassari-Teule M (2009) The formation of recent and remote memory is associated with time-dependent formation of dendritic spines in the hippocampus and anterior cingulate cortex. J Neurosci 29:8206–8214
Restivo L, Niibori Y, Mercaldo V, Josselyn SA, Frankland PW (2015) Development of adult-generated cell connectivity with excitatory and inhibitory cell populations in the hippocampus. J Neurosci 35:10600–10612
Roberts TF, Tschida KA, Klein ME, Mooney R (2010) Rapid spine stabilization and synaptic enhancement at the onset of behavioural learning. Nature 463:948–952
Roybon L, Hjalt T, Stott S, Guillemot F, Li JY, Brundin P (2009) Neurogenin2 directs granule neuroblast production and amplification while NeuroD1 specifies neuronal fate during hippocampal neurogenesis. PLoS One 4:e4779
Sahay A, Wilson DA, Hen R (2011) Pattern separation: A common function for new neurons in hippocampus and olfactory bulb. Neuron 70:582–588
Sase S et al (2015) Individual phases of contextual fear conditioning differentially modulate dorsal and ventral hippocampal GluA1-3, GluN1-containing receptor complexes and subunits. Hippocampus 25:1501–1516
Schiller D, Cain CK, Curley NG, Schwartz JS, Stern SA, Ledoux JE, Phelps EA (2008) Evidence for recovery of fear following immediate extinction in rats and humans. Learn Mem 15:394–402
Snyder JS, Hong NS, McDonald RJ, Wojtowicz JM (2005) A role for adult neurogenesis in spatial long-term memory. Neuroscience 130:843–852
Suarez-Pereira I, Carrion AM (2015) Updating stored memory requires adult hippocampal neurogenesis. Sci Rep 5:13993
Toni N, Sultan S (2011) Synapse formation on adult-born hippocampal neurons. Eur J Neurosci 33:1062–1068
Toni N, Laplagne DA, Zhao C, Lombardi G, Ribak CE, Gage FH, Schinder AF (2008) Neurons born in the adult dentate gyrus form functional synapses with target cells. Nat Neurosci 11:901–907
Trinchero MF et al (2015) Effects of spaced learning in the water maze on development of dentate granule cells generated in adult mice. Hippocampus 25:1314–1326
Tronel S, Fabre A, Charrier V, Oliet SH, Gage FH, Abrous DN (2010) Spatial learning sculpts the dendritic arbor of adult-born hippocampal neurons. Proc Natl Acad Sci USA 107:7963–7968
Trouche S, Bontempi B, Roullet P, Rampon C (2009) Recruitment of adult-generated neurons into functional hippocampal networks contributes to updating and strengthening of spatial memory. Proc Natl Acad Sci USA 106:5919–5924
Tse D et al (2007) Schemas and memory consolidation. Science 316:76–82
Wang SH, Teixeira CM, Wheeler AL, Frankland PW (2009) The precision of remote context memories does not require the hippocampus. Nat Neurosci 12:253–255
Wiltgen BJ et al (2010) The hippocampus plays a selective role in the retrieval of detailed contextual memories. Curr Biol 20:1336–1344
Wu MV, Hen R (2014) Functional dissociation of adult-born neurons along the dorsoventral axis of the dentate gyrus. Hippocampus 24:751–761
Xu T et al (2009) Rapid formation and selective stabilization of synapses for enduring motor memories. Nature 462:915–919
Yang Y, Wang XB, Frerking M, Zhou Q (2008) Spine expansion and stabilization associated with long-term potentiation. J Neurosci 28:5740–5751
Yang G, Pan F, Gan WB (2009) Stably maintained dendritic spines are associated with lifelong memories. Nature 462:920–924
Yuste R, Bonhoeffer T (2001) Morphological changes in dendritic spines associated with long-term synaptic plasticity. Annu Rev Neurosci 24:1071–1089
Zhang Y, Cudmore RH, Lin DT, Linden DJ, Huganir RL (2015) Visualization of NMDA receptor-dependent AMPA receptor synaptic plasticity in vivo. Nat Neurosci 18:402–407
Zhao C, Teng EM, Summers RG Jr, Ming GL, Gage FH (2006) Distinct morphological stages of dentate granule neuron maturation in the adult mouse hippocampus. J Neurosci 26:3–11
Zhao C, Jou J, Wolff LJ, Sun H, Gage FH (2014) Spine morphogenesis in newborn granule cells is differentially regulated in the outer and middle molecular layers. J Comp Neurol 522:2756–2766
Acknowledgements
This work was financially supported by the CNRS, UPS, Agence Nationale de la Recherche (ANR SIMI7 GRAL-12-BS07-0011, ANR-14-CE13-0029-03 ThalaMe). P. P. was supported by a doctoral fellowship from the Région Midi-Pyrénées (#13050241). T. A. was supported by a fellowship from the Ministry of Research, Technology and Higher Education, Republic of Indonesia. We thank Hélène Halley, Stéphane Pech and Brice Ronsin at Toulouse University III for their technical support. We thank Salah El Mestikawy for the gift of VGLUT1 antibody and Marie-Christine Miquel for her help with VGLUT1 immunohistochemistry, Pascal Roullet for advice on behavioral procedures. We also thank the ABC facility from ANEXPLO, Toulouse, for housing mice and Toulouse Réseau Imagerie, FRBT-CBI, for the microscopic platform. Finally, we thank Marie-Christine Miquel, Laure Verret and Lionel Mouledous for helpful comments on the manuscript.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
The authors declare that they have no conflict of interest.
Electronic supplementary material
Below is the link to the electronic supplementary material.
429_2016_1359_MOESM1_ESM.tif
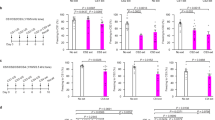
Overview of the experimental groups and paradigms used in this study. Experimental time lines to evaluate the impact of learning (a), consolidation (b), memory retrieval (c, d), novelty (e, f), and extinction (g). (HC: home cage; CE: context exposure; CFC: contextual fear conditioning; A: familiar context A; B: novel context B; EXT: extinction; No-EXT: no extinction, sac: sacrifice) (TIF 22219 KB)
Rights and permissions
About this article
Cite this article
Petsophonsakul, P., Richetin, K., Andraini, T. et al. Memory formation orchestrates the wiring of adult-born hippocampal neurons into brain circuits. Brain Struct Funct 222, 2585–2601 (2017). https://doi.org/10.1007/s00429-016-1359-x
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00429-016-1359-x