Abstract
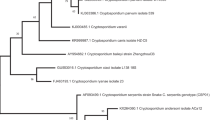
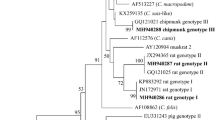
The purpose of this study was to genetically characterize and phylogenetically analyze the Cryptosporidium spp. isolated from exotic birds commercialized in popular markets, commercial aviaries, and pet shops located in Rio de Janeiro, Brazil. Fecal samples from individually housed birds were collected and subjected to centrifuge–flotation technique using saturated sugar solution. DNA was isolated from Cryptosporidium positive samples, and 18S subunit rDNA was amplified and processed using nested-polymerase chain reaction (PCR). To identify the protozoan species, the PCR amplicons were used for restriction fragment length polymorphism and sequencing analyses. Of the 103 analyzed fecal samples, seven (6.8%) were positive for Cryptosporidium oocysts. Sequencing and further phylogenetic analyses allowed us to identify the following species: Cryptosporidium parvum in Bengalese finch (Lonchura striata domestica) and avian genotype III in Java sparrow (Padda oryzivora) and cockatiel (Nymphicus hollandicus). The sequences of the Cryptosporidium spp. isolated from canaries (Serinus canarius) were not identifiable within the groups of known species, but they presented a higher genetic similarity with C. parvum. This is the first report in Brazil showing that C. parvum parasitizes Bengalese finches and that avian genotype III parasitizes Java sparrows.

Similar content being viewed by others
References
Antunes RG, Simões DC, Nakamura AA, Meireles MV (2008) Natural infection with Cryptosporidium galli in canaries (Serinus canaria), in a cockatiel (N. hollandicus), and in lesser seed-finches (Oryzoborus angolensis) from Brazil. Avian Dis 52:702–705. doi:10.1637/8356-051208-Case.1
Current WL, Upton SJ, Haynes TB (1986) The life cycle of Cryptosporidium baileyi, n. sp. (Apicomplexa: Cryptosporiidae) infecting chickens. J Protozool 33:289
Graczyk TK, Cranfield MR, Fayer R, Anderson MS (1996) Viability and infectivity of Cryptosporidium parvum oocysts are retained upon intestinal passage though a refractory avian host. Appl Environ Microbiol 62:3234–3237
Graczyk TK, Fayer R, Trout JM, Lewis EJ, Farley CA, Sulaiman I, Lal Altaf AA (1998) Cysts Giardia and infections Cryptosporidium parvum oocysts in the feces of migratory Canada geese (Branta canadensis). Appl Environ Microbiol 64:2736–2738
Huber F, Bomfim TCB, Gomes RS (2005) Comparison between natural infection by Cryptosporidium sp., Giardia sp. in dogs in two living situations in the west zone of the city of Rio de Janeiro. Vet Parasitol 130:69–72. doi:10.1016/j.vetpar.2005.03.012
Huber F, Da Silva S, Bomfim TCB, Teixeira KRS, Bello AR (2007) Genotypic characterization and phylogenetic analysis of Cryptosporidium sp. from domestic animals in Brazil. Vet Parasitol 150:65–74. doi:10.1016/j.vetpar.2007.08.018
Jellison KL, Distel DL, Hemond HF, Schauer DB (2004) Phylogenetic analysis of the hypervariable region of the 18S r RNA gene of Cryptosporidium oocysts in feces of Canada geese (Branta canadensis): evidence for five novel genotypes. Appl Environ Microbiol 70:452–458. doi:10.1128/AEM.70.1.452-458.2004
Kim Y, Howert EW, Shin NS, Knon SW, Terrell SP, Kim DY (2005) Disseminated visceral coccidiosis and cloacal cryptosporidioseis in a Japanese white-naped crane (Grus vipio). J Parasitol 91:199–201. doi:10.1645/GE-378R
Kuhn RC, Rock CM, Oshima KH (2002) Occurrence of Cryptosporidium and Giardia in wild ducks along the Rio Grande River Valley in Southern New Mexico. Appl Environ Microbiol 68:161–165. doi:10.1128/AEM.68.1.161-165.2002
Kwon YK, Wee SH, Kook JH, Lee CG (2005) Outbreak of enteric cryptosporidiosis in cockatiels (N. hollandicus). Vet Res 12:210–211. doi:10.1136/vr.156.7.210
Latimer KS, Steffens IWL, Rakich PM, Richie BW, Niagro FD, Kircher LM, Lukert PD (1992) Cryptosporidiosis in four cockatoos with psittacine beak and feather disease. J Am Vet Med Ass 200:707–710
Lindsay DS, Blagburn BL, Sundermann CA (1986) Host specificity of Cryptosporidium sp. isolated from chickens. J Parasitol 72:565–568
Lindsay DS, Blagburn BL, Hoerr FJ (1990) Small intestinal cryptosporidiosis in cockatiels associated with Cryptosporidium baileyi-like oocysts. Avian Dis 34:791–793. doi:10.2307/1591283
Meireles MV, Soares RM, Dos Santos MM, Gennari SM (2006) Biological studies and molecular characterization of a Cryptosporidium isolate from ostriches (Struthio camelus). J Parasitol 92:623–626. doi:10.1645/002395(2006)92[623:BSAMCO]2.0.CO
Morgan UM, Thompson RCA (1998) PCR Detection of Cryptosporidium: the way forward? Parasitol Today 14:241–245. doi:10.1016/S0169-4758(98)01247-2
Morgan RUM, Monis P, Possenti A, Crisanti A, Spano F (2001) Cloning and phylogenetic analysis of the ribosomal internal transcribed spacer-1 (ITS1) of Cryptosporidium wrairi and its relationship to C. parvum genotypes. Parasitol 43:159–163. doi:10.1016/S0020-7519(00)00164-8
Nakamura AA, Simões DC, Antunes RG, Silva Da DC, Meireles MV (2009) Molecular characterization of Cryptosporidium spp. from fecal samples of birds kept in captivity in Brazil. Vet Parasitol 166:47–51. doi:10.1016/j.vetpar.2009.07.033
Ng J, Pavlasek I, Ryan U (2006) Identification of novel Cryptosporidium genotypes from avian hosts. Appl Environ Microbiol 72:548–553. doi:10.1128/AEM.01352-06
Palkovic L, Marousek V (1989) The pathogenicity of Cryptosporidium parvum Tyzzer, 1912 and C. baileyi Current, Upton et Haynes, 1986 for chickens. Folia Parasitol 36:209–217
Ryan U (2010) Cryptosporidium in birds, fish and amphibians. Exp Parasitol 124:113–120. doi:10.1016/j.exppara.2009.02.002
Ryan UM, Xiao L, Read C, Sulaiman IM, Monis P, LAL A, Fayer R, Pavlasek I (2003) A redescription of Cryptosporidium galli, Pavlasek, 1999 (Apicomplexa: Cryptosporidiidae) from birds. J Parasitol 9:809–813. doi:10.1016/j.vetpar.2010.09.031
Ryan UM, O'hara A, Xiu O (2004) Molecular and biological characterization of a Cryptosporidium molnari-like isolate from a guppy (Poecilia reticulata). Appl Environ Microbiol 70:3761–3765. doi:10.1128/AEM.70.6.3761-3765.2004
Sevá AP, Funada MR, Richtzenhain L, Gimarães MB, Souza SO, Allegretti L, Sinhorini JA, Duarte VV, Soares RM (2011) Genotyping of Cryptosporidium spp. free-living wild birds from Brazil. Vet Parasitol 37:27–32. doi:10.1016/j.vetpar.2010.09.031
Slavin D (1955) Cryptosporidium meleagridis. J Comp Pathol 65:262–266
Sréter T, Varga I (2000) Cryptosporidiosis in birds—a review. Vet Parasitol 87:261–279. doi:10.1016/S0304-4017(99)00178-8
Xiao L, Sulaiman M, Ryan UM, Zhou L, Atwill ER, Tischler ML, Zhang X, Fayer R, Lal LL (2002) Host adaptation and host–parasite co-evolution in Cryptosporidium: implications for taxonomy and public health. Int J Parasitol 32:1773–1785. doi:10.1016/S0020-7519(02)00197-2
Xiao L, Altaf AL, Jiang J (2004) Detection and differentiation of Cryptosporidium oocysts in water by PCR-RFLP. In: Spencer JFT, Spencer ALR (eds) Public health microbiology: methods and protocols. Humana Press, Totowana, pp 163–176
Zhou L, Kassa H, Tiscler L, Xiao L (2004) Host-adapted Cryptosporidium spp. in Canada geese (Branta canadensis). Appl Environ Microbiol 70:4211–4215. doi:10.1128/AEM.70.7.4211-4215.2004
Zylan K, Bailey T, Smith HV, Silvanose C, Kinne J, Schuster RK, Hyland K (2008) An outbreak of cryptosporidiosis in a collection of stone curlews (Burhinus oedicnemus) in Dubai. Avian Pathol 37:521–526. doi:10.1080/03079450802357019
Acknowledgments
We thank the Conselho Nacional de Desenvolvimento Científico e Tecnológico (CNPq) and the Fundação de Amparo a Pesquisa do Estado do Rio de Janeiro (FAPERJ).
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Gomes, R.S., Huber, F., da Silva, S. et al. Cryptosporidium spp. parasitize exotic birds that are commercialized in markets, commercial aviaries, and pet shops. Parasitol Res 110, 1363–1370 (2012). https://doi.org/10.1007/s00436-011-2636-5
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00436-011-2636-5