Abstract
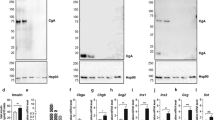
Chromogranin A (CgA) is a prohormone and granulogenic factor in neuroendocrine tissues with a regulated secretory pathway. The impact of CgA depletion on secretory granule formation has been previously demonstrated in cell culture. However, studies linking the structural effects of CgA deficiency with secretory performance and cell metabolism in the adrenomedullary chromaffin cells in vivo have not previously been reported. Adrenomedullary content of the secreted adrenal catecholamines norepinephrine (NE) and epinephrine (EPI) was decreased 30–40 % in Chga-KO mice. Quantification of NE and EPI-storing dense core (DC) vesicles (DCV) revealed decreased DCV numbers in chromaffin cells in Chga-KO mice. For both cell types, the DCV diameter in Chga-KO mice was less (100–200 nm) than in WT mice (200–350 nm). The volume density of the vesicle and vesicle number was also lower in Chga-KO mice. Chga-KO mice showed an ~47 % increase in DCV/DC ratio, implying vesicle swelling due to increased osmotically active free catecholamines. Upon challenge with 2 U/kg insulin, there was a diminution in adrenomedullary EPI, no change in NE and a very large increase in the EPI and NE precursor dopamine (DA), consistent with increased catecholamine biosynthesis during prolonged secretion. We found dilated mitochondrial cristae, endoplasmic reticulum and Golgi complex, as well as increased synaptic mitochondria, synaptic vesicles and glycogen granules in Chga-KO mice compared to WT mice, suggesting that decreased granulogenesis and catecholamine storage in CgA-deficient mouse adrenal medulla is compensated by increased VMAT-dependent catecholamine update into storage vesicles, at the expense of enhanced energy expenditure by the chromaffin cell.









Similar content being viewed by others
Abbreviations
- CA:
-
Catecholamines
- CgA:
-
Chromogranin A protein
- Chga:
-
Mouse Chromogranin A gene
- DA:
-
Dopamine
- DC:
-
Dense core
- DCV:
-
DC vesicle
- EPI:
-
Epinephrine
- GC:
-
Golgi complex
- KO:
-
Knockout
- NE:
-
Norepinephrine
- SDCV:
-
Decreased DC with swelled halo
- SEV:
-
Swelled empty vesicle
- SV:
-
Synaptic vesicle
- TEM:
-
Transmission electron microscopy
- VMAT:
-
Vesicular monoamine transporter
- WT:
-
Wild-type
References
Aardal S, Helle KB, Elsayed S, Reed RK, Serck-Hanssen G (1993) Vasostatins, comprising the N-terminal domain of chromogranin A, suppress tension in isolated human blood vessel segments. J Neuroendocrinol 5:405–412
Angelone T, Quintieri AM, Brar BK, Limchaiyawat PT, Tota B, Mahata SK, Cerra MC (2008) The antihypertensive chromogranin a peptide catestatin acts as a novel endocrine/paracrine modulator of cardiac inotropism and lusitropism. Endocrinology 149:4780–4793
Arruda AP, Pers BM, Parlakgul G, Guney E, Inouye K, Hotamisligil GS (2014) Chronic enrichment of hepatic endoplasmic reticulum-mitochondria contact leads to mitochondrial dysfunction in obesity. Nat Med 20:1427–1435
Bandyopadhyay GK, Lu M, Avolio E, Siddiqui JA, Gayen JR, Wollam J, Vu CU, Chi NW, O'Connor DT, Mahata SK (2015) Pancreastatin-dependent inflammatory signaling mediates obesity-induced insulin resistance. Diabetes 64:104–116
Bandyopadhyay GK, Vu CU, Gentile S, Lee H, Biswas N, Chi NW, O'Connor DT, Mahata SK (2012) Catestatin (chromogranin A(352–372)) and novel effects on mobilization of fat from adipose tissue through regulation of adrenergic and leptin signaling. J Biol Chem 287:23141–23151
Bartolomucci A, Possenti R, Mahata SK, Fischer-Colbrie R, Loh YP, Salton SR (2011) The extended granin family: structure, function, and biomedical implications. Endocr Rev 32:755–797
Berneis KH, Goetz U, Da Prada M, Pletscher A (1973) Interaction of aggregated catecholamines and nucleotides with intragranular proteins. Naunyn Schmiedebergs Arch Pharmacol 277:291–296
Borges R, Diaz-Vera J, Dominguez N, Arnau MR, Machado JD (2010) Chromogranins as regulators of exocytosis. J Neurochem 114:335–343
Borkowski KR, Quinn P (1985) Adrenaline and the development of spontaneous hypertension in rats. J Auton Pharmacol 5:89–100
Brodwick MS, Curran M, Edwards C (1992) Effects of osmotic stress on mast cell vesicles of the beige mouse. J Membr Biol 126:159–169
Chanat E, Huttner WB (1991) Milieu-induced, selective aggregation of regulated secretory proteins in the trans-Golgi network. J Cell Biol 115:1505–1519
Cheville NF (2013) Ultrastructural pathology and interorganelle cross talk in hepatotoxicity. Toxicol Pathol 41:210–226
Colliver TL, Pyott SJ, Achalabun M, Ewing AG (2000) VMAT-Mediated changes in quantal size and vesicular volume. J Neurosci 20:5276–5282
Coupland RE (1965) Electron microscopic observations on the structure of the rat adrenal medulla: II. Normal innervation. J Anat 99:255–272
Coupland RE, Hopwood D (1966) Mechanism of a histochemical reaction differentiating between adrenaline- and noradrenaline-storing cells in the electron microscope. Nature 209:590–591
Crosson SM, Khan A, Printen J, Pessin JE, Saltiel AR (2003) PTG gene deletion causes impaired glycogen synthesis and developmental insulin resistance. J Clin Invest 111:1423–1432
David G, Barrett EF (2003) Mitochondrial Ca2+ uptake prevents desynchronization of quantal release and minimizes depletion during repetitive stimulation of mouse motor nerve terminals. J Physiol 548:425–438
de Champlain J, Karas M, Toal C, Nadeau R, Larochelle P (1999) Effects of antihypertensive therapies on the sympathetic nervous system. Can J Cardiol 15(Suppl A):8A–14A
Dev NB, Gayen JR, O'Connor DT, Mahata SK (2010) Chromogranin A and the autonomic system: Decomposition of heart rate variability by time and frequency domains, along with non-linear characteristics during chromogranin A ablation, with "rescue" by its catestatin. Endocrinology 151:2760–2768
Diaz-Vera J, Camacho M, Machado JD, Dominguez N, Montesinos MS, Hernandez-Fernaud JR, Lujan R, Borges R (2012) Chromogranins A and B are key proteins in amine accumulation, but the catecholamine secretory pathway is conserved without them. FASEB J 26:430–438
Eirin A, Lerman A, Lerman LO (2015) Mitochondria: a pathogenic paradigm in hypertensive renal disease. Hypertension 65:264–270
Elias S, Delestre C, Ory S, Marais S, Courel M, Vazquez-Martinez R, Bernard S, Coquet L, Malagon MM, Driouich A, Chan P, Gasman S, Anouar Y, Montero-Hadjadje M (2012) Chromogranin A induces the biogenesis of granules with calcium- and actin-dependent dynamics and exocytosis in constitutively secreting cells. Endocrinology 153:4444–4456
Fargali S, Garcia AL, Sadahiro M, Jiang C, Janssen WG, Lin WJ, Cogliani V, Elste A, Mortillo S, Cero C, Veitenheimer B, Graiani G, Pasinetti GM, Mahata SK, Osborn JW, Huntley GW, Phillips GR, Benson DL, Bartolomucci A, Salton SR (2014) The granin VGF promotes genesis of secretory vesicles, and regulates circulating catecholamine levels and blood pressure. FASEB J 28:2120–2133
Fernandez-Busnadiego R, Asano S, Oprisoreanu AM, Sakata E, Doengi M, Kochovski Z, Zurner M, Stein V, Schoch S, Baumeister W, Lucic V (2013) Cryo-electron tomography reveals a critical role of RIM1alpha in synaptic vesicle tethering. J Cell Biol 201:725–740
Gayen JR, Gu Y, O’Connor DT, Mahata SK (2009a) Global disturbances in autonomic function yield cardiovascular instability and hypertension in the chromogranin A null mouse. Endocrinology 150:5027–5035
Gayen JR, Saberi M, Schenk S, Biswas N, Vaingankar SM, Cheung WW, Najjar SM, O'Connor DT, Bandyopadhyay G, Mahata SK (2009b) A novel pathway of insulin sensitivity in chromogranin a null mice: A crucial role for pancreastatin in glucose homeostasis. J Biol Chem 284:28498–28509
Gayen JR, Zhang K, Ramachandrarao SP, Mahata M, Chen Y, Kim H-S, Naviaux RK, Sharma K, Mahata SK, O'Connor DT (2010) Role of reactive oxygen species in hyperadrenergic hypertension: Biochemical, physiological, and pharmacological evidence from targeted ablation of the chromogranin A gene. Circ Cardiovasc Genet 3:414–425
Goldstein DS (1983) Plasma catecholamines and essential hypertension. An analytical review. Hypertension 5:86–99
Gong LW, Hafez I, Alvarez de Toledo G, Lindau M (2003) Secretory vesicles membrane area is regulated in tandem with quantal size in chromaffin cells. J Neurosci 23:7917–7921
Grabner CP, Fox AP (2006) Stimulus-dependent alterations in quantal neurotransmitter release. J Neurophysiol 96:3082–3087
Guo X, Macleod GT, Wellington A, Hu F, Panchumarthi S, Schoenfield M, Marin L, Charlton MP, Atwood HL, Zinsmaier KE (2005) The GTPase dMiro is required for axonal transport of mitochondria to Drosophila synapses. Neuron 47:379–393
Guo Y, Darshi M, Ma Y, Perkins GA, Shen Z, Haushalter KJ, Saito R, Chen A, Lee YS, Patel HH, Briggs SP, Ellisman MH, Olefsky JM, Taylor SS (2013) Quantitative proteomic and functional analysis of liver mitochondria from high fat diet (HFD) diabetic mice. Mol Cell Proteomics 12:3744–3758
Haigh JR, Parris R, Phillips JH (1989) Free concentrations of sodium, potassium and calcium in chromaffin granules. Biochem J 259:485–491
Hamelink C, Tjurmina O, Damadzic R, Young WS, Weihe E, Lee HW, Eiden LE (2002) Pituitary adenylate cyclase-activating polypeptide is a sympathoadrenal neurotransmitter involved in catecholamine regulation and glucohomeostasis. Proc Natl Acad Sci U S A 99:461–466
Helle KB, Reed RK, Pihl KE, Serck-Hanssen G (1985) Osmotic properties of the chromogranins and relation to osmotic pressure in catecholamine storage granules. Acta Physiol Scand 123:21–33
Hendy GN, Li T, Girard M, Feldstein RC, Mulay S, Desjardins R, Day R, Karaplis AC, Tremblay ML, Canaff L (2006) Targeted ablation of the chromogranin a (Chga) gene: normal neuroendocrine dense-core secretory granules and increased expression of other granins. Mol Endocrinol 20:1935–1947
Henry JP, Botton D, Sagne C, Isambert MF, Desnos C, Blanchard V, Raisman-Vozari R, Krejci E, Massoulie J, Gasnier B (1994) Biochemistry and molecular biology of the vesicular monoamine transporter from chromaffin granules. J Exp Biol 196:251–262
Hosaka M, Suda M, Sakai Y, Izumi T, Watanabe T, Takeuchi T (2004) Secretogranin III binds to cholesterol in the secretory granule membrane as an adapter for chromogranin A. J Biol Chem 279:3627–3634
Hosaka M, Watanabe T (2010) Secretogranin III: a bridge between core hormone aggregates and the secretory granule membrane. Endocr J 57:275–286
Huang JB, Ding Y, Huang DS, Liang AJ, Zeng WK, Zeng ZP, Qin CQ, Barden B (2013) Inhibition of the PI3K/AKT pathway reduces tumor necrosis factor-alpha production in the cellular response to wear particles in vitro. Artif Organs 37:298–307
Iacangelo AL, Eiden LE (1995) Chromogranin A: current status as a precursor for bioactive peptides and a granulogenic/sorting factor in the regulated secretory pathway. Regul Pept 58:65–88
Jiang Z, Hu Z, Zeng L, Lu W, Zhang H, Li T, Xiao H (2011) The role of the Golgi apparatus in oxidative stress: is this organelle less significant than mitochondria? Free Radic Biol Med 50:907–917
Johnson RG Jr (1988) Accumulation of biological amines into chromaffin granules: a model for hormone and neurotransmitter transport. Physiol Rev 68:232–307
Kim T, Tao-Cheng J, Eiden LE, Loh YP (2001) Chromogranin A, an "On/Off" Switch Controlling Dense-Core Secretory Granule Biogenesis. Cell 106:499–509
Kim T, Zhang CF, Sun Z, Wu H, Loh YP (2005) Chromogranin A deficiency in transgenic mice leads to aberrant chromaffin granule biogenesis. J Neurosci 25:6958–6961
Kirsch DM, Baumgarten M, Deufel T, Rinninger F, Kemmler W, Haring HU (1983) Catecholamine-induced insulin resistance of glucose transport in isolated rat adipocytes. Biochem J 216:737–745
Kopell WN, Westhead EW (1982) Osmotic pressures of solutions of ATP and catecholamines relating to storage in chromaffin granules. J Biol Chem 257:5707–5710
Ma H, Cai Q, Lu W, Sheng ZH, Mochida S (2009) KIF5B motor adaptor syntabulin maintains synaptic transmission in sympathetic neurons. J Neurosci 29:13019–13029
Mahapatra NR, Mahata M, Hazra PP, McDonough PM, O'Connor DT, Mahata SK (2004) A dynamic pool of calcium in catecholamine storage vesicles: exploration in living cells by a novel vesicle-targeted chromogranin A/aequorin chimeric photoprotein. J Biol Chem 279:51107–51121
Mahapatra NR, O'Connor DT, Vaingankar SM, Hikim AP, Mahata M, Ray S, Staite E, Wu H, Gu Y, Dalton N, Kennedy BP, Ziegler MG, Ross J, Mahata SK (2005) Hypertension from targeted ablation of chromogranin A can be rescued by the human ortholog. J Clin Invest 115:1942–1952
Mahata SK, Mahapatra NR, Mahata M, Wang TC, Kennedy BP, Ziegler MG, O'Connor DT (2003) Catecholamine secretory vesicle stimulus-transcription coupling in vivo. Demonstration by a novel transgenic promoter/photoprotein reporter and inhibition of secretion and transcription by the chromogranin A fragment catestatin. J Biol Chem 278:32058–32067
Mahata SK, Mahata M, Fung MM, O'Connor DT (2010) Catestatin: a multifunctional peptide from chromogranin A. Regul Pept 162:33–43
Mahata SK, O'Connor DT, Mahata M, Yoo SH, Taupenot L, Wu H, Gill BM, Parmer RJ (1997) Novel autocrine feedback control of catecholamine release. A discrete chromogranin A fragment is a noncompetitive nicotinic cholinergic antagonist. J Clin Invest 100:1623–1633
Medler K, Gleason EL (2002) Mitochondrial Ca(2+) buffering regulates synaptic transmission between retinal amacrine cells. J Neurophysiol 87:1426–1439
Montesinos MS, Machado JD, Camacho M, Diaz J, Morales YG, Alvarez de la Rosa D, Carmona E, Castaneyra A, Viveros OH, O'Connor DT, Mahata SK, Borges R (2008) The crucial role of chromogranins in storage and exocytosis revealed using chromaffin cells from chromogranin A null mouse. J Neurosci 28:3350–3358
Mulder AH, Tack CJ, Olthaar AJ, Smits P, Sweep FC, Bosch RR (2005) Adrenergic receptor stimulation attenuates insulin-stimulated glucose uptake in 3T3-L1 adipocytes by inhibiting GLUT4 translocation. Am J Physiol Endocrinol Metab 289:E627–E633
Noh YH, Kim KY, Shim MS, Choi SH, Choi S, Ellisman MH, Weinreb RN, Perkins GA, Ju WK (2013) Inhibition of oxidative stress by coenzyme Q10 increases mitochondrial mass and improves bioenergetic function in optic nerve head astrocytes. Cell Death Dis 4:e820
Oakes SA, Papa FR (2015) The role of endoplasmic reticulum stress in human pathology. Annu Rev Pathol 10:173–194
Pacak K, Palkovits M, Yadid G, Kvetnansky R, Kopin IJ, Goldstein DS (1998) Heterogeneous neurochemical responses to different stressors: a test of Selye's doctrine of nonspecificity. Am J Physiol 275:R1247–R1255
Patzak A, Winkler H (1986) Exocytotic exposure and recycling of membrane antigens of chromaffin granules: ultrastructural evaluation after immunolabeling. J Cell Biol 102:510–515
Philp A, Hargreaves M, Baar K (2012) More than a store: regulatory roles for glycogen in skeletal muscle adaptation to exercise. Am J Physiol Endocrinol Metab 302:E1343–E1351
Plattner H, Artalejo AR, Neher E (1997) Ultrastructural organization of bovine chromaffin cell cortex-analysis by cryofixation and morphometry of aspects pertinent to exocytosis. J Cell Biol 139:1709–1717
Revel JP, Napolitano L, Fawcett DW (1960) Identification of glycogen in electron micrographs of thin tissue sections. J Biophys Biochem Cytol 8:575–589
Roach PJ, Depaoli-Roach AA, Hurley TD, Tagliabracci VS (2012) Glycogen and its metabolism: some new developments and old themes. Biochem J 441:763–787
Sanchez-Margalet V, Gonzalez-Yanes C, Najib S, Santos-Alvarez J (2010) Metabolic effects and mechanism of action of the chromogranin A-derived peptide pancreastatin. Regul Pept 161:8–14
Schuldiner S (1994) A molecular glimpse of vesicular monoamine transporters. J Neurochem 62:2067–2078
Sengupta D, Linstedt AD (2011) Control of organelle size: the Golgi complex. Annu Rev Cell Dev Biol 27:57–77
Sterling CR, Tank AW (2001) Adrenal tyrosine hydroxylase activity and gene expression are increased by intraventricular administration of nicotine. J Pharmacol Exp Ther 296:15–21
Stowers RS, Megeath LJ, Gorska-Andrzejak J, Meinertzhagen IA, Schwarz TL (2002) Axonal transport of mitochondria to synapses depends on milton, a novel Drosophila protein. Neuron 36:1063–1077
Sudhof TC (1982) Core structure, internal osmotic pressure and irreversible structural changes of chromaffin granules during osmometer behaviour. Biochim Biophys Acta 684:27–39
Tatemoto K, Efendic S, Mutt V, Makk G, Feistner GJ, Barchas JD (1986) Pancreastatin, a novel pancreatic peptide that inhibits insulin secretion. Nature 324:476–478
Taupenot L, Harper KL, Mahapatra NR, Parmer RJ, Mahata SK, O'Connor DT (2002) Identification of a novel sorting determinant for the regulated pathway in the secretory protein chromogranin A. J Cell Sci 115:4827–4841
Theurl M, Schgoer W, Albrecht K, Jeschke J, Egger M, Beer AG, Vasiljevic D, Rong S, Wolf AM, Bahlmann FH, Patsch JR, Wolf D, Schratzberger P, Mahata SK, Kirchmair R (2010) The neuropeptide catestatin acts as a novel angiogenic cytokine via a basic fibroblast growth factor-dependent mechanism. Circ Res 107:1326–1335
Thoenen H, Mueller RA, Axelrod J (1969) Increased tyrosine hydroxylase activity after drug-induced alteration of sympathetic transmission. Nature 221:1264
Tota B, Angelone T, Mazza R, Cerra MC (2008) The chromogranin A-derived vasostatins: new players in the endocrine heart. Curr Med Chem 15:1444–1451
Tota B, Gentile S, Pasqua T, Bassino E, Koshimizu H, Cawley NX, Cerra MC, Loh YP, Angelone T (2012) The novel chromogranin A-derived serpinin and pyroglutaminated serpinin peptides are positive cardiac beta-adrenergic-like inotropes. FASEB J 26:2888–2898
Tsuda K, Masuyama Y (1991) Presynaptic regulation of neurotransmitter release in hypertension. Clin Exp Pharmacol Physiol 18:455–467
Videen JS, Mezger MS, Chang YM, O'Connor DT (1992) Calcium and catecholamine interactions with adrenal chromogranins. Comparison of driving forces in binding and aggregation. J Biol Chem 267:3066–3073
Wakade AR, Wakade TD, Malhotra RK (1988) Restoration of catecholamine content of previously depleted adrenal medulla in vitro: importance of synthesis in maintaining the catecholamine stores. J Neurochem 51:820–829
Westfall TC, Meldrum MJ (1985) Alterations in the release of norepinephrine at the vascular neuroeffector junction in hypertension. Annu Rev Pharmacol Toxicol 25:621–641
Winkler H, Fischer-Colbrie R (1992) The chromogranins A and B: the first 25 years and future perspectives. Neuroscience 49:497–528
Winkler H, Sietzen M, Schober M (1987) The life cycle of catecholamine-storing vesicles. Ann N Y Acad Sci 493:3–19
Winkler H, Westhead E (1980) The molecular organization of adrenal chromaffin granules. Neuroscience 5:1803–1823
Yoo SH, Albanesi JP (1991) High capacity, low affinity Ca2+ binding of chromogranin A. Relationship between the pH-induced conformational change and Ca2+ binding property. J Biol Chem 266:7740–7745
Acknowledgments
Transmission Electron Microscopy was conducted at the Cellular & Molecular Medicine Electron Microscopy Core Facility at UCSD. A Research Career Scientist Award from the Department of Veterans Affairs supports S.K.M. Mahata’s personal fund and UCSD Academic Senate Grant (RO091B) supported this work.
Conflict of interest
The authors have nothing to declare.
Author information
Authors and Affiliations
Corresponding author
Additional information
Teresa Pasqua and Sumana Mahata contributed equally to this work.
Rights and permissions
About this article
Cite this article
Pasqua, T., Mahata, S., Bandyopadhyay, G.K. et al. Impact of Chromogranin A deficiency on catecholamine storage, catecholamine granule morphology and chromaffin cell energy metabolism in vivo. Cell Tissue Res 363, 693–712 (2016). https://doi.org/10.1007/s00441-015-2316-3
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00441-015-2316-3