Abstract
Purpose
Fatigue is a common problem among rectal cancer patients and can affect their quality of life. This study conducted a systematic review to better understand changes in fatigue severity in rectal cancer patients before, during, and after they undergo therapy.
Methods
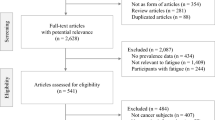
We used preset keywords to search the Cochrane Library, the Cumulative Index to Nursing and Allied Health Literature (CINAHL), PubMed, and ProQuest databases for relevant studies published between 2000 and 2018, and data analysis was performed using Comprehensive Meta-Analysis (CMA) software (version 2.2.048) and SPSS software (version 19.0). In total, nine articles with complete data were included in our meta-analysis.
Results
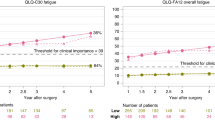
Fatigue conditions were compared before the start of therapy (baseline) and at 1 month (time 1), 3 months (time 2), 6 months (time 3), and 12 months (time 4) after the start of therapy. The standardized mean differences (SMDs) of the pooling effects size were 1.013 (95% confidence interval (CI) 0.217–1.810), − 0.551 (95% CI − 0.647 to − 0.456), − 0.330 (95% CI − 0.427 to − 0.233), and − 0.149 (95% CI − 0.221 to − 0.078), respectively. Subsequent analysis with a linear mixed effect model revealed that the estimate of the time variable was − 0.226 (p = 0.047), which indicates that the severity of fatigue varies over time and over the course of treatment. The results reveal that fatigue affects rectal cancer patients even before they start therapy.
Conclusion
Although fatigue worsened during the first month after cancer therapy, it gradually improved thereafter.




Similar content being viewed by others
References
Curt GA, Breitbart W, Cella D, Groopman JE, Horning SJ, Itri LM, Johnson DH, Miaskowski C, Scherr SL, Portenoy RK, Vogelzang NJ (2000) Impact of cancer-related fatigue on the lives of patients: new findings from the fatigue coalition. Oncologist 5:353–360. https://doi.org/10.1634/theoncologist.5-5-353
Teunissen SC, Wesker W, Kruitwagen C, de Haes HC, Voest EE, de Graeff A (2007) Symptom prevalence in patients with incurable cancer: a systematic review. J Pain Symptom Manag 34:94–104. https://doi.org/10.1016/j.jpainsymman.2006.10.015
Charalambous A, Kouta C (2016) Cancer related fatigue and quality of life in patients with advanced prostate cancer undergoing chemotherapy. Biomed Res Int 2016:3989286. https://doi.org/10.1155/2016/3989286
Chen HL, Liu K, You QS (2018) Self-efficacy, cancer-related fatigue, and quality of life in patients with resected lung cancer. Eur J Cancer Care (Engl) 27:e12934. https://doi.org/10.1111/ecc.12934
Fabi A, Falcicchio C, Giannarelli D, Maggi G, Cognetti F, Pugliese P (2017) The course of cancer related fatigue up to ten years in early breast cancer patients: what impact in clinical practice? Breast 34:44–52. https://doi.org/10.1016/j.breast.2017.04.012
Grenon NN, Chan J (2009) Managing toxicities associated with colorectal cancer chemotherapy and targeted therapy: a new guide for nurses. Clin J Oncol Nurs 13:285–296. https://doi.org/10.1188/09.CJON.285-296
Wang XS, Janjan NA, Guo H, Johnson BA, Engstrom MC, Crane CH, Mendoza TR, Cleeland CS (2001) Fatigue during preoperative chemoradiation for resectable rectal cancer. Cancer 92:1725–1732. https://doi.org/10.1002/1097-0142(20010915)92:6+<1725::aid-cncr1504>3.0.co;2-d
Yeom SS, Park IJ, Jung SW, Oh SH, Lee JL, Yoon YS, Kim CW, Lim SB, Kim N, Yu CS, Kim JC (2017) Outcomes of patients with abdominoperineal resection (APR) and low anterior resection (LAR) who had very low rectal cancer. Medicine 96:e8249. https://doi.org/10.1097/MD.0000000000008249
Li Y, Wang J, Ma X, Tan L, Yan Y, Xue C, Hui B, Liu R, Ma H, Ren J (2016) A review of neoadjuvant chemoradiotherapy for locally advanced rectal cancer. Int J Biol Sci 12:1022–1031. https://doi.org/10.7150/ijbs.15438
Petersen SH, Harling H, Kirkeby LT, Wille-Jørgensen P, Mocellin S (2012) Postoperative adjuvant chemotherapy in rectal cancer operated for cure. Cochrane Database Syst Rev 3:CD004078. https://doi.org/10.1002/14651858.CD004078.pub2
Kim CG, Ahn JB, Shin SJ, Beom SH, Heo SJ, Park HS, Kim JH, Choe EA, Koom WS, Hur H, Min BS, Kim NK, Kim H, Kim C, Jung I, Jung M (2017) Role of adjuvant chemotherapy in locally advanced rectal cancer with ypT0-3N0 after preoperative chemoradiation therapy and surgery. BMC Cancer 17:615. https://doi.org/10.1186/s12885-017-3624-7
Joanna Briggs Institute (2011) Module 3-the appraisal, extraction and pooling of quantitative data. http://www.joannabriggs.edu.au.pdf. Accessed 30 August 2019
Li SX, Liu BB, Lu JH (2014) Longitudinal study of cancer-related fatigue in patients with colorectal cancer. Asian Pac J Cancer Prev 15:3029–3033. https://doi.org/10.7314/apjcp.2014.15.7.3029
Park HC, Janjan NA, Mendoza TR, Lin EH, Vadhan-Raj S, Hundal M, Zhang Y, Delclos ME, Crane CH, Das P, Wang XS, Cleeland CS, Krishnan S (2009) Temporal patterns of fatigue predict pathologic response in patients treated with preoperative chemoradiation therapy for rectal cancer. Int J Radiat Oncol Biol Phys 75:775–781. https://doi.org/10.1016/j.ijrobp.2008.11.027
Pucciarelli S, Del Bianco P, Efficace F et al (2011) Patient-reported outcomes after neoadjuvant chemoradiotherapy for rectal cancer: a multicenter prospective observational study. Ann Surg 253:71–77. https://doi.org/10.1097/SLA.0b013e3181fcb856
Couwenberg AM, Burbach JPM, van Grevenstein WMU, Smits AB, Consten ECJ, Schiphorst AHW, Wijffels NAT, Heikens JT, Intven MPW, Verkooijen HM (2018) Effect of neoadjuvant therapy and rectal surgery on health-related quality of life in patients with rectal cancer during the first 2 years after diagnosis. Clin Colorectal Cancer 17:e499–e512. https://doi.org/10.1016/j.clcc.2018.03.009
Schmidt CE, Bestmann B, Küchler T, Longo WE, Kremer B (2005) Prospective evaluation of quality of life of patients receiving either abdominoperineal resection or sphincter-preserving procedure for rectal cancer. Ann Surg Oncol 12:117–123. https://doi.org/10.1245/ASO.2005.12.036
Zhang JK, Fang LL, Zhang DW, Jin Q, Wu XM, Liu JC, Zhang CD, Dai DQ (2016) Type D personality is associated with delaying patients to medical assessment and poor quality of life among rectal cancer survivors. Int J Color Dis 31:75–85. https://doi.org/10.1007/s00384-015-2333-4
Grumann MM, Noack EM, Hoffmann IA, Schlag PM (2001) Comparison of quality of life in patients undergoing abdominoperineal extirpation or anterior resection for rectal cancer. Ann Surg 233:149–156. https://doi.org/10.1097/00000658-200102000-00001
Monastyrska E, Hagner W, Jankowski M, Głowacka I, Zegarska B, Zegarski W (2016) Prospective assessment of the quality of life in patients treated surgically for rectal cancer with lower anterior resection and abdominoperineal resection. Eur J Surg Oncol 42:1647–1653. https://doi.org/10.1016/j.ejso.2016.07.007
Herrle F, Sandra-Petrescu F, Weiss C, Post S, Runkel N, Kienle P (2016) Quality of life and timing of stoma closure in patients with rectal cancer undergoing low anterior resection with diverting stoma: a multicenter longitudinal observational study. Dis Colon Rectum 59:281–290. https://doi.org/10.1097/DCR.0000000000000545
Väyrynen JP, Tuomisto A, Väyrynen SA, Klintrup K, Karhu T, Mäkelä J, Herzig KH, Karttunen TJ, Mäkinen MJ (2018) Preoperative anemia in colorectal cancer: relationships with tumor characteristics, systemic inflammation, and survival. Sci Rep 8:1126. https://doi.org/10.1038/s41598-018-19572-y
Bondurant KL, Lundgreen A, Herrick JS, Kadlubar S, Wolff RK, Slattery ML (2013) Interleukin genes and associations with colon and rectal cancer risk and overall survival. Int J Cancer 132:905–915. https://doi.org/10.1002/ijc.27660
Macciò A, Madeddu C, Gramignano G et al (2015) The role of inflammation, iron, and nutritional status in cancer-related anemia: results of a large, prospective, observational study. Haematologica 100:124–132. https://doi.org/10.3324/haematol.2014.112813
Manocha M, Khan WI (2012) Serotonin and GI disorders: an update on clinical and experimental studies. Clin Transl Gastroenterol 3:e13. https://doi.org/10.1038/ctg.2012.8
Coleman EA, Goodwin JA, Coon SK, Richards K, Enderlin C, Kennedy R, Stewart CB, McNatt P, Lockhart K, Anaissie EJ, Barlogie B (2011) Fatigue, sleep, pain, mood, and performance status in patients with multiple myeloma. Cancer Nurs 34:219–227. https://doi.org/10.1097/NCC.0b013e3181f9904d
Bower JE (2014) Cancer-related fatigue--mechanisms, risk factors, and treatments. Nat Rev Clin Oncol 11:597–609. https://doi.org/10.1038/nrclinonc.2014.127
Johnson RL, Amin AR, Matzo M (2012) Cancer-related fatigue. Am J Nurs 112:57–60. https://doi.org/10.1097/01.NAJ.0000413462.26668.96
O'Higgins CM, Brady B, O'Connor B, Walsh D, Reilly RB (2018) The pathophysiology of cancer-related fatigue: current controversies. Support Care Cancer 26:3353–3364. https://doi.org/10.1007/s00520-018-4318-7
Goedendorp MM, Gielissen MF, Verhagen CA, Peters ME, Bleijenberg G (2008) Severe fatigue and related factors in cancer patients before the initiation of treatment. Br J Cancer 99:1408–1414. https://doi.org/10.1038/sj.bjc.6604739
Silverman MN, Heim CM, Nater UM, Marques AH, Sternberg EM (2010) Neuroendocrine and immune contributors to fatigue. PM R 2:338–346. https://doi.org/10.1016/j.pmrj.2010.04.008
Gil F, Costa G, Hilker I, Benito L (2012) First anxiety, afterwards depression: psychological distress in cancer patients at diagnosis and after medical treatment. Stress Health 28:362–367. https://doi.org/10.1002/smi.2445
Turgay AS, Khorshid L, Eser I (2008) Effect of the first chemotherapy course on the quality of life of cancer patients in Turkey. Cancer Nurs 31:E19–E23. https://doi.org/10.1097/01.NCC.0000339248.37829.c2
Park IJ, You YN, Agarwal A, Skibber JM, Rodriguez-Bigas MA, Eng C, Feig BW, Das P, Krishnan S, Crane CH, Hu CY, Chang GJ (2012) Neoadjuvant treatment response as an early response indicator for patients with rectal cancer. J Clin Oncol 30:1770–1776. https://doi.org/10.1200/JCO.2011.39.7901
Given B, Given CW, McCorkle R, Kozachik S, Cimprich B, Rahbar MH, Wojcik C (2002) Pain and fatigue management: results of a nursing randomized clinical trial. Oncol Nurs Forum 29:949–956. https://doi.org/10.1188/02.ONF.949-956
Saber MM, Al-Mahallawi AM, Nassar NN, Stork B, Shouman SA (2018) Targeting colorectal cancer cell metabolism through development of cisplatin and metformin nano-cubosomes. BMC Cancer 18:822. https://doi.org/10.1186/s12885-018-4727-5
Banipal RPS, Singh H, Singh B (2017) Assessment of cancer-related fatigue among cancer patients receiving various therapies: a cross-sectional observational study. Indian J Palliat Care 23:207–211. https://doi.org/10.4103/IJPC.IJPC_135_16
Hofman M, Ryan JL, Figueroa-Moseley CD, Jean-Pierre P, Morrow GR (2007) Cancer-related fatigue: the scale of the problem. Oncologist 12:4–10. https://doi.org/10.1634/theoncologist.12-S1-4
Piper BF, Borneman T, Sun VC, Koczywas M, Uman G, Ferrell B, James RL (2008) Cancer-related fatigue: role of oncology nurses in translating National Comprehensive Cancer Network assessment guidelines into practice. Clin J Oncol Nurs 12:37–47. https://doi.org/10.1188/08.CJON.S2.37-47
Okuyama T, Akechi T, Kugaya A et al (2000) Development and validation of the cancer fatigue scale: a brief, three-dimensional, self-rating scale for assessment of fatigue in cancer patients. J Pain Symptom Manag 19:5–14. https://doi.org/10.1016/s0885-3924(99)00138-4
Wu HS, McSweeney M (2001) Measurement of fatigue in people with cancer. Oncol Nurs Forum 28:1371–1384
Cleeland CS, Mendoza TR, Wang XS et al (2000) Assessing symptom distress in cancer patient: the M. D. Anderson Symptom Inventory. Cancer 89:1634–1646. https://doi.org/10.1002/1097-0142(20001001)89:7<1634::aid-cncr29>3.0.co;2-v
Greimel ER, Kuljanic Vlasic K, Waldenstrom AC, Duric VM, Jensen PT, Singer S, Chie W, Nordin A, Bjelic Radisic V, Wydra D, European Organization for Research and Treatment of Cancer Quality-of-Life Group (2006) The European Organization for Research and Treatment of Cancer (EORTC) quality-of-life questionnaire cervical cancer module: EORTC QLQ-CX24. Cancer 107:1812–1822. https://doi.org/10.1002/cncr.22217
Knobel H, Loge JH, Brenne E, Fayers P, Hjermstad MJ, Kaasa S (2003) The validity of EORTC QLQ-C30 fatigue scale in advanced cancer patients and cancer survivors. Palliat Med 17:664–672. https://doi.org/10.1191/0269216303pm841oa
Glaus A (1993) Assessment of fatigue in cancer and non-cancer patients and in healthy individuals. Support Care Cancer 1:305–315. https://doi.org/10.1007/bf00364968
Munch TN, Strömgren AS, Pedersen L, Petersen MA, Hoermann L, Groenvold M (2006) Multidimensional measurement of fatigue in advanced cancer patients in palliative care: an application of the multidimensional fatigue inventory. J Pain Symptom Manag 31:533–541. https://doi.org/10.1016/j.jpainsymman.2005.11.012
Jean-Pierre P, Figueroa-Moseley CD, Kohli S, Fiscella K, Palesh OG, Morrow GR (2007) Assessment of cancer-related fatigue: implications for clinical diagnosis and treatment. Oncologist 12:11–21. https://doi.org/10.1634/theoncologist.12-S1-11
Acknowledgments
We thank the anonymous reviewers and editor for their comments.
Author information
Authors and Affiliations
Contributions
Conceptualization: Wen-Pei Chang; Hsiu-Ju Jen.
Data curation: Wen-Pei Chang.
Formal analysis: Wen-Pei Chang; Hsiu-Ju Jen.
Methodology: Wen-Pei Chang.
Project administration: Wen-Pei Chang.
Resources: Wen-Pei Chang.
Software: Wen-Pei Chang; Hsiu-Ju Jen.
Supervision: Wen-Pei Chang.
Validation: Wen-Pei Chang; Hsiu-Ju Jen.
Visualization: Wen-Pei Chang.
Writing, original draft: Wen-Pei Chang.
Writing, review and editing: Wen-Pei Chang.
Corresponding author
Ethics declarations
Conflict of interest
The authors declare that they have no conflict of interest.
Additional information
Publisher’s note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Wen-Pei, C., Hsiu-Ju, J. Changes in fatigue in rectal cancer patients before and after therapy: a systematic review and meta-analysis. Support Care Cancer 28, 2513–2522 (2020). https://doi.org/10.1007/s00520-020-05325-z
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00520-020-05325-z