Abstract
Purpose
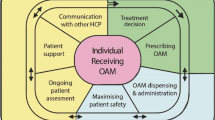
The use of oral cancer drugs (OAD) has increased over the last two decades. The objective of this study was to measure the impact of a nurse-led telephone follow-up in the therapeutic management of patients treated with an OAD regarding toxicity, medication adherence and quality of life.
Methods
A randomized, multicenter, controlled trial was conducted. All consecutive over 18-year-old patients, treated in medical oncology, radiotherapy, or hematology departments, receiving OAD for any cancer were invited to participate to the study. A total of 183 patients treated for solid or hematological cancers with an OAD were randomly assigned to receive a nurse-led telephone follow-up or standard care for 24 weeks. Data were collected between 2015 and 2018.
Results
Nurse telephone follow-up did not improve the global score toxicity in the intervention group. However, telephone calls directed by trained nurses induced a significant decrease in number of patients with grade 3 adverse events throughout the follow-up [OR 0.45 (IC à 95%) (0.23, 0.9)](P = 0.03). There was no significant difference in quality of life and medication adherence between groups at any follow-up time point.
Conclusions
In this first French real-life study, the advice provided by qualified nurses via phone calls improved the management of grade 3 toxicities but failed to demonstrate an improvement of all grades of toxicities. More prospective studies are needed to confirm the impact of telephone calls on the toxicities related to OAD.
Trial registration
Clinical trial registration is NCT02459483.
Protection committee SUD-ESTI registration is 2015-A00527-42 on 13 April 2015. National Agency for the Safety of Medicines and Health Products registration is 150619-B on the 27 may 2015.





Similar content being viewed by others
Data availability
All data are available on request.
References
UNICANCER - Quelle prise en charge des cancers en 2020? http://www.unicancer.fr/patients/quelle-prise-charge-cancers-2020. Accessed 25 Feb 2020
Van Cutsem E, Twelves C, Cassidy J et al (2001) Oral capecitabine compared with intravenous fluorouracil plus leucovorin in patients with metastatic colorectal cancer: results of a large phase III study. J Clin Oncol 19:4097–4106. https://doi.org/10.1200/JCO.2001.19.21.4097
Walko CM, Lindley C (2005) Capecitabine: a review. Clin Ther 27:23–44. https://doi.org/10.1016/j.clinthera.2005.01.005
Faithfull S, Deery P (2004) Implementation of capecitabine (Xeloda) into a cancer centre: UK experience. Eur J Oncol Nurs 8(Suppl 1):S54–S62. https://doi.org/10.1016/j.ejon.2004.06.009
Findlay M, von Minckwitz G, Wardley A (2008) Effective oral chemotherapy for breast cancer: pillars of strength. Ann Oncol 19:212–222. https://doi.org/10.1093/annonc/mdm285
Halfdanarson TR, Jatoi A (2010) Oral cancer chemotherapy: the critical interplay between patient education and patient safety. Curr Oncol Rep 12:247–252. https://doi.org/10.1007/s11912-010-0103-6
Weingart SN, Brown E, Bach PB, Eng K, Johnson SA, Kuzel TM, Langbaum TS, Leedy RD, Muller RJ, Newcomer LN, O’Brien S, Reinke D, Rubino M, Saltz L, Walters RS (2008) NCCN Task Force Report: Oral chemotherapy. J Natl Compr Canc Netw 6(Suppl 3):S1–S14
Shah NN, Casella E, Capozzi D, McGettigan S, Gangadhar TC, Schuchter L, Myers JS (2016) Improving the Safety of Oral Chemotherapy at an Academic Medical Center. J Oncol Pract 12:e71–e76. https://doi.org/10.1200/JOP.2015.007260
Zerillo JA, Goldenberg BA, Kotecha RR, Tewari AK, Jacobson JO, Krzyzanowska MK (2018) Interventions to improve oral chemotherapy safety and quality: a systematic review. JAMA Oncol 4:105–117. https://doi.org/10.1001/jamaoncol.2017.0625
Von Korff M, Gruman J, Schaefer J et al (1997) Collaborative management of chronic illness. Ann Intern Med 127:1097–1102. https://doi.org/10.7326/0003-4819-127-12-199712150-00008
Wasson J, Gaudette C, Whaley F, Sauvigne A, Baribeau P, Welch HG (1992) Telephone care as a substitute for routine clinic follow-up. JAMA 267:1788–1793
Jayakody A, Bryant J, Carey M, Hobden B, Dodd N, Sanson-Fisher R (2016) Effectiveness of interventions utilising telephone follow up in reducing hospital readmission within 30 days for individuals with chronic disease: a systematic review. BMC Health Serv Res 16:403. https://doi.org/10.1186/s12913-016-1650-9
Molassiotis A, Brearley S, Saunders M, Craven O, Wardley A, Farrell C, Swindell R, Todd C, Luker K (2009) Effectiveness of a home care nursing program in the symptom management of patients with colorectal and breast cancer receiving oral chemotherapy: a randomized, controlled trial. J Clin Oncol 27:6191–6198. https://doi.org/10.1200/JCO.2008.20.6755
Craven O, Hughes CA, Burton A, Saunders MP, Molassiotis A (2013) Is a nurse-led telephone intervention a viable alternative to nurse-led home care and standard care for patients receiving oral capecitabine? Results from a large prospective audit in patients with colorectal cancer. Eur J Cancer Care (Engl) 22:413–419. https://doi.org/10.1111/ecc.12047
Harris PA, Taylor R, Thielke R, Payne J, Gonzalez N, Conde JG (2009) Research electronic data capture (REDCap)--a metadata-driven methodology and workflow process for providing translational research informatics support. J Biomed Inform 42:377–381. https://doi.org/10.1016/j.jbi.2008.08.010
Willis K, Lewis S, Ng F, Wilson L (2015) The experience of living with metastatic breast cancer--a review of the literature. Health Care Women Int 36:514–542. https://doi.org/10.1080/07399332.2014.896364
Hendriksen E, Williams E, Sporn N, Greer J, DeGrange A, Koopman C (2015) Worried together: a qualitative study of shared anxiety in patients with metastatic non-small cell lung cancer and their family caregivers. Support Care Cancer 23:1035–1041. https://doi.org/10.1007/s00520-014-2431-9
Di Maio M, Basch E, Bryce J, Perrone F (2016) Patient-reported outcomes in the evaluation of toxicity of anticancer treatments. Nat Rev Clin Oncol 13:319–325. https://doi.org/10.1038/nrclinonc.2015.222
Eldeib HK, Abbassi MM, Hussein MM, Salem SE, Sabry NA (2019) The effect of telephone-based follow-up on adherence, efficacy, and toxicity of oral capecitabine-based chemotherapy. Telemed J E Health 25:462–470. https://doi.org/10.1089/tmj.2018.0077
Hakamies-Blomqvist L, Luoma ML, Sjöström J et al (2001) Timing of quality of life (QoL) assessments as a source of error in oncological trials. J Adv Nurs 35:709–716. https://doi.org/10.1046/j.1365-2648.2001.01903.x
Greer JA, Amoyal N, Nisotel L, Fishbein JN, MacDonald J, Stagl J, Lennes I, Temel JS, Safren SA, Pirl WF (2016) A systematic review of adherence to oral antineoplastic therapies. Oncologist 21:354–376. https://doi.org/10.1634/theoncologist.2015-0405
Schneider SM, Hess K, Gosselin T (2011) Interventions to promote adherence with oral agents. Semin Oncol Nurs 27:133–141. https://doi.org/10.1016/j.soncn.2011.02.005
Skrabal Ross X, Gunn KM, Patterson P, Olver I (2018) Mobile-based oral chemotherapy adherence–enhancing interventions: scoping review. JMIR Mhealth Uhealth 6. https://doi.org/10.2196/11724
Drzayich Antol D, Waldman Casebeer A, Khoury R, et al (2018) The relationship between comorbidity medication adherence and health related quality of life among patients with cancer. J Patient Rep Outcomes 2. https://doi.org/10.1186/s41687-018-0057-2
Salgado TM, Mackler E, Severson JA, Lindsay J, Batra P, Petersen L, Farris KB (2017) The relationship between patient activation, confidence to self-manage side effects, and adherence to oral oncolytics: a pilot study with Michigan oncology practices. Support Care Cancer 25:1797–1807. https://doi.org/10.1007/s00520-017-3584-0
Basch E, Deal AM, Dueck AC, Scher HI, Kris MG, Hudis C, Schrag D (2017) Overall survival results of a trial assessing patient-reported outcomes for symptom monitoring during routine cancer treatment. JAMA 318:197–198. https://doi.org/10.1001/jama.2017.7156
Denis F, Lethrosne C, Pourel N, et al (2017) Randomized trial comparing a web-mediated follow-up with routine surveillance in lung cancer patients. J Natl Cancer Inst 109. https://doi.org/10.1093/jnci/djx029
Walter T, Wang L, Chuk K, Ng P, Tannock IF, Krzyzanowska MK (2014) Assessing adherence to oral chemotherapy using different measurement methods: lessons learned from capecitabine. J Oncol Pharm Pract 20:249–256. https://doi.org/10.1177/1078155213501100
Acknowledgments
The authors gratefully acknowledges all pivot nurses of Lucien Neuwirth Cancer Institute for their help and involvement in this study.
The authors gratefully acknowledges the funding agencies, as well as Mr. Eric- Alban Giroux, the investigators, the collaborators, the medical and paramedical nurses, and all clinical research associates.
The authors would like to thank Miss Sandrine Sotton for English editing services.
Funding
This study was supported by “Le réseau espace santé cancer Rhones-Alpes: INNOV’RA 2014”, “La ligue contre le cancer” and “Novartis Pharma SAS” and the financial support of the Institute of Cancerology Lucien Neuwirth; no grant number is applicable.
Author information
Authors and Affiliations
Contributions
Conceptualization, Mathieu Oriol; Aurélie Bourmaud; Cécile Vassal; Methodology, Mathieu Oriol; Aurélie Bourmaud; Cécile Vassal; Fabien Tinquaut; Software, Sidonie Pupier; Validation, Cécile Vassal; Wafa Bouleftour; Fabien Tinquaut; Formal Analysis, Wafa Bouleftour; Fabien Tinquaut; Cécile Vassal; Investigation, Thierry Muron; Aline Guillot; Romain Rivoirard; Jean-Philippe Jacquin; Léa Saban-Roche; Karima Boussoualim; Emmanuelle Tavernier; Karine Augeul-Meunier; Olivier Collard; Benoite Mery; Pierre Fournel; Writing—Original Draft Preparation, Wafa Bouleftour; Writing—Review and Editing, all authors
Corresponding author
Ethics declarations
Conflict of interest/Competing interests
The authors declare that they have no conflicts of interest.
Ethics approval
All procedures performed in studies involving human participants were in accordance with the ethical standards of the institutional and/or national research committee and with the 1964 Helsinki declaration and its later amendments or comparable ethical standards.
Consent to participate
Informed consent was obtained from all individual participants included in the study.
Consent for publication
All the authors gave their consent for the publication of this work.
Code availability
NA
Additional information
Publisher’s note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Bouleftour, W., Muron, T., Guillot, A. et al. Effectiveness of a nurse-led telephone follow-up in the therapeutic management of patients receiving oral antineoplastic agents: a randomized, multicenter controlled trial (ETICCO study). Support Care Cancer 29, 4257–4267 (2021). https://doi.org/10.1007/s00520-020-05955-3
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00520-020-05955-3