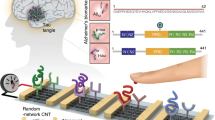
Abstract
An innovative sensing assay is described for point-of-care (PoC) quantification of a biomarker of Alzheimer’s disease, amyloid β-42 (Aβ-42). This device is based on a cellulose paper-dye test strip platform in which the corresponding detection layer is integrated by applying a molecularly imprinted polymer (MIP) to the cellulose paper surface. Briefly, the cellulose paper is chemically modified with a silane to subsequently apply the MIP detection layer. The imprinting process is confirmed by the parallel preparation of a control material, namely a non-imprinted polymer (NIP). The chemical changes of the surface were evaluated by Fourier transform infrared spectroscopy (FTIR), contact angle, and thermogravimetric analysis (TG). Proteins and peptides can be quantified by conventional staining methods. For this purpose, Coomassie blue (CB) was used as a staining dye for the detection and quantification of Aβ-42. Quantitative determination is made possible by taking a photograph and applying an appropriate mathematical treatment to the color coordinates provided by the ImageJ program. The MIP shows a linear range between 1.0 ng/mL and 10 μg/mL and a detection limit of 0.71 ng/mL. Overall, this cellulose-based assay is suitable for the detection of peptides or proteins in a sample by visual comparison of color change. The test strip provides a simple, instrument-free, and cost-effective method with high chemical stability, capable of detecting very small amounts of peptides or proteins in a sample, and can be used for the detection of any (bio)molecule of interest.
Graphical abstract







Similar content being viewed by others
References
Mascini M, Tombelli S (2008) Biosensors for biomarkers in medical diagnostics. Biomarkers. 13(7–8):637–657. https://doi.org/10.1080/13547500802645905
Luo Y, Zhang B, Chen M, Jiang T, Zhou D, Huang J, Fu W (2012) Sensitive and rapid quantification of C-reactive protein using quantum dot-labeled microplate immunoassay. J Transl Med 10:10. https://doi.org/10.1186/1479-5876-10-24
Kuntaruk S, Tatu T, Keowkarnkah T, Kasinrerk W (2010) Sandwich ELISA for hemoglobin A(2) quantification and identification of beta-thalassemia carriers nt. J Hematol 91(2):219–228. https://doi.org/10.1007/s12185-009-0490-3
Clarke NJ, Tomlinson AJ, Ohyagi Y, Younkin S, Naylor S (1998) Detection and quantitation of cellularly derived amyloid beta peptides by immunoprecipitation-HPLC-MS. FEBS Lett 430(3):419–423. https://doi.org/10.1016/s0014-5793(98)00706-6
Schuder S, Wittenberg JB, Haseltine B, Wittenberg BA (1979) Spectrophotometric determination of myoglobin in cardiac and skeletal-muscle - separation from hemoglobin by subunit-exchange chromatography. Anal Biochem 192(2):473–481. https://doi.org/10.1016/0003-2697(79)90687-0
Loo L, Wu W, Shih WY, Shih W-H, Borghaei H, Pourrezaei K, Adams GP (2011) A rapid method to regenerate piezoelectric microcantilever sensors (PEMS). Sensors 11(5):5520–5528. https://doi.org/10.3390/s110505520
Ma L, Wang C, Zhang M (2011) Detecting protein adsorption and binding using magnetic nanoparticle probes. Sensor Actuat B-Chem 160(1). https://doi.org/10.1016/j.snb.2011.08.043
Mohammed M-I, Desmulliez MPY (2011) Lab-on-a-chip based immunosensor principles and technologies for the detection of cardiac biomarkers: a review. Lab Chip 11(4):569–595. https://doi.org/10.1039/c0lc00204f
Sanchez-Portillo P, Hernandez-Sirio A, Godoy-Alcantar C, Lacroix PG, Agarwal V, Santillan R et al (2021) Colorimetric metal ion (II) sensors based on imine boronic esters functionalized with pyridine. Dyes Pigm 108991. https://doi.org/10.1016/j.dyepig.2020.108991
Kang BH, Park M, Jeong KH (2017) Colorimetric Schirmer strip for tear glucose detection. Biochip J 11(4):294–299. https://doi.org/10.1007/s13206-017-1405-7
Kudo H, Maejima K, Hiruta Y, Citterio D (2020) Microfluidic paper-based analytical devices for colorimetric detection of lactoferrin. Slas Technology 25(1):47–57. https://doi.org/10.1177/2472630319884031
Zeng L, Liu LQ, Kuang H, Cui G, Xu CL (2019) A paper-based colorimetric assay for rapid detection of four macrolides in milk. Mat Chem Front 3(10):2175–2183. https://doi.org/10.1039/c9qm00429g
Carrell C, Kava A, Nguyen M, Menger R, Munshi Z, Call Z, Nussbaum M, Henry C (2019) Beyond the lateral flow assay: a review of paper-based microfluidics. Microelectron Eng 206:45–54. https://doi.org/10.1016/j.mee.2018.12.002
Bushman ET, Jauk VC, Szychowski JM, Mazzoni S, Tita AT, Harper LM (2020) Utility of routine dipstick urinalysis for glucose screening as a predictor for gestational diabetes. Amer J Obst Gynec 222(1):S161–S1S2. https://doi.org/10.1016/j.ajog.2019.11.248
Hu J, Wang SQ, Wang L, Li F, Pingguan-Murphy B, Lu TJ, Xu F (2014) Advances in paper-based point-of-care diagnostics. Biosens Bioelectron 54:585–597. https://doi.org/10.1016/j.bios.2013.10.075
Albareda-Sirvent M, Merkoci A, Alegret S (2000) Configurations used in the design of screen-printed enzymatic biosensors. A review. Sensors Actuat B-Chem 269(1–2):153–163. https://doi.org/10.1016/s0925-4005(00)00536-0
Chen Z, Xi F, Yang S, Wu Q, Lin X (2008) Development of a bienzyme system based on sugar-lectin biospecific interactions for amperometric determination of phenols and aromatic amines. Sensors Actuat B-Chem 130(2):900–907. https://doi.org/10.1016/j.snb.2007.10.075
Cai H, Wang YQ, He PG, Fang YH (2002) Electrochemical detection of DNA hybridization based on silver-enhanced gold nanoparticle label. Anal Chim Acta 469(2):165–172. https://doi.org/10.1016/s0003-2670(02)00670-0
Chang Z, Pan H, Zhao K, Chen M, He P, Fang Y (2008) Electrochemical DNA biosensors based on palladium nanoparticles combined with carbon nanotubes. Electroanalysis 20(2):131–136. https://doi.org/10.1002/elan.200704023
Moreira FTC, Sharma S, Dutra RAF, Noronha JPC, Cass AEG, Sales MGF (2013) Smart plastic antibody material (SPAM) tailored on disposable screen printed electrodes for protein recognition: application to myoglobin detection. Biosens Bioelectron 45:237–244. https://doi.org/10.1016/j.bios.2013.02.012
Chen L, Wang X, Lu W, Wu X, Li J (2016) Molecular imprinting: perspectives and applications. Chem Soc Rev 45(8):2137–2211
Guan GJ, Liu BH, Wang ZY, Zhang ZP (2008) Imprinting of molecular recognition sites on nanostructures and its applications in chemosensors. Sensors. 8(12):8291–8320. https://doi.org/10.3390/s8128291
Lee GH, Hah SS (2012) Coomassie blue is sufficient for specific protein detection of aptamer-conjugated chips. Chemistry Letters is Bioorganic Med 22(4):1520–1522. https://doi.org/10.1016/j.bmcl.2012.01.012
Eynard L, Lauriere M (1998) The combination of Indian ink staining with immunochemiluminescence detection allows precise identification of antigens on blots: application to the study of glycosylated barley storage proteins. Electrophoresis. 19(8–9):1394–1396. https://doi.org/10.1002/elps.1150190833
Mitra P, Pal AK, Basu D, Hati RN (1994) A staining procedure using coomassie brilliant blue g-250 in phosphoric-acid for detection of protein bands with high-resolution in polyacrylamide-gel and nitrocellulose membrane. Anal Biochem 223(2):327–329. https://doi.org/10.1006/abio.1994.1594
Simpson RJ. Rapid coomassie blue staining of protein gels. (2010) Cold Spring Harb prot (4):pdb.prot5413-pdb.prot. doi: https://doi.org/10.1101/pdb.prot5413
Castellanos-Serra LR, Hardy E, Proenza W, Huerta V, Gonzalez LJ, Le-Caer JP et al (2000) Reversible negative staining of protein on electrophoresis gels by imidazole-zinc salts: micropreparative applications to proteome analysis by mass spectrometry. Prot Protein Anal:29–52. https://doi.org/10.1007/978-3-642-59631-5_3
Chevallet M, Luche S, Rabilloud T (2006) Silver staining of proteins in polyacrylamide gels. Nat Prot 1(4):1852–1858. https://doi.org/10.1038/nprot.2006.288
Derayea SM, Nagy DM (2018) Application of a xanthene dye, eosin Y, as spectroscopic probe in chemical and pharmaceutical analysis; a review. Rev Anal Chem 37(3). https://doi.org/10.1515/revac-2017-0020
Pereira MV, Marques AC, Oliveira D, Martins R, Moreira FTC, Sales MGF, Fortunato E (2020) Paper-based platform with an in situ molecularly imprinted polymer for beta-amyloid. Acs Omega 5(21):12057–12066. https://doi.org/10.1021/acsomega.0c00062
Moreira FTC, Sales MGF (2017) Smart naturally plastic antibody based on poly(alpha-cyclodextrin) polymer for beta-amyloid-42 soluble oligomer detection. Sensors Actuat B-Chem 240:229–238. https://doi.org/10.1016/j.snb.2016.08.150
Bonini F, Piletsky S, Turner APF, Speghini A, Bossi A (2007) Surface imprinted beads for the recognition of human serum albumin. Biosens Bioelectron 22(9–10):2322–2328. https://doi.org/10.1016/j.bios.2006.12.034
Migneault I, Dartiguenave C, Bertrand MJ, Waldron KC (2004) Glutaraldehyde: behavior in aqueous solution, reaction with proteins, and application to enzyme crosslinking. Biotechniques 37(5):790–802. https://doi.org/10.2144/04375RV01
Hossain MM, Trinh QH, Nguyen DB, Sudhakaran MSP, Mok YS (2019) Robust hydrophobic coating on glass surface by an atmospheric-pressure plasma jet for plasma-polymerisation of hexamethyldisiloxane conjugated with (3-aminopropyl) triethoxysilane. Surf Eng 35(5):466–475. https://doi.org/10.1080/02670844.2018.1524037
Yin YY, Ma JJ, Tian XZ, Jiang X, Wang HB, Gao WD (2018) Cellulose nanocrystals functionalized with amino-silane and epoxy-poly(ethylene glycol) for reinforcement and flexibilization of poly(lactic acid): material preparation and compatibility mechanism. Cellulose. 25(11):6447–6463. https://doi.org/10.1007/s10570-018-2033-7
Schmidt B (2019) Hydrophilic polymers. Polymers 11(4). https://doi.org/10.3390/polym11040693
Fazekas SDS, Webster RG, Datyner A (1963) New staining procedures for quantitative estimation of proteins on electrophoretic strips. Biochim Biophys Acta 71(2):377–391. https://doi.org/10.1016/0006-3002(63)91092-8
Li SS, Lin CW, Wei KC, Huang CY, Hsu PH, Liu HL, Lu YJ, Lin SC, Yang HW, Ma CCM (2016) Non-invasive screening for early Alzheimer’s disease diagnosis by a sensitively immunomagnetic biosensor. Sci Rep 6:1–11. https://doi.org/10.1038/srep25155
Diba FS, Kim S, Lee HJ (2017) Electrochemical immunoassay for amyloid-beta 1-42 peptide in biological fluids interfacing with a gold nanoparticle modified carbon surface. Cat Today 295:41–47. https://doi.org/10.1016/j.cattod.2017.02.039
Lien TTN, Takamura Y, Tamiya E, Vestergaard MC (2015) Modified screen printed electrode for development of a highly sensitive label-free impedimetric immunosensor to detect amyloid beta peptides. Anal Chim Acta 892:69–76. https://doi.org/10.1016/j.aca.2015.08.036
Hu T, Lu SS, Chen CX, Sun J, Yang XR (2017) Colorimetric sandwich immunosensor for A beta((1-42)) based on dual antibody-modified gold nanoparticles. Sensors Actuat B-Chem 243:792–799. https://doi.org/10.1016/j.snb.2016.12.052
Dai YF, Molazemhosseini A, Liu CC (2017) In vitro quantified determination of beta-amyloid 42 peptides, a biomarker of neuro-degenerative disorders, in PBS and human serum using a simple, cost-effective thin gold film biosensor. Biosensors-Basel. 7(3):1–29. https://doi.org/10.3390/bios7030029
Wang JX, Zhuo Y, Zhou Y, Wang HJ, Yuan R, Chai YQ (2016) Ceria doped zinc oxide nanoflowers enhanced luminol-based electrochemiluminescence immunosensor for amyloid-beta detection. ACS Appl Mater Interfaces 8(20):12968–12975. https://doi.org/10.1021/acsami.6b00021
Kang DY, Lee JH, Oh BK, Choi JW (2009) Ultra-sensitive immunosensor for beta-amyloid (1-42) using scanning tunneling microscopy-based electrical detection. Biosens Bioelectron 24(5):1431–1436. https://doi.org/10.1016/j.bios.2008.08.018
Ly TN, Park S (2020) High performance detection of Alzheimer’s disease biomarkers based on localized surface plasmon resonance. J Ind Eng Chem 91:182–190. https://doi.org/10.1016/j.jiec.2020.07.051
Zhao GH, Wang YG, Li XJ, Yue Q, Dong X, Du B et al (2019) Dual-quenching electrochemiluminescence strategy based on three-dimensional metal-organic frameworks for ultrasensitive detection of amyloid-beta. Anal Chem 91(3):1989–1996. https://doi.org/10.1021/acs.analchem.8b04332
Liu L, Xia N, Zhang JB, Mao WH, Wu YY, Ge XX (2015) A graphene oxide-based fluorescent platform for selective detection of amyloid-beta oligomers. Anal Methods 7(20):8727–8732. https://doi.org/10.1039/c5ay02018b
Qin J, Jo DG, Cho M, Lee Y (2018) Monitoring of early diagnosis of Alzheimer’s disease using the cellular prion protein and poly(pyrrole-2-carboxylic acid) modified electrode. Biosens Bioelectron 113:82–87. https://doi.org/10.1016/j.bios.2018.04.061
Deng CY, Liu H, Si SH, Zhu XJ, Tu QY, Jin Y, Xiang J (2020) An electrochemical aptasensor for amyloid-beta oligomer based on double-stranded DNA as “conductive spring”. Microchim Acta 187(4):1–8. https://doi.org/10.1007/s00604-020-4217-8
Liu YBA, Xu Q, Zhang YN, Ren BY, Huang LM, Cai H, Xu T, Liu Q, Zhang X (2021) An electrochemical aptasensor based on AuPt alloy nanoparticles for ultrasensitive detection of amyloid-beta oligomers. Talanta. 231:122360. https://doi.org/10.1016/j.talanta.2021.122360
Zhang X, Liu S, Song XL, Wang HW, Wang JF, Wang Y, Huang J, Yu J (2019) Robust and universal SERS sensing platform for multiplexed detection of Alzheimer’s disease core biomarkers using PAapt-AuNPs conjugates. Acs Sensors 4(8):2140–2149. https://doi.org/10.1021/acssensors.9b00974
Moreira FTC, Rodriguez BAG, Dutra RAF, Sales MGF (2018) Redox probe-free readings of a beta-amyloid-42 plastic antibody sensory material assembled on copper@carbon nanotubes. Sensors Actuat B-Chem 264:1–9. https://doi.org/10.1016/j.snb.2018.02.166
Ozcan N, Medetalibeyoglu H, Akyildirim O, Atar N, Yola ML (2020) Electrochemical detection of amyloid-beta protein by delaminated titanium carbide MXene/multi-walled carbon nanotubes composite with molecularly imprinted polymer. Mater Today Commun 23:101097. https://doi.org/10.1016/j.mtcomm.2020.101097
Acknowledgements
0624_2IQBIONEURO_6_E, Impulso de una red de I + i en química biológica para diagnóstico y tratamiento de enfermedades neurológicas EP - INTERREG V A España Portugal (POCTEP).
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
There is no any financial interests/personal relationships which may be considered as potential competing interests.
Additional information
Publisher’s note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary information
ESM 1
(DOCX 2256 kb)
Rights and permissions
About this article
Cite this article
Moreira, F.T., Correia, B.P., Sousa, M.P. et al. Colorimetric cellulose-based test-strip for rapid detection of amyloid β-42. Microchim Acta 188, 334 (2021). https://doi.org/10.1007/s00604-021-04996-7
Received:
Accepted:
Published:
DOI: https://doi.org/10.1007/s00604-021-04996-7