Abstract
Epibrassinolide (EBR), a polyhydroxysteroid belongs to plant growth regulator family, brassinosteroids and has been shown to have a similar chemical structure to mammalian steroid hormones. Our findings indicated that EBR could trigger apoptosis in cancer cells via induction of endoplasmic reticulum (ER) stress, caused by protein folding disturbance in the ER. Normal cells exhibited a remarkable resistance to EBR treatment and avoid from apoptotic cell death. The unfolded protein response clears un/misfolded proteins and restore ER functions. When stress is chronic, cells tend to die due to improper cellular functions. To understand the effect of EBR in non-malign cells, mouse embryonic fibroblast (MEF) cells were investigated in detail for ER stress biomarkers, autophagy, and polyamine metabolism in this study. Evolutionary conserved autophagy mechanism is a crucial cellular process to clean damaged organelles and protein aggregates through lysosome under the control of autophagy-related genes (ATGs). Cells tend to activate autophagy to promote cell survival under stress conditions. Polyamines are polycationic molecules playing a role in the homeostasis of important cellular events such as cell survival, growth, and, proliferation. The administration of PAs has been markedly extended the lifespan of various organisms via inducing autophagy and inhibiting oxidative stress. Our data indicated that ER stress is induced following EBR treatment in MEF cells as well as MEF Atg5−/− cells. In addition, autophagy is activated following EBR treatment by targeting PI3K/Akt/mTOR in wildtype (wt) cells. However, EBR-induced autophagy targets ULK1 in MEF cells lacking Atg5 expression. Besides, EBR treatment depleted the PA pool in MEF cells through the alterations of metabolic enzymes. The administration of Spd with EBR further increased autophagic vacuole formation. In conclusion, EBR is an anticancer drug candidate with selective cytotoxicity for cancer cells, in addition the induction of autophagy and PA metabolism are critical for responses of normal cells against EBR.









Similar content being viewed by others
References
Alasiri G, Fan LY, Zona S, Goldsbrough IG, Ke HL, Auner HW, Lam EW (2018) ER stress and cancer: the FOXO forkhead transcription factor link. Mol Cell Endocrinol 462(Pt B):67–81. https://doi.org/10.1016/j.mce.2017.05.027
Antonucci L, Fagman JB, Kim JY, Todoric J, Gukovsky I, Mackey M, Ellisman MH, Karin M (2015) Basal autophagy maintains pancreatic acinar cell homeostasis and protein synthesis and prevents ER stress. Proc Natl Acad Sci USA 112(45):E6166–6174. https://doi.org/10.1073/pnas.1519384112
Arisan ED, Akkoc Y, Akyuz KG, Kerman EM, Obakan P, Coker-Gurkan A, Palavan Unsal N (2015) Polyamines modulate the roscovitine-induced cell death switch decision autophagy vs. apoptosis in MCF-7 and MDA-MB-231 breast cancer cells. Mol Med Rep 11(6):4532–4540. https://doi.org/10.3892/mmr.2015.3303
Arisan ED, Obakan P, Coker-Gurkan A, Calcabrini A, Agostinelli E, Unsal NP (2014) CDK inhibitors induce mitochondria-mediated apoptosis through the activation of polyamine catabolic pathway in LNCaP, DU145 and PC3 prostate cancer cells. Curr Pharm Des 20(2):180–188
Cao S, Yan B, Lu Y, Zhang G, Li J, Guo W, Zhao Y, Zhang S (2015) C/EBP homologous protein-mediated endoplasmic reticulum stress-related renal apoptosis is involved in rats with brain death. Transplant Proc 47(2):354–358. https://doi.org/10.1016/j.transproceed.2014.10.012
Cheng Y, Yang JM (2011) Survival and death of endoplasmic-reticulum-stressed cells: role of autophagy. World J Biol Chem 2(10):226–231. https://doi.org/10.4331/wjbc.v2.i10.226
Cnop M, Toivonen S, Igoillo-Esteve M, Salpea P (2017) Endoplasmic reticulum stress and eIF2alpha phosphorylation: the Achilles heel of pancreatic beta cells. Mol Metab 6(9):1024–1039. https://doi.org/10.1016/j.molmet.2017.06.001
Codogno P, Meijer AJ (2006) Atg5: more than an autophagy factor. Nat Cell Biol 8(10):1045–1047. https://doi.org/10.1038/ncb1006-1045
Coker A, Arisan ED, Palavan-Unsal N (2012) Silencing of the polyamine catabolic key enzyme SSAT prevents CDK inhibitor-induced apoptosis in Caco-2 colon cancer cells. Mol Med Rep 5(4):1037–1042. https://doi.org/10.3892/mmr.2012.768
Copp J, Manning G, Hunter T (2009) TORC-specific phosphorylation of mammalian target of rapamycin (mTOR): phospho-Ser2481 is a marker for intact mTOR signaling complex 2. Can Res 69(5):1821–1827. https://doi.org/10.1158/0008-5472.CAN-08-3014
Coskun D, Obakan P, Arisan ED, Coker-Gurkan A, Palavan-Unsal N (2015) Epibrassinolide alters PI3K/MAPK signaling axis via activating Foxo3a-induced mitochondria-mediated apoptosis in colon cancer cells. Exp Cell Res 338(1):10–21. https://doi.org/10.1016/j.yexcr.2015.08.015
Cubillos-Ruiz JR, Bettigole SE, Glimcher LH (2017) Tumorigenic and immunosuppressive effects of endoplasmic reticulum stress in cancer. Cell 168(4):692–706. https://doi.org/10.1016/j.cell.2016.12.004
Cybulsky AV (2017) Endoplasmic reticulum stress, the unfolded protein response and autophagy in kidney diseases. Nat Rev Nephrol 13(11):681–696. https://doi.org/10.1038/nrneph.2017.129
Damiani E, Wallace HM (2018) Polyamines and cancer. Methods Mol Biol 1694:469–488. https://doi.org/10.1007/978-1-4939-7398-9_39
de Cabo R, Navas P (2016) Spermidine to the rescue for an aging heart. Nat Med 22(12):1389–1390. https://doi.org/10.1038/nm.4243
Deldicque L (2013) Endoplasmic reticulum stress in human skeletal muscle: any contribution to sarcopenia? Front Physiol 4:236. https://doi.org/10.3389/fphys.2013.00236
Dewaele M, Martinet W, Rubio N, Verfaillie T, de Witte PA, Piette J, Agostinis P (2011) Autophagy pathways activated in response to PDT contribute to cell resistance against ROS damage. J Cell Mol Med 15(6):1402–1414. https://doi.org/10.1111/j.1582-4934.2010.01118.x
Dijkers PF, Medema RH, Lammers JW, Koenderman L, Coffer PJ (2000) Expression of the pro-apoptotic Bcl-2 family member Bim is regulated by the forkhead transcription factor FKHR-L1. Curr Biol 10(19):1201–1204
Fujita E, Kouroku Y, Isoai A, Kumagai H, Misutani A, Matsuda C, Hayashi YK, Momoi T (2007) Two endoplasmic reticulum-associated degradation (ERAD) systems for the novel variant of the mutant dysferlin: ubiquitin/proteasome ERAD(I) and autophagy/lysosome ERAD(II). Hum Mol Genet 16(6):618–629. https://doi.org/10.1093/hmg/ddm002
Guo H, Lin W, Zhang X, Zhang X, Hu Z, Li L, Duan Z, Zhang J, Ren F (2017) Kaempferol induces hepatocellular carcinoma cell death via endoplasmic reticulum stress-CHOP-autophagy signaling pathway. Oncotarget 8(47):82207–82216. https://doi.org/10.18632/oncotarget.19200
Gong C, Hu C, Gu F, Xia Q, Yao C, Zhang L, Qiang L, Gao S, Gao Y (2017) Co-delivery of autophagy inhibitor ATG7 siRNA and docetaxel for breast cancer treatment. J Control Release 266:272–286
Gonzalez Y, Aryal B, Chehab L, Rao VA (2014) Atg7- and Keap1-dependent autophagy protects breast cancer cell lines against mitoquinone-induced oxidative stress. Oncotarget 5(6):1526–1537. https://doi.org/10.18632/oncotarget.1715
Gwinn DM, Shackelford DB, Egan DF, Mihaylova MM, Mery A, Vasquez DS, Turk BE, Shaw RJ (2008) AMPK phosphorylation of raptor mediates a metabolic checkpoint. Mol Cell 30(2):214–226. https://doi.org/10.1016/j.molcel.2008.03.003
Hassler J, Cao SS, Kaufman RJ (2012) IRE1, a double-edged sword in pre-miRNA slicing and cell death. Dev Cell 23(5):921–923. https://doi.org/10.1016/j.devcel.2012.10.025
Heras-Sandoval D, Perez-Rojas JM, Hernandez-Damian J, Pedraza-Chaverri J (2014) The role of PI3K/AKT/mTOR pathway in the modulation of autophagy and the clearance of protein aggregates in neurodegeneration. Cell Signal 26(12):2694–2701. https://doi.org/10.1016/j.cellsig.2014.08.019
Hsieh WC, Hsu PC, Liao YF, Young ST, Wang ZW, Lin CL, Tsay GJ, Lee H, Hung HC, Liu GY (2010) Overexpression of ornithine decarboxylase suppresses thapsigargin-induced apoptosis. Mol Cells 30(4):311–318. https://doi.org/10.1007/s10059-010-0120-1
Igarashi K, Kashiwagi K (2000) Polyamines: mysterious modulators of cellular functions. Biochem Biophys Res Commun 271(3):559–564. https://doi.org/10.1006/bbrc.2000.2601
Itakura E, Kishi C, Inoue K, Mizushima N (2008) Beclin 1 forms two distinct phosphatidylinositol 3-kinase complexes with mammalian Atg14 and UVRAG. Mol Biol Cell 19(12):5360–5372. https://doi.org/10.1091/mbc.E08-01-0080
Kahana C (2018) The antizyme family for regulating polyamines. J Biol Chem 293(48):18730–18735. https://doi.org/10.1074/jbc.TM118.003339
Kahana C (2009) Antizyme and antizyme inhibitor, a regulatory tango. Cell Mol Life Sci 66:2479–2488. https://doi.org/10.1007/s00018-009-0033-3
Kang R, Zeh HJ, Lotze MT, Tang D (2011) The Beclin 1 network regulates autophagy and apoptosis. Cell Death Differ 18(4):571–580. https://doi.org/10.1038/cdd.2010.191
Kucheryavenko O, Nelson G, von Zglinicki T, Korolchuk VI, Carroll B (2019) The mTORC1-autophagy pathway is a target for senescent cell elimination. Biogerontology. https://doi.org/10.1007/s10522-019-09802-9
Landau G, Ran A, Bercovich Z, Feldmesser E, Horn-Saban S, Korkotian E, Jacob-Hirsh J, Rechavi G, Ron D, Kahana C (2012) Expression profiling and biochemical analysis suggest stress response as a potential mechanism inhibiting proliferation of polyamine-depleted cells. J Biol Chem 287(43):35825–35837. https://doi.org/10.1074/jbc.M112.381335
Lei K, Davis RJ (2003) JNK phosphorylation of Bim-related members of the Bcl2 family induces Bax-dependent apoptosis. Proc Natl Acad Sci USA 100(5):2432–2437. https://doi.org/10.1073/pnas.0438011100
Li W, Saud SM, Young MR, Chen G, Hua B (2015) Targeting AMPK for cancer prevention and treatment. Oncotarget 6(10):7365–7378. https://doi.org/10.18632/oncotarget.3629
Li W, Zhu J, Dou J, She H, Tao K, Xu H, Yang Q, Mao Z (2017) Phosphorylation of LAMP2A by p38 MAPK couples ER stress to chaperone-mediated autophagy. Nat Commun 8(1):1763. https://doi.org/10.1038/s41467-017-01609-x
Luo B, Lee AS (2013) The critical roles of endoplasmic reticulum chaperones and unfolded protein response in tumorigenesis and anticancer therapies. Oncogene 32(7):805–818. https://doi.org/10.1038/onc.2012.130
Madeo F, Carmona-Gutierrez D, Kepp O, Kroemer G (2018) Spermidine delays aging in humans. Aging (Albany NY) 10(8):2209–2211. https://doi.org/10.18632/aging.101517
Madeo F, Eisenberg T, Buttner S, Ruckenstuhl C, Kroemer G (2010) Spermidine: a novel autophagy inducer and longevity elixir. Autophagy 6(1):160–162
Marquez RT, Xu L (2012) Bcl-2: Beclin 1 complex: multiple, mechanisms regulating autophagy/apoptosis toggle switch. Am J Cancer Res 2(2):214–221
Massey AC, Kaushik S, Sovak G, Kiffin R, Cuervo AM (2006) Consequences of the selective blockage of chaperone-mediated autophagy. Proc Natl Acad Sci USA 103(15):5805–5810. https://doi.org/10.1073/pnas.0507436103
Meijer AJ, Lorin S, Blommaart EF, Codogno P (2015) Regulation of autophagy by amino acids and MTOR-dependent signal transduction. Amino Acids 47(10):2037–2063. https://doi.org/10.1007/s00726-014-1765-4
Minois N, Rockenfeller P, Smith TK, Carmona-Gutierrez D (2014) Spermidine feeding decreases age-related locomotor activity loss and induces changes in lipid composition. PLoS ONE 9(7):e102435. https://doi.org/10.1371/journal.pone.0102435
Morselli E, Galluzzi L, Kepp O, Criollo A, Maiuri MC, Tavernarakis N, Madeo F, Kroemer G (2009) Autophagy mediates pharmacological lifespan extension by spermidine and resveratrol. Aging (Albany NY) 1(12):961–970. https://doi.org/10.18632/aging.100110
Murray Stewart T, Dunston TT, Woster PM, Casero RA Jr (2018) Polyamine catabolism and oxidative damage. J Biol Chem 293(48):18736–18745. https://doi.org/10.1074/jbc.TM118.003337
Nishida Y, Arakawa S, Fujitani K, Yamaguchi H, Mizuta T, Kanaseki T, Komatsu M, Otsu K, Tsujimoto Y, Shimizu S (2009) Discovery of Atg5/Atg7-independent alternative macroautophagy. Nature 461(7264):654–658. https://doi.org/10.1038/nature08455
Obakan-Yerlikaya P, Arisan ED, Coker-Gurkan A, Adacan K, Ozbey U, Somuncu B, Baran D, Palavan-Unsal N (2017) Calreticulin is a fine tuning molecule in epibrassinolide-induced apoptosis through activating endoplasmic reticulum stress in colon cancer cells. Mol Carcinog 56(6):1603–1619. https://doi.org/10.1002/mc.22616
Obakan P, Arisan ED, Calcabrini A, Agostinelli E, Bolkent S, Palavan-Unsal N (2014a) Activation of polyamine catabolic enzymes involved in diverse responses against epibrassinolide-induced apoptosis in LNCaP and DU145 prostate cancer cell lines. Amino Acids 46(3):553–564. https://doi.org/10.1007/s00726-013-1574-1
Obakan P, Arisan ED, Coker-Gurkan A, Palavan-Unsal N (2014b) Epibrassinolide-induced apoptosis regardless of p53 expression via activating polyamine catabolic machinery, a common target for androgen sensitive and insensitive prostate cancer cells. Prostate 74(16):1622–1633. https://doi.org/10.1002/pros.22879
Obakan P, Barrero C, Coker-Gurkan A, Arisan ED, Merali S, Palavan-Unsal N (2015) SILAC-based mass spectrometry analysis reveals that epibrassinolide induces apoptosis via activating endoplasmic reticulum stress in prostate cancer cells. PLoS ONE 10(9):e0135788. https://doi.org/10.1371/journal.pone.0135788
Onal G, Kutlu O, Gozuacik D, Dokmeci Emre S (2017) Lipid droplets in health and disease. Lipids Health Dis 16(1):128. https://doi.org/10.1186/s12944-017-0521-7
Pincus D, Chevalier MW, Aragon T, van Anken E, Vidal SE, El-Samad H, Walter P (2010) BiP binding to the ER-stress sensor Ire1 tunes the homeostatic behavior of the unfolded protein response. PLoS Biol 8(7):e1000415. https://doi.org/10.1371/journal.pbio.1000415
Pyo JO, Jang MH, Kwon YK, Lee HJ, Jun JI, Woo HN, Cho DH, Choi B, Lee H, Kim JH, Mizushima N, Oshumi Y, Jung YK (2005) Essential roles of Atg5 and FADD in autophagic cell death: dissection of autophagic cell death into vacuole formation and cell death. J Biol Chem 280(21):20722–20729. https://doi.org/10.1074/jbc.M413934200
Pyo JO, Nah J, Kim HJ, Lee HJ, Heo J, Lee H, Jung YK (2008) Compensatory activation of ERK1/2 in Atg5-deficient mouse embryo fibroblasts suppresses oxidative stress-induced cell death. Autophagy 4(3):315–321
Rueden CT, Schindelin J, Hiner MC, DeZonia BE, Walter AE, Arena ET, Eliceiri KW (2017) Image J2: ImageJ for the next generation of scientific image data. BMC Bioinform 18(1):529. https://doi.org/10.1186/s12859-017-1934-z
Sacitharan PK, Lwin S, Gharios GB, Edwards JR (2018) Spermidine restores dysregulated autophagy and polyamine synthesis in aged and osteoarthritic chondrocytes via EP300. Exp Mol Med 50(9):123. https://doi.org/10.1038/s12276-018-0149-3
Sano R, Reed JC (2013) ER stress-induced cell death mechanisms. Biochim Biophys Acta 1833(12):3460–3470. https://doi.org/10.1016/j.bbamcr.2013.06.028
Shanware NP, Bray K, Abraham RT (2013) The PI3K, metabolic, and autophagy networks: interactive partners in cellular health and disease. Annu Rev Pharmacol Toxicol 53:89–106. https://doi.org/10.1146/annurev-pharmtox-010611-134717
Song C, Mitter SK, Qi X, Beli E, Rao HV, Ding J, Ip CS, Gu H, Akin D, Dunn WA Jr, Bowes Rickman C, Lewin AS, Grant MB, Boulton ME (2017) Oxidative stress-mediated NFkappaB phosphorylation upregulates p62/SQSTM1 and promotes retinal pigmented epithelial cell survival through increased autophagy. PLoS ONE 12(2):e0171940. https://doi.org/10.1371/journal.pone.0171940
Song S, Tan J, Miao Y, Zhang Q (2018) Crosstalk of ER stress-mediated autophagy and ER-phagy: involvement of UPR and the core autophagy machinery. J Cell Physiol 233(5):3867–3874. https://doi.org/10.1002/jcp.26137
Steigerova J, Oklestkova J, Levkova M, Rarova L, Kolar Z, Strnad M (2010) Brassinosteroids cause cell cycle arrest and apoptosis of human breast cancer cells. Chem Biol Interact 188(3):487–496. https://doi.org/10.1016/j.cbi.2010.09.006
Steigerova J, Rarova L, Oklest'kova J, Krizova K, Levkova M, Svachova M, Kolar Z, Strnad M (2012) Mechanisms of natural brassinosteroid-induced apoptosis of prostate cancer cells. Food Chem Toxicol 50(11):4068–4076. https://doi.org/10.1016/j.fct.2012.08.031
Tong D, Hill JA (2017) Spermidine promotes cardioprotective autophagy. Circ Res 120(8):1229–1231. https://doi.org/10.1161/CIRCRESAHA.117.310603
Vanhaesebroeck B, Alessi DR (2000) The PI3K–PDK1 connection: more than just a road to PKB. Biochem J 346(Pt 3):561–576
Vanrell MC, Cueto JA, Barclay JJ, Carrillo C, Colombo MI, Gottlieb RA, Romano PS (2013) Polyamine depletion inhibits the autophagic response modulating Trypanosoma cruzi infectivity. Autophagy 9(7):1080–1093. https://doi.org/10.4161/auto.24709
Wei Y, Pattingre S, Sinha S, Bassik M, Levine B (2008) JNK1-mediated phosphorylation of Bcl-2 regulates starvation-induced autophagy. Mol Cell 30(6):678–688. https://doi.org/10.1016/j.molcel.2008.06.001
Xie J, Proud CG (2014) Signaling crosstalk between the mTOR complexes. Translation (Austin) 2(1):e28174
Yang L, Li P, Fu S, Calay ES, Hotamisligil GS (2010) Defective hepatic autophagy in obesity promotes ER stress and causes insulin resistance. Cell Metab 11(6):467–478. https://doi.org/10.1016/j.cmet.2010.04.005
Yue W, Yang CS, DiPaola RS, Tan XL (2014) Repurposing of metformin and aspirin by targeting AMPK-mTOR and inflammation for pancreatic cancer prevention and treatment. Cancer Prev Res (Phila) 7(4):388–397. https://doi.org/10.1158/1940-6207.CAPR-13-0337
Zahedi K, Barone S, Destefano-Shields C, Brooks M, Murray-Stewart T, Dunworth M, Li W, Doherty JR, Hall MA, Smith RD, Cleveland JL, Casero RA Jr, Soleimani M (2017) Activation of endoplasmic reticulum stress response by enhanced polyamine catabolism is important in the mediation of cisplatin-induced acute kidney injury. PLoS ONE 12(9):e0184570. https://doi.org/10.1371/journal.pone.0184570
Acknowledgements
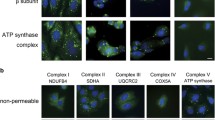
This study was supported by Istanbul Kultur University. We are thankful for technical assistance to Mert Meseli, Dilay Burmabiyikoglu and Tugce Buse Gormek for Fig. 6a. All authors interpreted the data and were involved in the writing of the manuscript.
Author information
Authors and Affiliations
Contributions
KA and RIK carried out the experiments. The project was designed by POY, EDA, ACG, and NPU. All authors prepared the manuscript and interpreted data.
Corresponding author
Ethics declarations
Conflict of interest
No conflict of interest was declared by the authors.
Ethical statement
As we are the authors of this study, we declare that there is no data given obtained from human participants or animals. We all read and confirmed the final version of the manuscript.
Additional information
Handling editor: E. Agostinelli.
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Electronic supplementary material
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Adacan, K., Obakan-Yerlikaya, P., Arisan, E.D. et al. Epibrassinolide-induced autophagy occurs in an Atg5-independent manner due to endoplasmic stress induction in MEF cells. Amino Acids 52, 871–891 (2020). https://doi.org/10.1007/s00726-020-02857-w
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00726-020-02857-w