Abstract
Objectives
This study was performed to characterise oral shedding of herpesviruses in patients who underwent allogeneic hematopoietic stem cell transplantation (alloHSCT) and to investigate its relationship with oral mucositis (OM).
Materials and Methods
PCR and enzymatic digestion were conducted to identify oral shedding of herpesviruses and its correlation with OM development in 31 patients. The samples were collected at three sites in the oral cavity and at 5 times during follow-up; two additional collections were made from patients who developed ulcerative OM.
Results
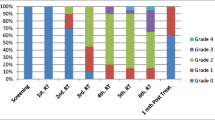
HSV-1, EBV, CMV, HHV-6A, HHV-6B, and HHV-7 were detected in 4.97%, 16.02%, 4.41%, 2.20%, 3.31%, and 68% of the oral mucosal samples, respectively; 4.41%, 16.57%, 5.52%, 2.20%, 5.52%, and 63.53% of supragingival samples, respectively, and 4.41%, 18.23%, 2.76%, 1.65%, 2.75%, and 35.91% of subgingival samples, respectively. OM was diagnosed in 13 patients. The presence of HHV-7 in C1 (oral mucosa: p = 0.032) and C2 (supragingival: p = 0.009; subgingival: p = 0.002) was significantly increased in patients who developed OM, and patients exhibiting HHV-7 shedding in the oral cavity were 3.32-fold more likely to develop OM.
Conclusions
Patients who developed OM showed higher HHV-7 shedding in the oral cavity at nadir (immediately prior to OM development), suggesting modifications to the inflammatory microenvironment.
Clinical Relevance
HHV-7 may be involved in oral dysbiosis in HSCT-related OM; enhanced understanding of its role in the pathogenesis of OM may lead to the development of strategies for managing and preventing this common side effect of alloHSCT.



Similar content being viewed by others
Abbreviations
- alloHSCT:
-
allogenic hematopoietic stem cell transplantation
- CMV:
-
Cytomegalovirus
- EBV:
-
Epstein-Barr virus
- HHV-6:
-
Human herpesvirus 6
- HHV-7:
-
Human herpesvirus 7
- HHV-8:
-
Human herpesvirus 8
- HSV-1:
-
Herpes simplex virus 1
- HSV-2:
-
Herpes simplex virus 2
- OM:
-
Oral mucositis
- RIC:
-
Reduced-intensity conditioning
- VZV:
-
Varicella Zoster virus
- WHO:
-
World Health Organization
References
Majhail NS, Farnia SH, Carpenter PA, Champlin RE, Crawford S, Marks DI, Omel JL, Orchard PJ, Palmer J, Saber W, Savani BN, Veys PA, Bredeson CN, Giralt SA, LeMaistre C (2015) Indications for autologous and allogeneic hematopoietic cell transplantation: guidelines from the American Society for Blood and Marrow Transplantation. Biol Blood Marrow Transplant 21:1863–1869. https://doi.org/10.1016/j.bbmt.2015.07.032
Sureda A, Bader P, Cesaro S, Dreger P, Duarte RF, Dufour C, Falkenburg JH, Farge-Bancel D, Gennery A, Kröger N, Lanza F, Marsh JC, Nagler A, Peters C, Velardi A, Mohty M, Madrigal A (2015) Indications for allo- and auto-SCT for haematological diseases , solid tumours and immune disorders: current practice in Europe. Bone Marrow Transplant 50:1037–1056. https://doi.org/10.1038/bmt.2015.6
Bezinelli LM, Eduardo FDP, Lopes RM d G, MGH B, CDP E, Correa L, Hamerschlak N et al (2014) Cost-effectiveness of the introduction of specialized oral care with laser therapy in hematopoietic stem cell transplantation. Hematol Oncol 32:31–39. https://doi.org/10.1002/hon
Villa A, Sonis ST (2015) Mucositis: pathobiology and management. Curr Opin Oncol 27:159–164. https://doi.org/10.1097/CCO.0000000000000180
Bowen J, Al-Dasooqi N, Bossi P, Wardill H, Van Sebille Y, Al-Azri A et al (2019) The pathogenesis of mucositis: updated perspectives and emerging targets. Support Care Cancer 27:4023–4033. https://doi.org/10.1007/s00520-019-04893-z
Al-Dasooqi N, Sonis ST, Bowen JM, Bateman E, Blijlevens N, Gibson RJ et al (2013) Emerging evidence on the pathobiology of mucositis. Support Care Cancer 21:2075–2083. https://doi.org/10.1007/s00520-013-1810-y
Silva LC, Sacono NT, Freire M d CM, Costa LR, Batista AC, GBL S (2015) The impact of low-level laser therapy on Oral Mucositis and quality of life in patients undergoing hematopoietic stem cell transplantation using the oral health impact profile and the functional assessment of cancer therapy-bone marrow transplantation questionnaires. Photomed Laser Surg 33:357–363. https://doi.org/10.1089/pho.2015.3911
Silva GBL, Mendonça EF, Bariani C, Antunes HS, Silva MAG (2011) The prevention of induced oral mucositis with low-level laser therapy in bone marrow transplantation patients: a randomized clinical trial. Photomed Laser Surg 29:27–31. https://doi.org/10.1089/pho.2009.2699
Zadik Y, Arany PR, Fregnani ER, Bossi P, Antunes HS, Bensadoun R-J, Gueiros LA, Majorana A, Nair RG, Ranna V, Tissing WJE, Vaddi A, Lubart R, Migliorati CA, Lalla RV, Cheng KKF, Elad S, On behalf of The Mucositis Study Group of the Multinational Association of Supportive Care in Cancer/International Society of Oral Oncology (MASCC/ISOO) (2019) Systematic review of photobiomodulation for the management of oral mucositis in cancer patients and clinical practice guidelines. Support Care Cancer 27:3969–3983. https://doi.org/10.1007/s00520-019-04890-2
Vagliano L, Feraut C, Gobetto G, Trunfio A, Errico A, Campani V et al (2011) Incidence and severity of oral mucositis in patients undergoing haematopoietic SCT — results of a multicentre study. Bone Marrow Transplant 46:727–732. https://doi.org/10.1038/bmt.2010.184
Robien K, Schubert MM, Bruemmer B, Lloid ME, Potter JD, Ulrich CM (2004) Predictors of Oral Mucositis in patients receiving hematopoietic cell transplants for chronic myelogenous leukemia. J Clin Oncol 22:1268–1275. https://doi.org/10.1200/JCO.2004.05.147
De Pagter PJA, Schuurman R, Keukens L, Schutten M, Cornelissen JJ, Van Baarle D et al (2013) Human herpes virus 6 reactivation: important predictor for poor outcome after myeloablative, but not non-myeloablative allo-SCT. Bone Marrow Transplant 48:1460–1464. https://doi.org/10.1038/bmt.2013.78
Gomes de Oliveira PGA, Ueda MYH, Real JM, De Sá ME, Rodrigues De Oliveira JS, Gonçalves MV et al (2016) Simultaneous quantification of the 8 human herpesviruses in allogeneic hematopoietic stem cell transplantation. Transplantation 100:1363–1370. https://doi.org/10.1097/TP.0000000000000986
Nicolatou-Galitis O, Athanassiadou P, Kouloulias V, Sotiropoulou-Lontou A, Polychronopoulou A, Gonidi M et al (2006) Herpes simplex virus-1 (HSV-1) infection in radiation-induced oral mucositis. Support Care Cancer 14:753–762. https://doi.org/10.1007/s00520-005-0006-5
Chen YK, Hou HA, Chow JM, Chen YC (2011) The impact of oral herpes simplex virus infection and candidiasis on chemotherapy-induced oral mucositis among patients with hematological malignancies. Eur J Microbiol Infect Dis 30:753–759. https://doi.org/10.1007/s10096-010-1148-z
Correa Sierra CB, Kourí Cardellá V, Pérez Santos L, Silverio CE, Hondal N, Florin J (2017) Herpesviruses excretion in saliva of pediatric transplant recipients. Transpl Infect Dis 19:2–10. https://doi.org/10.1111/tid.12771
Palmieri M, Martins VA d O, Sumita LM, Tozetto-Mendoza TR, Romano BB, Machado CM et al (2017) Oral shedding of human herpesviruses in patients undergoing radiotherapy/chemotherapy treatment for head and neck squamous cell carcinoma. Clin Oral Investig 21:2291–2301. https://doi.org/10.1007/s00784-016-2022-x
World Health Organization. (1979) Toxic effects. In: WHO Handbook for Reporting Results of |Cancer Treatment, WHO Offset Publication,Geneve, pp 14-21
Wu M, Huang F, Jiang X, Fan Z, Zhou H, Liu C et al (2013) Herpesvirus-associated central nervous system diseases after allogeneic hematopoietic stem cell transplantation. PLoS One 8:1e77805. https://doi.org/10.1371/journal.pone.0077805
Quintela A, Escuret V, Roux S, Bonnafous P, Gilis L, Barraco F et al (2016) HHV-6 infection after allogeneic hematopoietic stem cell transplantation: from chromosomal integration to viral co-infections and T-cell reconstitution patterns. J Inf Secur 72:214–222. https://doi.org/10.1016/j.jinf.2015.09.039
Olson AL, Dahi PB, Zheng J, Devlin SM, Lubin M, Gonzales AM, Giralt SA, Perales MA, Papadopoulos EB, Ponce DM, Young JW, Kernan NA, Scaradavou A, O'Reilly RJ, Small TN, Papanicolaou G, Barker JN (2014) Frequent human herpesvirus-6 viremia but low incidence of encephalitis in double-unit cord blood recipients transplanted without anti-thymocyte globulin. Biol Blood Marrow Transplant 20:787–793. https://doi.org/10.1016/j.cgh.2008.07.016.Cytokeratin
Inazawa N, Hori T, Hatakeyama N, Yamamoto M, Yoto Y, Nojima M, Suzuki N, Shimizu N, Tsutsumi H (2015) Large-scale multiplex polymerase chain reaction assay for diagnosis of viral reactivations after allogeneic hematopoietic stem cell transplantation. J Med Virol 87:1427–1435. https://doi.org/10.1002/jmv
Pereira CM, De Almeida OP, Corrêa MEP, Costa FF, De Souza C, Barjas-Castro ML (2007) Detection of human herpesvirus 6 in patients with oral chronic graft-vs-host disease following allogeneic progenitor cell transplantation. Oral Dis 13:329–334. https://doi.org/10.1111/j.1601-0825.2006.01294.x
Sarmento DJ d S, Tozetto-Mendoza TR, Sumita LM, Pierroti, Pallos D, Caliento R, Palmieri M, Martins VA d O et al (2017) Oral shedding of human herpesviruses in renal transplant recipients. Clin Oral Investig 9:e12356. https://doi.org/10.1007/s00784-017-2166-3
Van Der Beek MT, Laheij AMGA, Raber-Durlacher JE, Von Dem Borne PA, Wolterbeek R, Van Der Blij-De Brouwer CS et al (2012) Viral loads and antiviral resistance of herpesviruses and oral ulcerations in hematopoietic stem cell transplant recipients. Bone Marrow Transplant 47:1222–1228. https://doi.org/10.1038/bmt.2012.2
Lantzman E, Michman J (1970) Leukocyte counts in the saliva of adults before and after extraction of teeth. Oral Surg 30:766–773
Theda C, Hwang SH, Czajko A, Loke YJ, Leong P, Craig JM (2018) Quantitation of the cellular content of saliva and buccal swab samples. Sci Rep 8:4–11. https://doi.org/10.1038/s41598-018-25311-0
Wilborn BF, Brinkmann V, Schmidt CA, Neipel F, Gelderblom H, Siegert W (1994) Herpesvirus type 6 in patients undergoing bone marrow transplantation: serologic features and detection by polymerase chain reaction. Am Soc Hematol 83:3052–3059
Pereira CM, Gasparetto PF, Corrêa MEP, Costa FF, De Almeida OP, Barjas-Castro ML (2004) Human herpesvirus 6 in oral fluids from healthy individuals. Arch Oral Biol 49:1043–1046. https://doi.org/10.1016/j.archoralbio.2004.06.002
Zerr DM, Huang M, Corey L, Erickson M, Parker HL, Frenkel LM (2000) Sensitive method for detection of human herpesviruses 6 and 7 in saliva collected in field studies. J Clin Microbiol 38:1981–1983
de Mendonça RMH, De Araújo M, Levy CE, Morari J, Silva AR, Yunes AJ et al (2015) Oral mucositis in pediatric acute lymphoblastic leukemia patients: evaluation of microbiological and hematological factors. Pediatr Hematol Oncol 32:322–330. https://doi.org/10.3109/08880018.2015.1034819
Tang F-F, Zhao X-S, Xu L-P, Zhang X-H, Chen Y-H, Mo X-D, Sun YQ, Liu KY, Huang XJ (2017) Risk factors for herpes simplex virus-1/2 viremia and clinical outcomes following unmanipulated haploidentical haematopoietic stem cell transplantation. J Clin Virol 95:20–25. https://doi.org/10.1016/j.jcv.2017.07.018
Pankratova OS, Chukhlovin AB, Shiryaev SN, Eismont YA, Vavilov VN, Zubarovskaya LS, Afanasyev BV (2013) Herpesviruses and oral ulcerations in hematopoietic SCT recipients. Bone Marrow Transplant 48:1364–1365. https://doi.org/10.1038/bmt.2013.74
Correia AVL, Coêlho MRCD, Cahú GG d OM, Silva JL d A, Brasil C d MV, de Castro JFL (2014) Seroprevalence of HSV-1 / 2 and correlation with aggravation of oral mucositis in patients with squamous cell carcinoma of the head and neck region submitted to antineoplastic treatment. Support Care Cancer 23:2105–2111. https://doi.org/10.1007/s00520-014-2558-8
Djuric M, Jankovic L, Jovanovic T, Pavlica D, Brkic S, Knezevic A, Markovic D, Milasin J (2009) Prevalence of oral herpes simplex virus reactivation in cancer patients: a comparison of different techniques of viral detection. J Oral Pathol Med 38:167–173. https://doi.org/10.1111/j.1600-0714.2008.00684.x
Kawamura K, Wada H, Yamasaki R, Ishihara Y, Sakamoto K, Ashizawa M, Sato M, Machishima T, Terasako K, Kimura SI, Kikuchi M, Nakasone H, Yamazaki R, Kanda J, Kako S, Tanihara A, Nishida J, Kanda Y (2013) Low-dose acyclovir prophylaxis for the prevention of herpes simplex virus disease after allogeneic hematopoietic stem cell transplantation. Transpl Infect Dis 15:457–465. https://doi.org/10.1111/tid.12118
Taplitz RA, Kennedy EB, Bow EJ, Crews J, Gleason C, Hawley DK et al (2019) Antimicrobial prophylaxis for adult patients with cancer- related immunosuppression: ASCO and IDSA clinical practice guideline update. J Clin Oncol 36:3043–3054. https://doi.org/10.1200/JCO.18.00374
Saral R, Ambinder RF, Burns WH, Angelopulos CM, Griffin DE, Burke PJ, Lietman PS (1983) Acyclovir prophylaxis against herpes simplex virus infection in patients with leukemia. Ann Intern Med 99:773–776
Diaz PI, Valm AM (2019) Microbial interactions in oral communities mediate emergent biofilm properties. J Dent Res 99:18–25. https://doi.org/10.1177/0022034519880157
Kiedrowski MR, Gaston JR, Kocak BR, Coburn SL, Lee S, Pilewski JM et al (2018) Staphylococcus aureus biofilm growth on cystic fibrosis airway epithelial cells is enhanced during respiratory syncytial virus coinfection. MSphere 3:e00341–e00318. https://doi.org/10.1128/mSphere.00341-18
Slots J (2010) Human viruses in periodontitis. Periodontology 53:89–110. https://doi.org/10.1111/j.1600-0757.2009.00325.x
Marques Filho JS, Jr JG, Vargas G, Sumita LM, Shibli JA, Viana RG et al (2018) Clinical study cytokine levels and human herpesviruses in saliva from clinical periodontal healthy subjects with peri-implantitis: a case-control study. Mediat Inflamm 2018:1–7. https://doi.org/10.1155/2018/6020625
Caliento R, de Santana Sarmento DJ, Silva ÈMP, Tozetto-Mendoza TR, Tobouti PL, Benini V et al (2018) Oral shedding ogf HSV1 and EBV and oral manifestations in paediatric chronic kidney disease patients and renal transplant recipients. Acta Odontol Scand 76:539–544
Funding sources
This study was supported by grants from Sao Paulo Research Foundation – FAPESP, Sao Paulo, Brazil [grant number 2015/07727–9], National Council for Scientific and Technology Development (CNPq) [grant number 443004/2014–5], and Coordination for the Improvement of Higher Education Personnel (CAPES).
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interests
The authors declared that they have no conflict of interests.
Ethical approval
This study was approved by the local ethics committee (N° 1.414.217) and conducted in accordance with the Declaration of Helsinki.
Informed consent
All patients of this study who agreed to participate with this study signed the informed consent term.
Additional information
Publisher’s note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Miranda-Silva, W., Knebel, F.H., Tozetto-Mendozo, T.R. et al. Herpesviruses in the oral cavity of patients subjected to allogeneic hematopoietic stem cell transplantation and its relationship with oral mucositis. Clin Oral Invest 24, 3597–3608 (2020). https://doi.org/10.1007/s00784-020-03234-3
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00784-020-03234-3