Abstract
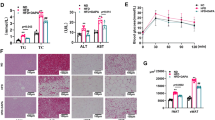
This study evaluated the anti-adipogenic effects and mechanisms underlying the action of Lactobacillus fermentum MG4231 and MG4244 strains on adipogenesis and lipid accumulation in 3T3-L1 preadipocytes. Treatment with cell-free extracts (CFEs) from the two strains reduced lipid accumulation and intracellular triglyceride production in 3T3-L1 adipocytes by more than 50%. The inhibitory effects of L. fermentum on lipid accumulation were mediated by the downregulation of FAS and aP2 resulting from the inhibition of PPARγ and C/EBPα gene expression. Moreover, AMPK and HSL phosphorylation was upregulated by CFE treatment. These results indicated that the anti-adipogenic and lipolysis activities of L. fermentum strains were caused by increased AMPK and HSL phosphorylation. Both strains displayed high leucine arylamidase and β-galactosidase enzymatic activity, with excellent adhesion to epithelial cells. Therefore, we identified L. fermentum as potential new probiotics for the prevention of obesity.



Similar content being viewed by others
Abbreviations
- CFE:
-
Cell-free extract
- DEX:
-
Dexamethasone
- IBMX:
-
Isobutylmethylxanthine
- DMEM:
-
Dulbecco modified Eagle’s medium
- FBS:
-
Fetal bovine serum
- TG:
-
Triglyceride
- MTT:
-
Tetrazolium bromide salt
- PPARγ:
-
Peroxisome proliferator-activated receptor γ
- C/EBPα:
-
CCAAT/enhancer-binding protein α
- FAS:
-
Fatty acid synthase
- aP2:
-
Adipose-specific fatty acid-binding protein
- SREBP1:
-
Sterol regulatory element-binding protein 1
- LPL:
-
Lipoprotein lipase
- ACC:
-
Acetyl CoA carboxylase
- AMPK:
-
AMP-activated protein kinase
- p-AMPK:
-
Phospho-AMP-activated protein kinase
- HSL:
-
Hormone-sensitive lipase
- p-HSL:
-
Phospho-hormone- sensitive lipase
References
Bäumler AJ, Sperandio V. Interactions between the microbiota and pathogenic bacteria in the gut. Nature 535: 85-93 (2016)
Blüher M. Obesity: global epidemiology and pathogenesis. Nat. Rev. Endocrinol. 15: 288-298 (2019)
Brun RP, Kim JB, Hu E, Altiok S, Spiegelman BM. Adipocyte differentiation: a transcriptional regulatory cascade. Curr. Opin. Cell Biol. 8: 826-832 (1996)
Carmen GY, Víctor SM. Signalling mechanisms regulating lipolysis. Cell Signal. 18: 401-408 (2006)
Castro-Gonzalez JM, Castro P, Sandoval H, Castro-Sandoval D. Probiotic Lactobacilli precautions. Front Microbiol. 10: 375 (2019)
Colombo M, Castilho NPA, Todorov SD, Nero LA. Beneficial properties of lactic acid bacteria naturally present in dairy production. BMC Microbiol. 18: 219 (2018)
Djouder N, Tuerk RD, Suter M, Salvioni P, Thali RF, Scholz R, Vaahtomeri K, Auchli Y, Rechsteiner H, Brunisholz RA, Viollet B, Makela TP, Wallimann T, Neumann D, Krek W. PKA phosphorylates and inactivates AMPK alpha to promote efficient lipolysis. EMBO J. 29: 469-481 (2010)
Endo H, Niioka M, Kobayashi N, Tanaka M, Watanabe T. Butyrate-producing probiotics reduce nonalcoholic fatty liver disease progression in rats: new insight into the probiotics for the gut-liver axis. PLoS ONE. 8: e63388 (2013)
Ghaben AL, Scherer PE. Adipogenesis and metabolic health. Nat. Rev. Mol. Cell Biol. 20: 242-258 (2019)
Gioacchini G, Rossi G, Camevali O. Host-probiotic interaction: new insight into the role of the endocannabinoid system by in vivo and ex vivo approaches. Sci. Rep. 7: 1261 (2017)
Gregoire FM, Smas CM, Sul HS. Understanding adipocyte differentiation. Physiol. Rev. 78: 783-809 (1998)
Husain Q. Beta galactosidases and their potential applications: a review. Crit. Rev. Biotechnol. 30: 41-62 (2010)
Iwamoto K, Kamo S, Takada Y, Ieda A, Yamashita T, Sato T, Zaima N, Moriyama T. Soyasapogenols reduce cellular triglyceride levels in 3T3-L1 mouse adipocyte cells by accelerating triglyceride lipolysis. Biochem. Biophys. Rep. 16: 44-49 (2018)
Jang HM, Han SK, Kim JK, Oh SJ, Jang HB, Kim DH. Lactobacillus sakei alleviates high-fat-diet-induced obesity and anxiety in mice by inducing AMPK activation and SIRT1 expression and inhibiting gut microbiota-mediated NF-κB activation. Mol. Nutr. Food Res. 63: e1800978 (2019)
Kang CH, Jeong YA, Han SH, Kim JS, Kim YG, Park HM, Choi SI, Paek NS. In vitro probiotic evaluation of potential antiobesity lactic acid bacteria isolated from human vagina and shellfish. Biotechnol. Bioprocess. Eng. 33: 161-167 (2018)
Kerry RG, Patra JK, Gouda S, Park Y, Shin HS, Das G. Benefaction of probiotics for human health: a review. J. Food Drug Anal. 26: 927-939 (2018)
Kim S, Huang E, Park S, Holzapfel W, Lim SD. Physiological characteristics and anti-obesity effect of Lactobacillus plantarum K10. Korean J. Food Sci. Anim. 38: 554-569 (2018)
Kleerebezem M, Hols P, Bernard E, Rolain T, Zhou M, Siezen RJ, Bron PA. The extracellular biology of the lactobacilli. FEMS Microbiol. Rev. 34: 199-230 (2010)
Kobyliak N, Conte C, Cammarota G, Haley AP, Styriak I, Gaspar L, Fusek J, Rodrigo L, Kruzliak P. Probiotics in prevention and treatment of obesity: a critical view. Nutr. Metab. 13: 14 (2016)
Lazar AD, Dinescu S, Costache M. Adipose tissue engineering and adipogenesis: a review. Rev. Biol. Biomed. Sci. 1: 17-26 (2018)
Lee SJ, Depoortere I, Hatt H. Therapeutic potential of ectopic olfactory and taste receptors. Nat. Rev. Drug Discov. 18: 116-138 (2019)
Mandard S, Müller M, Kersten S. Peroxisome proliferator-activated receptor alpha target genes. Cell. Mol. Life Sci. 61: 393-416 (2004)
Miyoshi M, Ogawa A, Higurashi S, Kadooka Y. Anti-obesity effect of Lactobacillus gasseri SBT2055 accompanied by inhibition of pro-inflammatory gene expression in the visceral adipose tissue in diet-induced obese mice. Eur. J. Nutr. 53: 599-606 (2014)
Mota de Sá P, Richard AJ, Hang H, Stephens JM. Transcriptional regulation of adipogenesis. Compr. Physiol. 7: 635-674 (2017)
Nagpal R, Kumar A, Kumar M, Behare PV, Jain S, Yadav H. Probiotics, their health benefits and applications for developing healthier foods: a review. FEMS Microbiol. Lett. 334: 1-15 (2012)
Ogawa A, Kobayashi T, Sakai F, Kadooka Y, Kawasaki Y. Lactobacillus gasseri SBT2055 suppresses fatty acid release through enlargement of fat emulsion size in vitro and promotes fecal fat excretion in healthy Japanese subjects. Lipids Health Dis. 14: 20 (2015)
Park DY, Ahn YT, Huh CS, Jeon SM, Choi MS. The inhibitory effect of Lactobacillus plantarum KY1032 cell extract on the adipogenesis of 3T3-L1 cells. J. Med. Food 14: 670-675 (2011)
Park SS, Lee YJ, Song S, Kim B, Kang H, Oh S, Kim E. Lactobacillus acidophilus NS1 attenuates diet-induced obesity and fatty liver. J. Endocrinol. 237: 87-100 (2018)
Patten DA, Laws AP. Lactobacillus-produced exopolysaccharides and their potential health benefits: A review. Benef. Microbes. 6: 457-471 (2015)
Plaza-Diaz J, Ruiz-Ojeda FJ, Gil-Campos M, Gil A. Mechanisms of action of probiotics. Adv. Nutr.10: S49-S66 (2019)
Polak-Berecka M, Waśko A, Paduch R, Skrzypek T, Sroka-Bartnicka A. The effect of cell surface components on adhesion ability of Lactobacillus rhamnosus. Anton. Leeuw. Int. J. G. 106: 751–762 (2014)
Prusty D, Park BF, Davis KE, Farmer SR. Activation of MEK/ERK signaling promotes adipogenesis by enhancing peroxisome proliferator-activated receptor γ (PPARγ) and C/EBPα gene expression during the differentiation of 3T3-L1 preadipocytes. J. Biol. Chem. 277: 46226-46232 (2002)
Rapold RA, Wueest S, Knoepfel A, Schoenle EJ, Konrad D. Fas activates lipolysis in a Ca2+-CaMKII-dependent manner in 3T3-L1 adipocytes. J. Lipid Res. 54: 63-70 (2013)
Ricci A, Levante A, Cirlini M, Calani L, Bernini V, Rio DD, Galaverna G, Neviani E, Lazzi C. The influence of viable cells and cell-free extracts of Lactobacillus casei on volatile compounds and polyphenolic profile of elderberry juice. Front Microbiol. 9: 2784 (2018)
Rizzatti V, Boschi F, Pedrotti M, Zoico E, Sbarbati A, Zamboni M. Lipid droplets characterization in adipocyte differentiated 3T3-L1 cells: size and optical density distribution. Eur. J. Histochem. 57: e24 (2013)
Rosen E, Eguchi J, Xu Z. Transcriptional targets in adipocyte biology. Expert Opin. Ther. Target 13: 975-986 (2009)
Saadatzadeh A, Fazeli MR, Jamalifar H, Dinarvand R. Probiotic properties of lyophilized cell free extract of Lactobacillus casei. Jundishapur J. Nat. Pharm. Prod. 8: 131-137 (2013)
Schwartz M, Seeley RJ, Zeltser LM, Drewnowski AD, Ravussin E, Redman LM, Leibel RL. Obesity pathogenesis: an endocrine society scientific statement. Endocr. Rev. 38: 267-296 (2017)
Seo JW, Yang HJ, Jeong SJ, Ryu MS, Ha G, Jeong SY, Jeong DY. Characterization of Lactobacillus brevis SCML 432 isolated from Meju in Sunchang and optimization of its culture conditions by statistical methods. Korean J. Food Preserv. 25: 397-410 (2018)
Smid EJ, Kleerebezem M. Production of aroma compounds in lactic fermentations. Annu. Rev. Food Sci. Technol. 5: 313-326 (2014)
Wlodarczyk M, Nowicka G. Obesity, DNA damage, and development of obesity-related diseases. Int. J. Mol. Sci. 20: 1146 (2019)
Wu CC, Weng WL, Lai WL, Tsai HP, Liu WH, Lee MH, Tsai YC. Effect of Lactobacillus plantarum strain K21 on high-fat diet-fed obese mice. Evid-Based Complem. Alter 2015: 391767 (2015)
Zhang L, Li N, Caicedo R., Neu J. Alive and dead Lactobacillus rhamnosus GG decrease tumor necrosis factor-α-induced interleukin-8 production in Caco-2 cells. J. Nutr. 135: 1752-1756 (2005).
Zhang Z, Zhou Z, Li Y, Zhou L, Ding Q, Xu L. Isolated exopolysaccharides from Lactobacillus rhamnosus GG alleviated adipogenesis mediated by TLR2 in mice. Sci. Rep. 6: 36083 (2016)
Zommiti M, Connil N, Hamida JB, Ferchichi M. Probiotic characteristics of Lactobacillus curvatus DN317, a strain isolated from chicken ceca. Probiotics Antimicrob. 9: 415-424 (2017)
Acknowledgements
This work was supported by a project for Collabo R&D between Industry, Academy, and Research Institute funded by the Korea Ministry of SMEs and Startups in 2019 (Project No. S2717946).
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
The authors declare that they have no conflict of interest.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Kim, S., Choi, SI., Jang, M. et al. Anti-adipogenic effect of Lactobacillus fermentum MG4231 and MG4244 through AMPK pathway in 3T3-L1 preadipocytes. Food Sci Biotechnol 29, 1541–1551 (2020). https://doi.org/10.1007/s10068-020-00819-2
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10068-020-00819-2