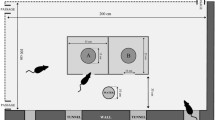
Abstract
In rodents, defensive behaviors increase the chances of survival during a predator encounter. Observable rodent defensive behaviors have been shown to be influenced by the presence of predator odors and nearby environmental cues such as cover, odors from conspecifics and food availability. Our experiment tested whether a predator scent cue influenced refuge preference in meadow voles within a laboratory setting. We placed voles in an experimental apparatus with bedding soaked in mink scent versus olive oil as a control across from four tubes that either contained (a) a dark plastic covering, (b) opposite-sex conspecific odor, (c) a food pellet, or (d) an empty, unscented space. A three-way interaction of tube contents, subject sex, and the presence of mink or olive oil on the preference of meadow voles to spend time in each area of the experimental apparatus and their latency to enter each area of the apparatus revealed sex differences in the environmental preference of meadow voles facing the risk of predation. The environmental preference of female, but not male, meadow voles was altered by the presence of mink urine or olive oil. A similar trend was found in the latency of males and females to enter each area of the experimental apparatus. These differences suggest that each sex utilizes different methods to increase their fitness when experiencing a predation risk. The observed sex differences may be explained by the natural history of voles owing to the differences in territorial range and the dynamics of evasion of terrestrial predators.



Similar content being viewed by others
Availability of data and material
The corresponding author can make the data collected during the completion of this experiment available upon, reasonable, individual request.
Code availability
The data utilized to obtain the results of this experiment were analyzed in R 4.0.2 (R Core Team, 2020) and RStudio 1.3.1093 (RStudio Team, 2020). The specific code used to analyze this data can be made available upon individual request.
References
Abramsky Z, Strauss E, Subach A, Riechman A, Kotler BP (1996) The effect of barn owls (Tyto alba) on the activity and microhabitat selection of Gerbillus allenbyi and G. pyramidum. Oecol 105:313–319. https://doi.org/10.1007/BF00328733
Apfelbach R, Blanchard CD, Blanchard RJ, Hayes RA, McGregor IS (2005) The effects of predator odors in mammalian prey species: a review of field and laboratory studies. Neurosci Biobehav Rev 29:1123–1144. https://doi.org/10.1016/j.neubiorev.2005.05.005
Arnold TW, Fritzell EK (1987) Food habits of prairie mink during the waterfowl breeding season. Can J Zool 65(9):2322–2324. https://doi.org/10.1139/z87-349
Boonstra R, Xia X, Pavone L (1993) Mating system of the meadow vole, Microtus pennsylvanicus. Behav Ecol 4:83–89. https://doi.org/10.1093/beheco/4.1.83
Bourin M, Hascoet M (2003) The mouse light/dark box test. Eur J Pharmacol 463:55–65. https://doi.org/10.1016/S0014-2999(03)01274-3
Brown JS, Kotler BP, Rosemary JS, Wirtz WO III (1988) The effect of owl predation on the foraging behavior of heteromyid rodents. Oecol 76:408–415. https://doi.org/10.1007/BF00377036
Edut S, Eilam D (2004) Protean behavior under barn-owl attack: voles alternate between freezing and fleeing and spiny mice flee in alternating patterns. Behav Brain Res 155:207–216. https://doi.org/10.1016/j.bbr.2004.04.018
Ferkin MH (2018) Odor communication and mate choice in rodents. Biology 7:13. https://doi.org/10.3390/biology7010013
Ferkin MH, Seamon JO (1987) Odor preferences and social behavior in meadow voles, Microtus pennsylvanicus: seasonal differences. Can J Zool 65:2931–2937. https://doi.org/10.1139/z87-445
Ferkin MH, Zucker I (1991) Seasonal variations in photoperiod and ovarian hormones induce the seasonal changes in odour preferences in meadow voles, Microtus pennsylvanicus. J Reprod Fert 92:433–441. https://doi.org/10.1530/jrf.0.0920433
Ferkin MH, Lee DN, Leonard ST (2004) The reproductive state of female voles affects their scent marking behavior and the responses of male conspecifics to such marks. J Ethol 110:257–272. https://doi.org/10.1111/j.1439-0310.2004.00961.x
Ferkin MH, delBarco-Trillo J, Petrulis A (2017) Communication by chemical signals: physiological mechanisms, ontogeny and learning, function, evolution and cognition. In: Pfaff DW, Joëls M (eds) Hormones, brain, and behavior, 3rd edn. Elsevier Press, Oxfordshire, pp 285–327
Fuelling O, Halle S (2004) Breeding suppression in free-ranging grey-sided voles under the influence of predator odour. Oecol 138:151–159. https://doi.org/10.1007/s00442-003-1417-y
Haapakoski M, Hardenbol AA, Matson KD (2018) Exposure to chemical cues from predator-exposed conspecifics increased reproduction in a wild-rodent. Sci Rep 8:1–9. https://doi.org/10.1038/s41598-018-35568-0
Hegab IM, Wei W (2014) Neuroendocrine changes upon exposure to predator odors. Physiol Behav 131:149–155. https://doi.org/10.1016/j.physbeh.2014.04.041
Johnson ML, Johnson S (1982) Voles: Microtus species. In: Chapman JA, Feldharmer GA (eds) Wild mammals of North America: biology, management, and economics. Johns Hopkins University Press, Maryland, pp 327–354
Kohl MT, Stahler DR, Metz MC, Forester JD, Kauffman MJ, Varley N, White PJ, Smith DW, MacNulty DR (2018) Diel predator activity drives a dynamic landscape of fear. Ecol Monogr 88:638–652. https://doi.org/10.1002/ecm.1313
Koskela E, Ylönen H (1995) Suppressed breeding in the field vole (Microtus agrestis): an adaptation to cyclically fluctuating predation risk. Behav Ecol 6:311–315. https://doi.org/10.1093/beheco/6.3.311
Lima SL, Bednekoff PA (1999) Temporal variation in danger drives antipredator behavior: the predation risk allocation hypothesis. Am Nat 153:649–659. https://doi.org/10.1086/303202
Madison DM (1980) Space use and social structure in meadow voles, Microtus pennsylvanicus. Behav Ecol Sociobiol 7:65–71. https://doi.org/10.1007/BF00302520
McShea WJ, Madison DM (1989) Measurements of reproductive traits in a field population of meadow voles. J Mammal 70:132–141. https://doi.org/10.2307/1381676
Merkens M, Harestead A, Sullivan T (1991) Cover and efficacy of predator-based repellents for Townsend’s vole (Microtus townsendii). J Chem Ecol 17:401–412. https://doi.org/10.1007/BF00994341
Norrdahl K, Korpimäki E (1998) Does mobility or sex of voles affect risk of predation by mammalian predators? Ecol 79:226–232. https://doi.org/10.1890/0012-9658(1998)079[0226:DMOSOV]2.0.CO;2
Orrock JL, Danielson BJ, Brinkerhoff RJ (2004) Rodent foraging is affected by indirect, but not by direct, cues of predation risk. Behav Ecol 15:433–437. https://doi.org/10.1093/beheco/arh031
Perrot-Sinal TS, Heale VR, Ossenkopp KP, Kavaliers M (1996) Sexually dimorphic aspects of spontaneous activity in meadow voles (Microtus pennsylvanicus): effects of exposure to fox odor. Behav Neurosci 110:1126–1132. https://doi.org/10.1037/0735-7044.110.5.1126
Perrot-Sinal TS, Ossenkopp KP, Kavaliers M (1999) Effects of repeated exposure to fox odor on locomotor activity levels and spatial movement patterns in breeding male and female meadow voles (Microtus pennsylvanicus). J Chem Ecol 25:1567–1584. https://doi.org/10.1023/A:1020836832455
Perrot-Sinal T, Ossenkopp KP, Kavaliers M (2000) Influence of a natural stressor (predator odor) on locomotor activity in the meadow vole (Microtus pennsylvanicus): modulation by sex, reproductive condition and gonadal hormones. Psychoneuroendocrinology 25:259–276. https://doi.org/10.1016/S0306-4530(99)00054-2
Pierson LM (2018) The effects of diet and food deprivation on the sexual and exploratory behaviors of meadow voles, Microtus Pennsylvanicus. Dissertation, University of Memphis
Pusenius J, Ostfeld RS (2000) Effects of stoat’s presence and auditory cues indicating its presence on tree seedling predation by meadow voles. Oikos 91:123–130. https://doi.org/10.1034/j.1600-0706.2000.910111.x
Pusenius J, Ostfeld RS (2002) Mammalian predator scent, vegetation cover and tree seedling predation by meadow voles. Ecography 25:481–487. https://doi.org/10.1034/j.1600-0706.2000.910111.x
Rhoades E, Blumstein DT (2007) Predicted fitness consequences of threat sensitive hiding behavior. Behav Ecol 18:937–943. https://doi.org/10.1093/beheco/arm064
Sievert T, Haapakoski M, Palme R, Voipio H, Ylönen H (2019) Secondhand horror: effects of direct and indirect predator cues on behavior and reproduction of the bank vole. Ecosphere 10:e02765. https://doi.org/10.1002/ecs2.2765
Spritzer MD, Meikle DB, Solomon NG (2004) The relationship between dominance rank and spatial ability among male meadow voles (Microtus pennsylvanicus). J Comp Psych 118:332–339. https://doi.org/10.1037/0735-7036.118.3.332
Spritzer MD, Meikle DB, Solomon NG (2005) Female choice based on male spatial ability and aggressiveness among meadow voles. Anim Behav 69:1121–1130. https://doi.org/10.1016/j.anbehav.2004.06.033
Takao K, Miyakawa T (2006) Light/dark transition test for mice. J vis Exp. https://doi.org/10.3791/104
Vlautin CT, Ferkin MH (2012) The influence of predator and conspecific odor on sex differences in path choice in meadow voles. Behaviour 149:133–152. https://doi.org/10.1163/156853912X630272
Vlautin CT, Hobbs NJ, Ferkin MH (2010) Male and female meadow voles, Microtus pennsylvanicus, differ in their responses to heterospecific/conspecific over-marks. Ethol 116:797–805. https://doi.org/10.1111/j.1439-0310.2010.01803.x
Wickham H (2016) ggplot2: elegant graphics for data analysis. Springer, New York
Wolff JO (1993) Why are female small mammals territorial? Oikos 68:364–370. https://doi.org/10.2307/3544853
Acknowledgements
The authors would like to thank Kelsey Clark for her advice in the writing of this manuscript, and the University of Memphis Department of Biological Sciences and the Jack H. Morris distinguished Professorship award presented to Dr. Michael H. Ferkin for the funds necessary to complete this manuscript.
Funding
The completion of this experiment was made possible with funds provided by The University of Memphis Department of Biological Sciences and the Jack H. Morris distinguished Professorship award presented to Dr. Michael H. Ferkin.
Author information
Authors and Affiliations
Contributions
The experimental concept was proposed by DMM and Dr. MHF. All authors contributed to the experimental methods. Data collection was completed by DMM and SSG. Data analysis was completed by KNR. First drafts of this manuscript were completed by DMM and SSG. All authors revised subsequent versions of the manuscript, and approved the final draft of this manuscript that was subsequently submitted for review.
Corresponding author
Ethics declarations
Conflict of interest
None of the authors have a conflict of interest to declare.
Ethics approval
All applicable international, national, and institutional guidelines for care and use of animals were followed. The authors specifically followed guidelines of the American Society of Mammalogists for research involving live mammals and Animal Protocol 0731, approved by the Institutional Animal Care and Use Committee at the Univeristy of Memphis, to design and complete this experiment. Human participants were not utilized to complete this experiment.
Consent to participate
Not applicable.
Consent for publication
Not applicable.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Midlick, D.M., Garris, S.S., Rohrer, K.N. et al. Sexual differences in responses of meadow voles to environmental cues in the presence of mink odor. Anim Cogn 25, 1003–1011 (2022). https://doi.org/10.1007/s10071-022-01606-8
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10071-022-01606-8