Abstract
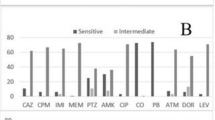
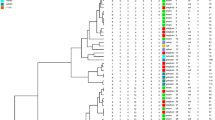
Pseudomonas aeruginosa is an important nosocomial pathogen with a capacity of resistance to multiple antibiotics and production of various extracellular and cell-associated virulence factors that clearly contribute to its pathogenicity. The objective of this study was to investigate the antibiotic susceptibility, virulence factors, and clonal relationship among clinical isolates of P. aeruginosa. Different clinical specimens from hospitalized patients were investigated for P. aeruginosa. Susceptibility of the isolates was evaluated by disc diffusion and broth microdilution methods, as described by the Clinical and Laboratory Standards Institute (CLSI) guideline. A total of 97 P. aeruginosa isolates were recovered from clinical specimens. The percentage of isolates resistant to antimicrobials was imipenem 25.77%, meropenem 15.46%, gentamicin 16.49%, tobramycin 15.46%, amikacin 16.49%, ciprofloxacin 20.61%, levofloxacin 24.74, ceftazidime 20.61%, piperacillin 15.46%, piperacillin/tazobactam 12.37%, colistin 9.27%, and polymyxin B 11.34%. Of isolates, 87.62% possessed β-hemolytic activity, 78.35% lecithinase, 59.8% elastase, 37.11% DNase, and 28.86% twitching motility. The frequency of virulence genes in isolates was lasB 82.47%, plcH 82.47%, exoA 58.76%, exoS 56.7%, and pilA 10.3%. ERIC-PCR typing clustered P. aeruginosa isolates to 19 common types (CT1-CT19) containing isolates from different hospitals and 43 single types (ST1-ST43). Colistin and polymyxin B were the most effective agents against the majority of P. aeruginosa isolates, emphasizing the effort to maintain their antibacterial activity as last-line therapy. The frequency of some virulence factors and genes was noticeably high, which is alarming. In addition, more effective strategies and surveillance are necessary to confine and prevent the inter-hospital and/or intra-hospital dissemination of P. aeruginosa between therapeutic centers.



Similar content being viewed by others
References
Aghaei SS, Javadi A, Sharifi Y, Morovvati A (2016) Detection of exotoxin A, Y, T, U, S genes of Pseudomonas aeruginosa isolates resistant to third-generation cephalosporins in clinical samples of hospitalized patients in hospitals of Qom city. Iran Qom Univ Med Sci J 10:48–55
Ahmad K, Ali A, Rahat S (2018) Prevalence of virulence genes among clinical isolates of Pseudomonas aeruginosa collected from Peshawar, Pakistan. J Pakistan Med Assoc 68:1787–1791
Al-Shimmary SM (2020) Molecular identification and prevalence of some virulence genes among Pseudomonas aeruginosa isolated from Iraqi patients. Int J Pharm Res. https://doi.org/10.31838/ijpr/2020.SP1.237
Al-Wrafy F, Brzozowska E, Górska S, Gamian A (2017) Pathogenic factors of Pseudomonas aeruginosa-the role of biofilm in pathogenicity and as a target for phage therapy. Postepy Hig Med Dosw 71:78–91. https://doi.org/10.5604/01.3001.0010.3792
Barbieri J, Sun J (2004) Pseudomonas aeruginosa ExoS and ExoT. Rev Physiol Biochem Pharmacol 152:79–92. https://doi.org/10.1007/s10254-004-0031-7
Bodey GP, Bolivar R, Fainstein V, Jadeja L (1983) Infections caused by Pseudomonas aeruginosa. Rev Infect Dis 5:279–313. https://doi.org/10.1093/clinids/5.2.279
Burrows LL (2012) Pseudomonas aeruginosa twitching motility: type IV pili in action. Annu Rev Microbiol 66:493–520. https://doi.org/10.1146/annurev-micro-092611-150055
Carter MQ, Chen J, Lory S (2010) The Pseudomonas aeruginosa pathogenicity island PAPI-1 is transferred via a novel type IV pilus. J Bacteriol 192:3249–3258. https://doi.org/10.1128/JB.00041-10
Charan AR, Reddy V, Reddy P, Reddy S, Sivaramakrishnan S (2011) Assessment of genetic diversity in Pseudomonas fluorescens using PCR-based methods. Bioremediat Biodivers Bioavailab 5:10–16
Clinical and laboratory standards institute (2015). Methods for dilution antimicrobial susceptibility tests for bacteria that grow aerobically. 10th ed. M7-A10. Wayne, PA, CLSI supplement M7-A10M100
Clinical and laboratory standards institute (2018). Performance standards for antimicrobial susceptibility testing. 28th ed. Wayne, PA, CLSI supplement M100
Corehtash ZG, Ahmad Khorshidi FF, Akbari H, Aznaveh AM (2015) Biofilm formation and virulence factors among Pseudomonas aeruginosa isolated from burn patients. Jundishapur J Microbiol 8:e22345. https://doi.org/10.5812/jjm.22345
de Bentzmann S, Aurouze M, Ball G, Filloux A (2006) FppA, a novel Pseudomonas aeruginosa prepilin peptidase involved in assembly of type IVb pili. J Bacteriol 188:4851–4860. https://doi.org/10.1128/JB.00345-06
Dogonchi AA, Ghaemi EA, Ardebili A, Yazdansetad S, Pournajaf A (2018) Metallo-β-lactamase-mediated resistance among clinical carbapenem-resistant Pseudomonas aeruginosa isolates in northern Iran: a potential threat to clinical therapeutics. T Ci Ji Yi Xue Za Zhi 30:90–96. https://doi.org/10.4103/tcmj.tcmj_101_17
Doustdar F, Karimi F, Abedinyfar Z, Amoli FA, Goudarzi H (2019) Genetic features of Pseudomonas aeruginosa isolates associated with eye infections referred to Farabi Hospital. Int Ophthalmol 39:1581–1587. https://doi.org/10.1007/s10792-018-0980-5
Eid D, EN W, Barwa R, El-Sokkary MA (2012) Phenotypic and genotypic characterization of some virulence factors in Pseudomonas aeruginosa strains isolated from different clinical sources in Mansoura University Hospitals. New Egypt J Microbiol 32:151–167
Elmouaden C, Laglaoui A, Ennanei L, Bakkali M, Abid M (2019) Virulence genes and antibiotic resistance of Pseudomonas aeruginosa isolated from patients in the Northwestern of Morocco. J Infect Dev Ctries 13:892–898. https://doi.org/10.3855/jidc.10675
European Centre for Disease Prevention and Control (2017) Antimicrobial resistance surveillance in Europe 2015. Annual Report of the European Antimicrobial Resistance Surveilance Network (EARS-Net). Stockholm: ECDC
Foley SL, Lynne AM, Nayak R (2009) Molecular typing methodologies for microbial source tracking and epidemiological investigations of Gram-negative bacterial foodborne pathogens. Infect Genet Evol 9:430–440. https://doi.org/10.1016/j.meegid.2009.03.004
Fukuda K, Ishida W, Fukushima A, Nishida T (2017) Corneal fibroblasts as sentinel cells and local immune modulators in infectious keratitis. Int J Mol Sci 18:1831. https://doi.org/10.3390/ijms18091831
Gajdács M, Baráth Z, Kárpáti K et al (2021) No correlation between biofilm formation, virulence factors, and antibiotic resistance in Pseudomonas aeruginosa: results from a laboratory based in vitro study. Antibiotics (Basel) 10:1134. https://doi.org/10.3390/antibiotics10091134
Georgescu M, Gheorghe I, Curutiu C, Lazar V, Bleotu C, Chifiriuc M-C (2016) Virulence and resistance features of Pseudomonas aeruginosa strains isolated from chronic leg ulcers. BMC Infect Dis 16(Suppl 1):92. https://doi.org/10.1186/s12879-016-1396-3
Gomila M, del Carmen GM, Fernández-Baca V et al (2013) Genetic diversity of clinical Pseudomonas aeruginosa isolates in a public hospital in Spain. BMC Microbiol 18(13):138. https://doi.org/10.1186/1471-2180-13-138
González-Olvera EM, Pérez-Morales R, Zamora AG, Castro-Escarpulli G, Palma-Martínez I, Alba-Romero JJ (2019) Antibiotic resistance, virulence factors and genotyping of Pseudomonas aeruginosa in public hospitals of northeastern Mexico. J Infect Dev Ctries 13:374–383. https://doi.org/10.3855/jidc.10953
Hassuna NA, Mandour SA, Mohamed ES (2020) Virulence constitution of multi-drug-resistant Pseudomonas aeruginosa in upper Egypt. Infect Drug Resist 13:587–595. https://doi.org/10.2147/IDR.S233694
Hughes DT, Sperandio V (2008) Inter-kingdom signalling: communication between bacteria and their hosts. Nat Rev Microbiol 6:111–120. https://doi.org/10.1038/nrmicro1836
Janda JM, Bottone EJ (1981) Pseudomonas aeruginosa enzyme profiling: predictor of potential invasiveness and use as an epidemiological tool. J Clin Microbiol 14:55–60. https://doi.org/10.1128/jcm.14.1.55-60.1981
Javanmardi F, Emami A, Pirbonyeh N, Keshavarzi A, Rajaee M (2019) A systematic review and meta-analysis on exo-toxins prevalence in hospital acquired Pseudomonas aeruginosa isolates. Infect Genet Evol 75:104037. https://doi.org/10.1016/j.meegid.2019.104037
Jurado-Martín I, Sainz-Mejías M, McClean S (2021) Pseudomonas aeruginosa: an audacious pathogen with an adaptable arsenal of virulence factors. Int J Mol Sci 22:3128. https://doi.org/10.3390/ijms22063128
Karatuna O, Yagci A (2010) Analysis of quorum sensing-dependent virulence factor production and its relationship with antimicrobial susceptibility in Pseudomonas aeruginosa respiratory isolates. Clin Microbiol Infect 16:1770–1775. https://doi.org/10.1111/j.14690691.2010.03177.x
Karimi E, Ghalibafan F, Esfandani A et al (2021) Antibiotic resistance pattern in Pseudomonasaeruginosa isolated from clinical samples other than burn samples in Iran. Avicenna J Med Biotechnol 13:35–41. https://doi.org/10.18502/ajmb.v13i1.4575
Khosravi AD, Shafie F, Montazeri EA, Rostami S (2016) The frequency of genes encoding exotoxin A and exoenzyme S in Pseudomonas aeruginosa strains isolated from burn patients. Burns 42:1116–1120. https://doi.org/10.1016/j.burns.2016.02.012
Kiewitz C, Tümmler B (2000) Sequence diversity of Pseudomonas aeruginosa: impact on population structure and genome evolution. J Bacteriol 182:3125–3135. https://doi.org/10.1128/JB.182.11.3125-3135.2000
Ktari S, Mnif B, Znazen A et al (2011) Diversity of β-lactamases in Pseudomonas aeruginosa isolates producing metallo-β-lactamase in two Tunisian hospitals. Microb Drug Resist 17:25–30. https://doi.org/10.1089/mdr.2010.0104
Lanotte P, Watt S, Mereghetti L et al (2004) Genetic features of Pseudomonas aeruginosa isolates from cystic fibrosis patients compared with those of isolates from other origins. J Med Microbiol 53:73–81. https://doi.org/10.1099/jmm.0.05324-0
Leone I, Chirillo M, Raso T, Zucca M, Savoia D (2008) Phenotypic and genotypic characterization of Pseudomonas aeruginosa from cystic fibrosis patients. Eur J Clin Microbiol Infect Dis 27:1093–1099. https://doi.org/10.1007/s10096-008-0551-1
Macin S, Akyon Y (2017) Phenotypic and genotypic virulence factors in Pseudomonasaeruginosa strains according to pigment presence. Acta Medica Mediterr 33:1033–1038. https://doi.org/10.19193/0393-6384_2017_6_163
Magiorakos A-P, Srinivasan A, Carey R et al (2012) Multidrug-resistant, extensively drug-resistant and pandrug-resistant bacteria: an international expert proposal for interim standard definitions for acquired resistance. Clin Microbiol Infect 18:268–281. https://doi.org/10.1111/j.1469-0691.2011.03570.x
Michalska M, Wolf P (2015) Pseudomonas exotoxin A: optimized by evolution for effective killing. Front Microbiol 15:963. https://doi.org/10.3389/fmicb.2015.00963
Mitov I, Strateva T, Markova B (2010) Prevalence of virulence genes among bulgarian nosocomial and cystic fibrosis isolates of Pseudomonas aeruginosa. Braz J Microbiol 41:588–595. https://doi.org/10.1590/S1517-83822010000300008
Mittal R, Khandwaha RK, Gupta V, Mittal P, Harjai K (2006) Phenotypic characters of urinary isolates of Pseudomonas aeruginosa & their association with mouse renal colonization. Indian J Med Res 123(1):67–72
Namaei MH, Yousefi M, Askari P et al (2021) High prevalence of multidrug-resistant non-fermentative Gram-negative bacilli harboring bla and bla metallo-beta-lactamase genes in Birjand, south-east Iran. Iran J Microbiol 13:470–479. https://doi.org/10.18502/ijm.v13i4.6971
Peña C, Cabot G, Gómez-Zorrilla S et al (2015) Influence of virulence genotype and resistance profile in the mortality of Pseudomonas aeruginosa bloodstream infections. Clin Infect Dis 15(60):539–548. https://doi.org/10.1093/cid/ciu866
Peymani A, Farivar TN, Ghanbarlou MM, Najafipour R (2015) Dissemination of Pseudomonas aeruginosa producing blaIMP-1 and blaVIM-1 in Qazvin and Alborz educational hospitals. Iran Iran J Microbiol 7:302–309
Pirnay JP, De Vos D, Cochez C et al (2002) Pseudomonas aeruginosa displays an epidemic population structure. Environ Microbiol 4:898–911. https://doi.org/10.1046/j.1462-2920.2002.00321.x
Pobiega M, Chmielarczyk A, Kozioł J et al (2018) Virulence factors genes and drug resistance in Pseudomonas aeruginosa strains derived from different forms of community and healthcare associated infections. Postepy Hig Med Dosw 72:751–759
Pournajaf A, Razavi S, Irajian G et al (2018) Integron types, antimicrobial resistance genes, virulence gene profile, alginate production and biofilm formation in Iranian cystic fibrosis Pseudomonas aeruginosa isolates. Infez Med 26:226–236
Priya JL, Prajna L, Mohankumar V (2015) Genotypic and phenotypic characterization of Pseudomonas aeruginosa isolates from post-cataract endophthalmitis patients. Microb Pathog 78:67–73. https://doi.org/10.1016/j.micpath.2014.11.014
Raafat MM, Ali-Tammam M, Ali AE (2016) Phenotypic and genotypic characterization of Pseudomonas aeruginosa isolates from Egyptian hospitals. African J Microbiol Res 10:1645–1653. https://doi.org/10.5897/AJMR2016.8254
Rad ZR, Rad ZR, Goudarzi H et al (2021) Detection of New Delhi Metallo-β-lactamase-1 among Pseudomonas aeruginosa isolated from adult and pediatric patients in Iranian hospitals. Gene Reports 23:101152. https://doi.org/10.1016/j.genrep.2021.101152
Römling U, Wingender J, Müller H, Tümmler B (1994) A major Pseudomonas aeruginosa clone common to patients and aquatic habitats. Appl Environ Microbiol 60:1734–1738. https://doi.org/10.1128/aem.60.6.1734-1738.1994
Ruimy R, Genauzeau E, Barnabe C, Beaulieu A, Tibayrenc M, Andremont A (2001) Genetic diversity of Pseudomonas aeruginosa strains isolated from ventilated patients with nosocomial pneumonia, cancer patients with bacteremia, and environmental water. Infect Immun 69:584–588. https://doi.org/10.1128/IAI.69.1.584-588.2001
Sadeghi H, Najafabadi A, Abedi D, Dehkordi A (2008) Identification of an isolate of Pseudomonas aeroginosa desposited in PTCC as a PHA producer strains: comparison of three different bacterial genomic DNA extraction methods. J Biol Sci 8:826–830. https://doi.org/10.3923/jbs.2008.826.830
Sader HS, Castanheira M, Duncan LR, Flamm RK (2018) Antimicrobial susceptibility of Enterobacteriaceae and Pseudomonas aeruginosa isolates from United States medical centers stratified by infection type: results from the International Network for Optimal Resistance Monitoring (INFORM) surveillance program, 2015–2016. Diagn Microbiol Infect Dis 92:69–74. https://doi.org/10.1016/j.diagmicrobio.2018.04.012
Sharifi H, Pouladfar G, Shakibaie MR, Pourabbas B, Mardaneh J, Mansouri S (2019) Prevalence of β-lactamase genes, class 1 integrons, major virulence factors and clonal relationships of multidrug-resistant Pseudomonasaeruginosa isolated from hospitalized patients in southeast of Iran. Iran J Basic Med Sci 22:806–812. https://doi.org/10.22038/ijbms.2019.35063.8340
Sheikh AF, Ghanbari F, Afzali M, Shahin M (2020) Isolation of oxidase-negative Pseudomonas aeruginosa from various specimens. Iranian J Public Health 49:1186–1188. https://doi.org/10.18502/ijph.v49i6.3376
Solomon SL, Oliver KB (2014) Antibiotic resistance threats in the United States: stepping back from the brink. Am Fam Physician 89:938–941
Sonbol FI, Khalil MAEF, Mohamed AB, Sameh SA (2015) Correlation between antibiotic resistance and virulence of Pseudomonas aeruginosa clinical isolates. Turk J Med Sci 45:568–577. https://doi.org/10.3906/sag-1406-58
Sorkh MAG, Shokoohizadeh L, Rashidi N, Tajbakhsh E (2017) Molecular analysis of Pseudomonas aeruginosa strains isolated from burn patients by repetitive extragenic palindromic-PCR (rep-PCR). Iran Red Crescent Med J 19:e43508
Strateva T, Mitov I (2011) Contribution of an arsenal of virulence factors to pathogenesis of Pseudomonas aeruginosa infections. Ann Microbiol 61:717–732. https://doi.org/10.1007/s13213-011-0273-y
Tielen P, Narten M, Rosin N et al (2011) (2011) Genotypic and phenotypic characterization of Pseudomonas aeruginosa isolates from urinary tract infections. Int J Med Microbiol 301:282–292. https://doi.org/10.1016/j.ijmm.2010.10.005
Timani RJ (2011) Molecular typing and biofilm characterization of Pseudomonas aeruginosa isolated from clinical samples in Lebanon.(c2011). Dissertation, Lebanese American University. https://doi.org/10.26756/th.2011.44
Tingpej P, Smith L, Rose B et al (2007) Phenotypic characterization of clonal and nonclonal Pseudomonas aeruginosa strains isolated from lungs of adults with cystic fibrosis. J Clin Microbiol 45:1697–1704. https://doi.org/10.1128/JCM.02364-06
Ullah W, Qasim M, Rahman H, Jie Y, Muhammad N (2017) Beta-lactamase-producing Pseudomonas aeruginosa: phenotypic characteristics and molecular identification of virulence genes. J Chin Med Assoc 80:173–177. https://doi.org/10.1016/j.jcma.2016.08.011
Van Belkum A, Tassios P, Dijkshoorn L et al (2007) Guidelines for the validation and application of typing methods for use in bacterial epidemiology. Clin Microbiol Infect 3:1–46. https://doi.org/10.1111/j.14690691.2007.01786.x
Walkty A, Lagace-Wiens P, Adam H et al (2017) Antimicrobial susceptibility of 2906 Pseudomonas aeruginosa clinical isolates obtained from patients in Canadian hospitals over a period of 8 years: results of the Canadian Ward surveillance study (CANWARD), 2008–. Diagn Microbiol Infect Dis 87:60–63. https://doi.org/10.1016/j.diagmicrobio.2016.10.003
Wolska K, Szweda P, Lada K et al (2014) Motility activity, slime production, biofilm formation and genetic typing by ERIC-PCR for Pseudomonas aeruginosa strains isolated from bovine and other sources (human and environment). Pol J Vet Sci 17:321–329. https://doi.org/10.2478/pjvs-2014-0044
Yousefi-Avarvand A, Khashei R, Sedigh Ebrahim-Saraie H, Emami A, Zomorodian K, Motamedifar M (2015) The frequency of exotoxin A and exoenzymes S and U genes among clinical isolates of Pseudomonas aeruginosa in Shiraz. Iran Int J Mol Cell Med 4:167–173
Zahedani SS, Tahmasebi H, Jahantigh M (2021) Coexistence of virulence factors and efflux pump genes in clinical isolates of Pseudomonas aeruginosa: analysis of biofilm-forming strains from Iran. Int J Microbiol 2021:5557361. https://doi.org/10.1155/2021/5557361
Zarei O, Shokoohizadeh L, Hossainpour H, Alikhani MY (2018) Molecular analysis of Pseudomonas aeruginosa isolated from clinical, environmental and cockroach sources by ERIC-PCR. BMC Res Notes 11:668. https://doi.org/10.1186/s13104-018-3765-z
Acknowledgements
The authors would like to thank the staff of the selected hospitals as well as all the colleagues at the Department of Microbiology, Golestan University of Medical Sciences, Gorgan, Iran, for their laboratory cooperation.
Funding
This work was financially supported by grant 110212 from the Golestan University of Medical Sciences, Gorgan, Iran.
Author information
Authors and Affiliations
Contributions
AA, AJ, and FNS conceptualized and designed study. FNS, AA, HB, and AH performed the experiments. FNS, AA, AJ, and MASBG interpreted results and analyzed data. FNS and AI wrote the original draft. AA reviewed the manuscript and critically revised it.
Corresponding author
Ethics declarations
Ethics approval
This study was approved by the Ethics Committee of Golestan University of Medical Sciences with ethical code number IR.GUMS. REC.1398.079. All patients were verbally informed about this study, and consensus was obtained from their close relatives. In order to maintain patient’s confidentiality, participants were anonymous, and no personal information was collected or included in the study.
Competing interests
The authors declare no competing interests.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Shahri, F.N., Izanloo, A., Goharrizi, M.A.S.B. et al. Antimicrobial resistance, virulence factors, and genotypes of Pseudomonas aeruginosa clinical isolates from Gorgan, northern Iran. Int Microbiol 25, 709–721 (2022). https://doi.org/10.1007/s10123-022-00256-7
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10123-022-00256-7