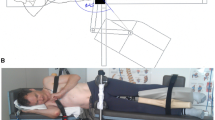
Abstract
The role of intra-abdominal pressure (IAP) in spinal load reduction has remained controversial, partly because previous musculoskeletal models did not introduce the pressure generating mechanism. In this study, an integrated computational methodology is proposed to combine the IAP change with core muscle activations. An ideal gas relationship was introduced to calculate pressure distribution within the abdominal cavity. Additionally, based on flexible multibody dynamics, a muscle membrane element was developed by incorporating the muscular fiber deformation, inter-fiber stiffness, and volume constancy. This element was then utilized in discretizing the diaphragm and transversus abdominis, forming an IAP–muscle coupling system of the abdominal cavity. Based on this methodology, a forward dynamic simulation of spinal flexion was presented to examine the unloading effect of abdominal breathing. The results confirm that core muscle contraction during the abdominal breathing cycle can substantially reduce the forces of spinal compression together with trunk extensor muscles, and this effect is more pronounced when the IAP increase is produced by contraction of the transversus abdominis. This unloading effect still holds even with the co-activation of other abdominal muscles, providing a potential choice when designing trunk movements during weight-lifting tasks.














Similar content being viewed by others
References
Angelillo M, Boriek AM, Rodarte JR, Wilson TA (1997) Theory of diaphragm structure and shape. J Appl Physiol 83(5):1486–1491
Arjmand N, Shirazi-Adl A (2005) Biomechanics of changes in lumbar posture in static lifting. Spine 30(23):2637–2648
Arjmand N, Shirazi-Adl A (2006a) Model and in vivo studies on human trunk load partitioning and stability in isometric forward flexions. J Biomech 39(3):510–521
Arjmand N, Shirazi-Adl A (2006b) Role of intra-abdominal pressure in the unloading and stabilization of the human spine during static lifting tasks. Eur Spine J 15(8):1265–1275
Arshad R, Zander T, Dreischarf M, Schmidt H (2016) Influence of lumbar spine rhythms and intra-abdominal pressure on spinal loads and trunk muscle forces during upper body inclination. Med Eng Phys 38(4):333–338
Ashton-Miller JA, Schultz AB (1997) Biomechanics of the human spine. Basic orthopaedic biomechanics. Lippincott Williams & Wilkins, New York, pp 353–393
Bayer A, Schmitt S, Günther M, Haeufle DFB (2017) The influence of biophysical muscle properties on simulating fast human arm movements. Comput Math Method Med 20(8):803–821
Bernstein IT (1997) The pelvic floor muscles. Muscle thickness in healthy and urinary incontinent women measured by perineal ultrasonography with reference to the effect of pelvic floor training. Estrogen receptors studies. Neurourol Urodyn 16(4):237–275
Blankevoort L, Kuiper JH, Huiskes R, Grootenboer HJ (1991) Articular contact in a three-dimensional model of the knee. J Biomech 24(11):1019–1031
Brown SH, McGill SM (2010) A comparison of ultrasound and electromyography measures of force and activation to examine the mechanics of abdominal wall contraction. Clin Biomech 25(2):115–123
Bruno AG, Bouxsein ML, Anderson DE (2015) Development and validation of a musculoskeletal model of the fully articulated thoracolumbar spine and rib cage. Biomech Eng Trans ASME 137(8):081003
Burkhart K, Grindle D, Bouxsein ML, Anderson DE (2020) Between-session reliability of subject-specific musculoskeletal models of the spine derived from optoelectronic motion capture data. J Biomech 112(110):044
de Cesare N, Trevisan C, Maghin E, Piccoli M, Pavan PG (2018) A finite element analysis of diaphragmatic hernia repair on an animal model. J Mech Behav Biomed Mater 86:33–42
Choi YT, Wereley NM (2005) Biodynamic response mitigation to shock loads using magnetorheological helicopter crew seat suspensions. J Aircr 42(5):1288–1295
Cholewicki J, Reeves N (2004) All abdominal muscles must be considered when evaluating the intra-abdominal pressure contribution to trunk extensor moment and spinal loading. J Biomech 37(6):953–954
Cholewicki J, Juluru K, McGill SM (1999) Intra-abdominal pressure mechanism for stabilizing the lumbar spine. J Biomech 32(1):13–17
Cholewicki J, Ivancic PC, Radebold A (2002) Can increased intra-abdominal pressure in humans be decoupled from trunk muscle co-contraction during steady state isometric exertions? Eur J Appl Physiol 87(2):127–133
Christophy M, Faruk Senan NA, Lotz JC, O’Reilly OM (2012) A musculoskeletal model for the lumbar spine. Biomech Model Mechanobiol 11(1):19–34
Daggfeldt K, Thorstensson A (1997) The role of intra-abdominal pressure in spinal unloading. J Biomech 30(11–12):1149–1155
Daggfeldt K, Thorstensson A (2003) The mechanics of back-extensor torque production about the lumbar spine. J Biomech 36(6):815–825
Delp SLL, Loan JPP, Hoy MGG, Zajac FE, Topp EL, Rosen JM (1990) An interactive graphics-based model of the lower extremity to study orthopedic surgical procedures. IEEE Trans Biomed Eng 37(8):757–767
Desailly E, Sardain P, Khouri N, Yepremian D, Lacouture P (2010) The convex wrapping algorithm: a method for identifying muscle paths using the underlying bone mesh. J Biomech 43(13):2601–2607
Diao H, Xin H, Jin Z (2018) Prediction of in vivo lower cervical spinal loading using musculoskeletal multi-body dynamics model during the head flexion/extension, lateral bending and axial rotation. Proc Inst Mech Eng Part H–J Eng Med 232(11):1071–1082
Eberlein R, Holzapfel GA, Fröhlich M (2004) Multi-segment FEA of the human lumbar spine including the heterogeneity of the annulus fibrosus. Comput Mech 34(2):147–163
Favre P, Gerber C, Snedeker JG (2010) Automated muscle wrapping using finite element contact detection. J Biomech 43(10):1931–1940
Fink RN, Lembo AJ (2001) Intestinal gas. Curr Treat Options Gastroenterol 4(4):333–337
Flores P, MacHado M, Silva MT, Martins JM (2011) On the continuous contact force models for soft materials in multibody dynamics. Multibody Syst Dyn 25(3):357–375
Frank C, Kobesova A, Kolar P (2013) Dynamic neuromuscular stabilization & sports rehabilitation. Int J Sports Phys Ther 8(1):62–73
Gantoi FM, Brown MA, Shabana AA (2010) ANCF finite element/multibody system formulation of the ligament/bone insertion site constraints. J Comput Nonlinear Dyn 5(3):031006
Gardiner JC, Weiss JA (2000) simple shear testing of parallel-fibered planar soft tissues. Biomech Eng Trans ASME 123(2):170–175
Gardner-Morse MG, Stokes IA (2004) Structural behavior of human lumbar spinal motion segments. J Biomech 37(2):205–212
Garner BA, Pandy MG (2000) The obstacle-set method for representing muscle paths in musculoskeletal models. Comput Math Method Med 3(1):1–30
Gfrerer MH, Simeon B (2021) Fiber-based modeling and simulation of skeletal muscles. Multibody Syst Dyn. https://doi.org/10.1007/s11044-021-09781-1
Gilad I, Nissan M (1986) A study of vertebra and disc geometric relations of the human cervical and lumbar spine. Spine 11(2):154–157
Goligher EC, Laghi F, Detsky ME, Farias P, Murray A, Brace D, Brochard LJ, Sebastien-Bolz S, Rubenfeld GD, Kavanagh BP, Ferguson ND (2015) Measuring diaphragm thickness with ultrasound in mechanically ventilated patients: feasibility, reproducibility and validity. Intensive Care Med 41(4):642–649
Grasa J, Sierra M, Lauzeral N, Muñoz M, Miana-Mena F, Calvo B (2016) Active behavior of abdominal wall muscles: Experimental results and numerical model formulation. J Mech Behav Biomed Mater 61:444–454
Günther M, Schmitt S, Wank V (2007) High-frequency oscillations as a consequence of neglected serial damping in Hill-type muscle models. Biol Cybern 97(1):63–79
Guo J, Huang H, Yu Y, Liang Z, Ambrósio J, Zhao Z, Ren G, Ao Y (2020a) Modeling muscle wrapping and mass flow using a mass-variable multibody formulation. Multibody Syst Dyn 49:315–336
Guo J, Sun Y, Hao Y, Cui L, Ren G (2020b) A mass-flowing muscle model with shape restrictive soft tissues: correlation with sonoelastography. Biomech Model Mechanobiol 19(3):911–926
Haasdonk B, Wenzel T, Santin G, Schmitt S (2020) Biomechanical surrogate modelling using stabilized vectorial greedy kernel methods. arXiv preprint arXiv:200412670
Halilaj E, Rajagopal A, Fiterau M, Hicks JL, Hastie TJ, Delp SL (2018) Machine learning in human movement biomechanics: best practices, common pitfalls, and new opportunities. J Biomech 81:1–11
Hammer M, Günther M, Haeufle DF, Schmitt S (2019) Tailoring anatomical muscle paths: a sheath-like solution for muscle routing in musculoskeletal computer models. Math Biosci 311:68–81
Hernández B, Peña E, Pascual G, Rodríguez M, Calvo B, Doblaré M, Bellón J (2011) Mechanical and histological characterization of the abdominal muscle. A previous step to modelling hernia surgery. J Mech Behav Biomed Mater 4(3):392–404
Hodges PW, Butler JE, McKenzie DK, Gandevia SC (1997) Contraction of the human diaphragm during rapid postural adjustments. J Physiol 505(2):539–548
Hodges PW, Cresswell AG, Daggfeldt K, Thorstensson A (2001) In vivo measurement of the effect of intra-abdominal pressure on the human spine. J Biomech 34(3):347–353
Hodges PW, Eriksson AE, Shirley D, Gandevia SC (2005) Intra-abdominal pressure increases stiffness of the lumbar spine. J Biomech 38(9):1873–1880
Hoyte L, Jakab M, Warfield SK, Shott S, Flesh G, Fielding JR (2004) Levator ani thickness variations in symptomatic and asymptomatic women using magnetic resonance-based 3-dimensional color mapping. Am J Obstet Gynecol 191(3):856–861
Hui GC, Amaral J, Stephens D, Atenafu E, John P, Temple M, Chait P, Connolly B (2005) Gas distribution in intraabdominal and pelvic abscesses on CT is associated with drainability. Am J Roentgenol 184(3):915–919
Hume DR, Navacchia A, Rullkoetter PJ, Shelburne KB (2019) A lower extremity model for muscle-driven simulation of activity using explicit finite element modeling. J Biomech 84:153–160
Ignasiak D, Ferguson SJ, Arjmand N (2016) A rigid thorax assumption affects model loading predictions at the upper but not lower lumbar levels. J Biomech 49(13):3074–3078
Ignasiak D, Valenzuela W, Reyes M, Ferguson SJ (2018) The effect of muscle ageing and sarcopenia on spinal segmental loads. Eur Spine J 27(10):2650–2659
Karajan N, Röhrle O, Ehlers W, Schmitt S (2013) Linking continuous and discrete intervertebral disc models through homogenisation. Biomech Model Mechanobiol 12(3):453–466
Khoddam-Khorasani P, Arjmand N, Shirazi-Adl A (2020) Effect of changes in the lumbar posture in lifting on trunk muscle and spinal loads: a combined in vivo, musculoskeletal, and finite element model study. J Biomech 104(109):728
Khurelbaatar T, Kim K, Hyuk Kim Y (2015) A cervico-thoraco-lumbar multibody dynamic model for the estimation of joint loads and muscle forces. Biomech Eng Trans ASME 137(11):111001
Kłodowski A, Rantalainen T, Mikkola A, Heinonen A, Sievänen H (2011) Flexible multibody approach in forward dynamic simulation of locomotive strains in human skeleton with flexible lower body bones. Multibody Syst Dyn 25(4):395–409
Kłodowski A, Valkeapää A, Mikkola A (2012) Pilot study on proximal femur strains during locomotion and fall-down scenario. Multibody Syst Dyn 28(3):239–256
Levitt MD (1971) Volume and composition of human intestinal gas determined by means of an intestinal washout technic. N Engl J Med 284(25):1394–1398
Li J, Lu Y, Miller SC, Jin Z, Hua X (2019) Development of a finite element musculoskeletal model with the ability to predict contractions of three-dimensional muscles. J Biomech 94:230–234
Liu C, Tian Q, Yan D, Hu H (2013) Dynamic analysis of membrane systems undergoing overall motions, large deformations and wrinkles via thin shell elements of ANCF. Comput Methods Appl Mech Eng 258:81–95
Liu T, Khalaf K, Adeeb S, El-Rich M (2019a) Effects of lumbo-pelvic rhythm on trunk muscle forces and disc loads during forward flexion: a combined musculoskeletal and finite element simulation study. J Biomech 82:116–123
Liu T, Khalaf K, Adeeb S, El-Rich M (2019b) Numerical investigation of intra-abdominal pressure effects on spinal loads and load-sharing in forward flexion. Front Bioeng Biotechnol 7:1–12
Loring SH, Yoshino K, Kimball WR, Barnas GM (1994) Gravitational and shear-associated pressure gradients in the abdomen. J Appl Physiol 77(3):1375–1382
Marras WS, Mirka GA (1996) Intra-abdominal pressure during trunk extension motions. Clin Biomech 11(5):267–274
Martins JAC, Pato MPM, Pires EB (2006) A finite element model of skeletal muscles. Virtual Phys Prototyp 1(3):159–170
Meng X, Bruno AG, Cheng B, Wang W, Bouxsein ML, Anderson DE (2015) Incorporating six degree-of-freedom intervertebral joint stiffness in a lumbar spine musculoskeletal model-method and performance in flexed postures. Biomech Eng Trans ASME 137(10):101008
Mens J, Hoek Van Dijke G, Pool-Goudzwaard A, Van Der Hulst V, Stam H (2006) Possible harmful effects of high intra-abdominal pressure on the pelvic girdle. J Biomech 39(4):627–635
Michael G, Röhrle O, Daniel FBH, Schmitt S (2012) Spreading out muscle mass within a Hill-type model: a computer simulation study. Comput Math Method Med 848:630
Mikkola AM, Shabana AA (2003) A non-incremental finite element procedure for the analysis of large deformation of plates and shells in mechanical system applications. Multibody Syst Dyn 9(3):283–309
Millard M, Delp S (2012) A computationally efficient muscle model. In: ASME 2012 summer bioengineering conference, American Society of Mechanical Engineers, Fajardo, Puerto Rico, 44809, pp 1055–1056
Millard M, Uchida T, Seth A, Delp SL (2013) Flexing computational muscle: modeling and simulation of musculotendon dynamics. J Biomech Eng Trans ASME 135(2):21005
Mohamed ANA, Brown MA, Shabana AA (2010) Study of the ligament tension and cross-section deformation using nonlinear finite element/multibody system algorithms. Multibody Syst Dyn 23(3):227–248
Mokhtarzadeh H, Farahmand F, Shirazi-Adl A, Arjmand N, Malekipour F, Parnianpour M (2012) The effects of intra-abdominal pressure on the stability and unloading of the spine. J Mech Med Biol 12(1):1250014
Mörl F, Günther M, Riede JM, Hammer M, Schmitt S (2020) Loads distributed in vivo among vertebrae, muscles, spinal ligaments, and intervertebral discs in a passively flexed lumbar spine. Biomech Model Mechanobiol 19:2015–2047
Nachemson ALFL, Andersson GBJ, Schultz AB (1986) Valsalva maneuver biomechanics: effects on lumbar trunk loads of elevated intraabdominal pressures. Spine 11(5):476–479
Nowakowska-Lipiec K, Michnik R, Linek P, Myśliwiec A, Jochymczyk-Woźniak K, Gzik M (2020) A numerical study to determine the effect of strengthening and weakening of the transversus abdominis muscle on lumbar spine loads. Comput Methods Biomech Biomed Eng 23(16):1287–1296
Pai DK (2010) Muscle mass in musculoskeletal models. J Biomech 43(11):2093–2098
Pandy MG, Zajac FE, Sim E, Levine WS (1990) An optimal control model for maximum-height human jumping. J Biomech 23(12):1185–1198
Panjabi MM, Takata K, Goel V, Federico D, Oxland T, Duranceau J, Krag M (1991) Thoracic human vertebrae quantitative three-dimensional anatomy. Spine 16(8):1487–1497
Panjabi MM, Oxland T, Takata K, Goel V, Duranceau J, Krag M (1993) Articular facets of the human spine quantitative three-dimensional anatomy. Spine 18(10):1298–1310
Pato MPM, Areias P (2010) Active and passive behaviors of soft tissues: pelvic floor muscles. Int J Numer Methods Biomed 26(6):667–680
Pato MPM, Santos NJG, Areias P, Pires EB, de Carvalho M, Pinto S, Lopes DS (2011) Finite element studies of the mechanical behaviour of the diaphragm in normal and pathological cases. Comput Methods Biomech Biomed Eng 14(6):505–513
Pel JJ, Spoor CW, Goossens RH, Pool-Goudzwaard AL (2008) Biomechanical model study of pelvic belt influence on muscle and ligament forces. J Biomech 41(9):1878–1884
Peng Y, Zhao Z, Zhou M, He J, Yang J, Xiao Y (2017) Flexible multibody model and the dynamics of the deployment of mesh antennas. J Guid Control Dyn 40(6):1499–1510
Pool-Goudzwaard A, Hoek Van Dijke G, Van Gurp M, Mulder P, Snijders C, Stoeckart R (2004) Contribution of pelvic floor muscles to stiffness of the pelvic ring. Clin Biomech 19(6):564–571
Rasmussen J, de Zee M, Damsgaard M, Christensen ST, Marek C, Siebertz K (2005) A general method for scaling musculo-skeletal models. In: 2005 international symposium on computer simulation in biomechanics
Ratnovsky A, Elad D, Halpern P (2008) Mechanics of respiratory muscles. Respir Physiol Neuro 163(1):82–89
Rupp TK, Ehlers W, Karajan N, Günther M, Schmitt S (2015) A forward dynamics simulation of human lumbar spine flexion predicting the load sharing of intervertebral discs, ligaments, and muscles. Biomech Model Mechanobiol 14(5):1081–1105
Saxby DJ, Killen BA, Pizzolato C, Carty CP, Diamond LE, Modenese L, Fernandez J, Davico G, Barzan M, Lenton G, da Luz SB, Suwarganda E, Devaprakash D, Korhonen RK, Alderson JA, Besier TF, Barrett RS, Lloyd DG (2020) Machine learning methods to support personalized neuromusculoskeletal modelling. Biomech Model Mechanobiol 19(4):1169–1185
Silva MPT, Ambrósio JAC (2003) Solution of redundant muscle forces in human locomotion with multibody dynamics and optimization tools. Mech Based Des Struct Mech 31(3):381–411
Stokes IAF, Gardner-Morse MG, Henry SM (2010) Intra-abdominal pressure and abdominal wall muscular function: spinal unloading mechanism. Clin Biomech 25(9):859–866
Sun J, Tian Q, Hu H, Pedersen NL (2019) Axially variable-length solid element of absolute nodal coordinate formulation. Acta Mech Sin 35(3):653–663
Takai Y, Katsumata Y, Kawakami Y, Kanehisa H, Fukunaga T (2011) Ultrasound method for estimating the cross-sectional area of the psoas major muscle. Med Sci Sports Exerc 43(10):2000–2004
Talmadge RJ, Roy RR, Caiozzo VJ, Edgerton VR (2002) Mechanical properties of rat soleus after long-term spinal cord transection. J Appl Physiol 93(4):1487–1497
Thelen DG (2003) Adjustment of muscle mechanics model parameters to simulate dynamic contractions in older adults. J Biomech Eng Trans ASME 125(1):70–77
Todros S, de Niccolò C, Concheri G, Natali AN, Pavan PG (2020) Numerical modelling of abdominal wall mechanics: the role of muscular contraction and intra-abdominal pressure. J Mech Behav Biomed Mater 103(103):578
Tran D, Podwojewski F, Beillas P, Ottenio M, Voirin D, Turquier F, Mitton D (2016) Abdominal wall muscle elasticity and abdomen local stiffness on healthy volunteers during various physiological activities. J Mech Behav Biomed Mater 60:451–459
Tuktamyshev V, Kasatova E, Truhacheva E, Kasatov A (2016) The study of compressibility of abdominal contents. Russ J Biomech 20(4):261–265
Tzelepis GE, Nasiff L, McCool FD, Hammond J (1996) Transmission of pressure within the abdomen. J Appl Physiol 81(3):1111–1114
Urquhart DM, Barker PJ, Hodges PW, Story IH, Briggs CA (2005) Regional morphology of the transversus abdominis and obliquus internus and externus abdominis muscles. Clin Biomech 20(3):233–241
Vinnars E, Bergstöm J, Fürst P (1975) Influence of the postoperative state on the intracellular free amino acids in human muscle tissue. Ann Surg 182(6):665–671
Wirtz D, Karajan N, Haasdonk B (2015) Surrogate modeling of multiscale models using kernel methods. Int J Numer Methods Eng 101(1):1–28
Wynarsky GT, Schultz AB (1991) Optimization of skeletal configuration: Studies of scoliosis correction biomechanics. J Biomech 24(8):721–732
Zanoni A, Cocco A, Masarati P (2020) Multibody dynamics analysis of the human upper body for rotorcraft-pilot interaction. Nonlinear Dyn 102(3):1517–1539
Zatsiorsky VM, Prilutsky BI (2012) Biomechanics of skeletal muscles. Human Kinetics, Champaign
de Zee M, Hansen L, Wong C, Rasmussen J, Simonsen EB (2007) A generic detailed rigid-body lumbar spine model. J Biomech 40(6):1219–1227
Acknowledgements
This work was supported in part by the China Postdoctoral Science Foundation under Grant 2020TQ0042, and Air Force Equipment Division under Grant KJ20182A030135. We are grateful to Prof. Qiang Tian and Mr. Peishui Zhang for their helpful comments and revisions of this paper.
Author information
Authors and Affiliations
Corresponding author
Appendices
Appendix A: Validation of the proposed muscular membrane element
Benchmark problem of the muscle membrane under pressure loading. a Membrane element discretization under simply supported boundary condition; b cable element discretization (Guo et al. 2020a) under simply supported boundary condition; c membrane element discretization under fixed boundary condition
1.1 Pressure compression benchmark
As shown in Fig. 15, the muscle membrane is under sinusoidal pressure \(p=(1 + 6.65 \sin {2 \pi t}){}{\hbox {kPa}}\), the amplitude of which represents the standing coughing state (Hoyte et al. 2004). A Cartesian coordinate system \(O-xyz\) was created with its origin located at the mass center of the muscle membrane. The muscle fiber was set along the y-axis under activation \(a = 0.05,~0.5,~1.0\). The muscular geometry and strength were adopted from the RA muscle in Bruno et al. (2015), and other parameters were taken from Pato and Areias (2010), as listed in Table 7.
In anatomy, in addition to muscle/bone insertion, other boundaries of RA are constrained by extramuscular connective tissues. As shown in Fig. 15, four different models were built here to understand the role of boundary conditions and inter-fiber tissue stiffness:
-
1.
The muscle was discretized by \(7 \times 1\) cable elements (Guo et al. 2020a), and its origin/insertion points were connected to the ground by a spherical joint.
-
2.
The muscle was discretized by \(7 \times 7\) membrane elements, taking only \(\varvec{\sigma }^{\rm {fiber}}\) into account, and its origin/insertion points were connected to the ground by a spherical joint.
-
3.
The muscle was discretized by \(7 \times 7\) membrane elements, taking \(\varvec{\sigma }^{\rm {fiber}}\), \(\varvec{\sigma }^{\rm {mat}}\), and \(\varvec{\sigma }^{\rm {bulk}}\) into account, and its origin/insertion points were connected to the ground by a spherical joint.
-
4.
The muscle was discretized by \(7 \times 7\) membrane elements, taking \(\varvec{\sigma }^{\rm {fiber}}\), \(\varvec{\sigma }^{\rm {mat}}\), and \(\varvec{\sigma }^{\rm {bulk}}\) into account, and all its boundaries were connected to the ground by a spherical joint.
The membrane deflection profile is illustrated in Fig. 16. Under the maximum pressure, this membrane deformed to a catenary curve along the y-axis at \(x=0\), and its maximum deflection occurred at the center of mass (Fig. 16a and b). When stimulated by minimal activation, the deflection of this muscle membrane behaves nonlinearly with the pressure change, yet the nonlinearity diminishes with the increase in the activation level (Fig. 16c and d). Moreover, the membrane deformation of Models 1 and 2 was identical, validating the proposed membrane element.
According to Fig. 16, different constraints and internal loading conditions strongly affected the membrane deformation distributions. To further analyze these effects, we further depicted the deformation distribution at different muscle activation levels in Fig. 17. As for Model 3, the compressive resistance of the muscle membrane originated from its contractile force at the maximum activation, producing the same results as Model 2. However, introducing \(\varvec{\sigma }^{\rm {mat}}\) and \(\varvec{\sigma }^{\rm {bulk}}\) increased the membrane stiffness when \(a=0.05\). Furthermore, the magnitude of the membrane displacement in Model 4 can be reduced by constraining the lateral boundaries.
1.2 Musculotendon benchmark
Another validation benchmark is based on the simulations performed by Millard et al. (2013). As shown in Fig. 18a, the musculotendon actuator consisted of a pennated muscle belly and a long tendon. The initial length of this whole actuator was \(l^{\rm {fiber}}_0 \cos {\alpha _0} + l^{\rm {tend}}_0\), where \(\alpha _0 = 30^\circ \) denotes the pennation angle. The left end of this actuator was fixed to the ground, and a sinusoidal displacement curve \(u(t) = l^{\rm {fiber}}_0 {\rm {sin}} 2 \pi t\) was applied to its free end. Meanwhile, the muscle was stimulated by constant activations ranged from 0.1 to 1. In the numerical model, each half of the muscle belly was discretized by a membrane element, and the \(\varvec{\sigma }^{\rm {mat}}\) and \(\varvec{\sigma }^{\rm {bulk}}\) were neglected within the membrane element to mimic the Hill-type nonlinear spring. Moreover, according to Günther et al. (2007), a linear damper was introduced and positioned in parallel with the tendon element to stabilize the forward dynamics integration. The musculotendon geometry and strength parameters were adopted from Millard et al. (2013), and its density values were taken from Talmadge et al. (2002), as listed in Table 8.
As shown in Fig. 19a, the fiber length change predicted by our membrane model is consistent with Millard et al. (2013). During the tendon lengthening, its passive tension tracts the muscle fibers toward the longitudinal direction, decreasing the pennation angle to \(\approx 20^\circ \). Afterward, the diminishing of tendon traction sharply increased the pennation angle to \(\approx 90^\circ \) regardless of the activation level. Compared with Millard et al. (2013), the flexible multibody model presented almost the same pennation results except for the slack stage (Fig. 19b).
The musculotendon force results are further depicted in Fig. 19c and d. Compared with Millard et al. (2013), our membrane model performed a quantitatively different muscle force configuration (Fig. 19c) because of the differences of the Hill-type relation. Note that the equilibrium equations of the proposed flexible element were cast in weak form. As a result, the summation of fiber forces projected to the longitudinal direction was not identical to the tendon one (Fig. 19d). Moreover, by including a damper in series (Günther et al. 2007), the membrane model with distributed mass did not perform any oscillation even stimulated, improving the performances compared with Millard and Delp (2012).
Simulation results of the musculotendon model of different activation levels (\(a = 0.1,~0.5,~1.0\)). Here, the solid line indicates our results, and the dashed line indicates the results performed by Millard et al. (2013); a muscle fiber length; b pennation angle; c muscle stress normalized by \(\sigma ^{\rm {fiber}}_0\); d tendon stress normalized by \(\sigma ^{\rm {fiber}}_0\). Norm. \(=\) normalized
Appendix B: Adjustment of diaphragmatic thickness using in vivo measurements
To obtain the diaphragmatic thickness in vivo, we performed an ultrasound-based measurement based on seven male participants (\(19.9 \pm 1.0\) years, \(m=61.6 \pm {2.3}{\hbox {kg}}\), \(h=170.9 \pm {1.2}{\hbox {cm}}\)). The study protocol was approved by the Ethical Committee of Air Force Medical Center, PLA (2020-157-PJ01), and the written informed consent was obtained from each subject. Following the procedure presented by Goligher et al. (2015), a linear array transducer was placed in the ninth or tenth intercostal place near the mid-auxiliary line. The diaphragmatic thickness was measured at end-expiration and peak-inspiration instants (Table 9), and the observed data were then averaged to obtain the subject-specific thickness value.
Meanwhile, to ensure the physiologic consistency between the measured thickness and the other PCSA values, we also measured the PS thickness at the L4-L5 level by B-mode ultrasound. According to Takai et al. (2011), the PS cross-sectional area was then obtained using linear interpolation. Finally, a linear scaling law (Rasmussen et al. 2005) is adopted to obtain the diaphragmatic thickness (\({4.46}{\hbox {mm}}\)) in this model.
Appendix C: Sensitivity results of different modeling parameters
Sensitivity study of muscle activations and IVD compression to muscle stiffness. a Activation with different \(\sigma ^{\rm {fiber}}_0\); b activation under different stages of muscle deterioration; c IVD compression at L3–L4 level with different \(\sigma ^{\rm {fiber}}_0\); (d) IVD compression at L3–L4 level under muscle deterioration
Rights and permissions
About this article
Cite this article
Guo, J., Guo, W. & Ren, G. Embodiment of intra-abdominal pressure in a flexible multibody model of the trunk and the spinal unloading effects during static lifting tasks. Biomech Model Mechanobiol 20, 1599–1626 (2021). https://doi.org/10.1007/s10237-021-01465-1
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10237-021-01465-1