Abstract
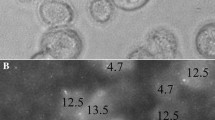
We provide an innovative, bioengineering, mechanobiology-based approach to rapidly (2-h) establish the in vivo metastatic likelihood of patient tumor-samples, where results are in direct agreement with clinical histopathology and patient outcomes. Cancer-related mortality is mostly due to local recurrence or to metastatic disease, thus early prediction of tumor-cell-fate may critically affect treatment protocols and survival rates. Metastasis and recurrence risks are currently predicted by lymph-node status, tumor size, histopathology and genetic testing, however, these are not infallible and results may require days/weeks. We have previously observed that subpopulations of invasive cancer-cells will rapidly (1–2 h) push into the surface of physiological-stiffness, synthetic polyacrylamide gels, reaching to cell-scale depths, while normal or noninvasive cells do not considerably indent gels. Here, we evaluate the mechanical invasiveness of established breast and pancreatic cell lines and of tumor-cells from fresh, suspected pancreatic cancer tumors. The mechanical invasiveness matches the in vitro metastatic potential in cell lines as determined with Boyden chamber assays. Moreover, the mechanical invasiveness directly agrees with the clinical histopathology in primary-site, pancreatic-tumors. Thus, the rapid, patient-specific, early prediction of metastatic likelihood, on the time-scale of initial resection/biopsy, can directly affect disease management and treatment protocols.





Similar content being viewed by others
References
Abuhattum, S., and D. Weihs. Asymmetry in traction forces produced by migrating preadipocytes is bounded to 33. Med. Eng. Phys. 38:834–838, 2016.
Albini, A., and R. Benelli. The chemoinvasion assay: a method to assess tumor and endothelial cell invasion and its modulation. Nat. Protoc. 2:504–511, 2007.
Alvarez-Elizondo, M. B., C. W. Li, A. Marom, Y.-T. Tung, G. Drillich, Y. Horesh, S. C. Lin, G.-J. Wang, and D. Weihs. Micropatterned topographies reveal measurable differences between cancer and benign cells. Med. Eng. Phys. 75:5–12, 2020.
Alvarez-Elizondo, M. B., and D. Weihs. Cell-gel mechanical interactions as an approach to rapidly and quantitatively reveal invasive subpopulations of metastatic cancer cells. Tissue Eng. Part C Methods 23:180–187, 2017.
Artym, V. V., K. M. Yamada, and S. C. Mueller. ECM degradation assays for analyzing local cell invasion. In: Methods in Molecular Biology (Clifton, N.J.). 2009, pp. 211–219.
Carrato, A., A. Falcone, M. Ducreux, J. W. Valle, A. Parnaby, K. Djazouli, K. Alnwick-Allu, A. Hutchings, C. Palaska, and I. Parthenaki. A systematic review of the burden of pancreatic cancer in Europe: real-world impact on survival, quality of life and costs. J. Gastrointest. Cancer 46:201–211, 2015.
Casciani, F., G. Marchegiani, G. Malleo, C. Bassi, and R. Salvia. Pancreatic cancer in the era of neoadjuvant therapy: a narrative overview. Chirurgia (Bucur) 113:307, 2018.
Cross, S. E., Y. S. Jin, J. Rao, and J. K. Gimzewski. Nanomechanical analysis of cells from cancer patients. Nat. Nanotechnol. 2:780–783, 2007.
Deer, E. L., J. Gonzalez-Hernandez, J. D. Coursen, J. E. Shea, J. Ngatia, C. L. Scaife, M. A. Firpo, and S. J. Mulvihill. Phenotype and genotype of pancreatic cancer cell lines. Pancreas 39:425–435, 2010.
Dvir, L., R. Nissim, M. B. Alvarez-Elizondo, and D. Weihs. Quantitative measures to reveal coordinated cytoskeleton-nucleus reorganization during in vitro invasion of cancer cells. New J. Phys. 17:043010, 2015.
Eslami Amirabadi, H., S. Saheb Ali, J. P. Frimat, R. Luttge, and J. M. J. Den Toonder. A novel method to understand tumor cell invasion: integrating extracellular matrix mimicking layers in microfluidic chips by “selective curing”. Biomed. Microdevices 19:1–11, 2017.
Friedl, P., and K. Wolf. Tumour-cell invasion and migration: diversity and escape mechanisms. Nat. Rev. Cancer 3:362–374, 2003.
Friedl, P., and K. Wolf. Plasticity of cell migration: a multiscale tuning model. J. Cell Biol. 188:11–19, 2010.
Gal, N., D. Lechtman-Goldstein, and D. Weihs. Particle tracking in living cells: a review of the mean square displacement method and beyond. Rheol. Acta 52:425–443, 2013.
Guck, J., S. Schinkinger, B. Lincoln, F. Wottawah, S. Ebert, M. Romeyke, D. Lenz, H. M. Erickson, R. Ananthakrishnan, D. Mitchell, J. Kas, S. Ulvick, and C. Bilby. Optical deformability as an inherent cell marker for testing malignant transformation and metastatic competence. Biophys. J. 88:3689–3698, 2005.
Gurcan, M. N., L. E. Boucheron, A. Can, A. Madabhushi, N. M. Rajpoot, and B. Yener. Histopathological image analysis: a review. IEEE Rev. Biomed. Eng. 2:147–171, 2009.
Holle, A. W., N. Govindan Kutty Devi, K. Clar, A. Fan, T. Saif, R. Kemkemer, and J. P. Spatz. Cancer cells invade confined microchannels via a self-directed mesenchymal-to-amoeboid transition. Nano Lett. 19:2280–2290, 2019.
Holliday, D. L., and V. Speirs. Choosing the right cell line for breast cancer research. Breast Cancer Res. 13:215, 2011.
Koch, T. M., S. Munster, N. Bonakdar, J. P. Butler, and B. Fabry. 3D Traction forces in cancer cell invasion. PLoS ONE 7:e33476, 2012.
Kraning-Rush, C. M., J. P. Califano, and C. A. Reinhart-King. Cellular traction stresses increase with increasing metastatic potential. PLoS ONE 7:e32572, 2012.
Kristal-Muscal, R., L. Dvir, and D. Weihs. Metastatic cancer cells tenaciously indent impenetrable, soft substrates. New J. Phys. 15:035022, 2013.
Levental, K. R., H. Yu, L. Kass, J. N. Lakins, M. Egeblad, J. T. Erler, S. F. T. Fong, K. Csiszar, A. Giaccia, W. Weninger, M. Yamauchi, D. L. Gasser, and V. M. Weaver. Matrix crosslinking forces tumor progression by enhancing integrin signaling. Cell 139:891–906, 2009.
Li, X., A. V. Valadez, P. Zuo, and Z. Nie. Microfluidic 3D cell culture: potential application for tissue-based bioassays. Bioassays. 4(12):1509–1525, 2012.
Liang, S. B., and L. W. Fu. Application of single-cell technology in cancer research. Biotechnol. Adv. 35(4):443–449, 2017.
Lloyd, S. Least squares quantization in PCM. IEEE Trans. Inf. Theory 28:129–137, 1982.
Massalha, S., and D. Weihs. Metastatic breast cancer cells adhere strongly on varying stiffness substrates, initially without adjusting their morphology. Biomech. Model. Mechanobiol. 16:961–970, 2017.
Merkher, Y., M. B. Alvarez-Elizondo, and D. Weihs. Taxol reduces synergistic, mechanobiological invasiveness of metastatic cells. Converg. Sci. Phys. Oncol. 3:044002, 2017.
Merkher, Y., and D. Weihs. Proximity of metastatic cells enhances their mechanobiological invasiveness. Ann. Biomed. Eng. 45:1399–1406, 2017.
Nyström, M., G. Thomas, M. Stone, I. Mackenzie, I. Hart, and J. Marshall. Development of a quantitative method to analyse tumour cell invasion in organotypic culture. J. Pathol. 205:468–475, 2005.
Peixoto, R. D., C. Speers, C. E. McGahan, D. J. Renouf, D. F. Schaeffer, and H. F. Kennecke. Prognostic factors and sites of metastasis in unresectable locally advanced pancreatic cancer. Cancer Med. 4:1171–1177, 2015.
Rahib, L., B. D. Smith, R. Aizenberg, A. B. Rosenzweig, J. M. Fleshman, and L. M. Matrisian. Projecting cancer incidence and deaths to 2030: the unexpected burden of thyroid, liver, and pancreas cancers in the United States. Cancer Res. 74:2913–2921, 2014.
Riihimaki, M., H. Thomsen, A. Hemminki, K. Sundquist, K. Hemminki, M. Riihimäki, H. Thomsen, A. Hemminki, K. Sundquist, K. Hemminki, M. Riihimaki, H. Thomsen, A. Hemminki, K. Sundquist, and K. Hemminki. Comparison of survival of patients with metastases from known versus unknown primaries: survival in metastatic cancer. BMC Cancer 13:36, 2013.
Sieuwerts, A. M., J. G. Klijn, and J. A. Foekens. Assessment of the invasive potential of human gynecological tumor cell lines with the in vitro Boyden chamber assay: influences of the ability of cells to migrate through the filter membrane. Clin. Exp. Metastasis 15:53–62, 1997.
Sleeman, J. P., I. Nazarenko, and W. Thiele. Do all roads lead to Rome? Routes to metastasis development. Int. J. Cancer 128:2511–2526, 2011.
Suker, M., B. R. Beumer, E. Sadot, L. Marthey, J. E. Faris, E. A. Mellon, B. F. El-Rayes, A. Wang-Gillam, J. Lacy, P. J. Hosein, S. Y. Moorcraft, T. Conroy, F. Hohla, P. Allen, J. Taieb, T. S. Hong, R. Shridhar, I. Chau, C. H. van Eijck, and B. G. Koerkamp. FOLFIRINOX for locally advanced pancreatic cancer: a systematic review and patient-level meta-analysis. Lancet Oncol. 17:801–810, 2016.
Sung, K. E., and D. J. Beebe. Microfluidic 3D models of cancer. Adv. Drug Deliv. Rev. 79–80:68–78, 2014.
Swaminathan, V., K. Mythreye, E. T. O’Brien, A. Berchuck, G. C. Blobe, and R. Superfine. Mechanical stiffness grades metastatic potential in patient tumor cells and in cancer cell lines. Cancer Res. 71:5075–5080, 2011.
Teng, Y., X. Xie, S. Walker, D. T. White, J. S. Mumm, and J. K. Cowell. Evaluating human cancer cell metastasis in zebrafish. BMC Cancer 13:453, 2013.
Toh, Y. C., A. Raja, H. Yu, and D. Van Noort. A 3D microfluidic model to recapitulate cancer cell migration and invasion. Bioengineering 5:10, 2018.
Truong, H. H., J. de Sonneville, V. P. S. S. Ghotra, J. Xiong, L. Price, P. C. W. W. Hogendoorn, H. H. Spaink, B. van de Water, and E. H. J. J. Danen. Automated microinjection of cell-polymer suspensions in 3D ECM scaffolds for high-throughput quantitative cancer invasion screens. Biomaterials 33:181–188, 2012.
Vinci, M., C. Box, and S. A. Eccles. Three-dimensional (3D) tumor spheroid invasion assay. J. Vis. Exp 2015. https://doi.org/10.3791/52686.
Wei, J. W., L. J. Tafe, Y. A. Linnik, L. J. Vaickus, N. Tomita, and S. Hassanpour. Pathologist-level classification of histologic patterns on resected lung adenocarcinoma slides with deep neural networks. Sci. Rep. 9:3358, 2019.
Weigelt, B., J. L. Peterse, and L. J. van’t Veer. Breast cancer metastasis markers and models. Nat. Rev. Cancer 5:591–602, 2005.
Weihs, D., and Y. Merkher. A device and method for determining cell indention activity, Patent pending. Patent: PCT/IL2019/050463, 2019.
Yankaskas, C. L., K. N. Thompson, C. D. Paul, M. I. Vitolo, P. Mistriotis, A. Mahendra, V. K. Bajpai, D. J. Shea, K. M. Manto, A. C. Chai, N. Varadarajan, et al. A microfluidic assay for the quantification of the metastatic propensity of breast cancer specimens. Nat. Biomed. Eng. 3:452–465, 2019.
Acknowledgments
The work was partially funded by the Technion Internal Elias Fund for Medical Research and by Polak Fund for Applied Research, and by the Ber-Lehmsdorf Foundation and the Gerald O. Mann Charitable Foundation.
Author information
Authors and Affiliations
Corresponding author
Additional information
Associate Editor Konstantinos Konstantopoulos oversaw the review of this article.
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Electronic supplementary material
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Merkher, Y., Horesh, Y., Abramov, Z. et al. Rapid Cancer Diagnosis and Early Prognosis of Metastatic Risk Based on Mechanical Invasiveness of Sampled Cells. Ann Biomed Eng 48, 2846–2858 (2020). https://doi.org/10.1007/s10439-020-02547-4
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10439-020-02547-4