Abstract
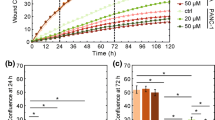
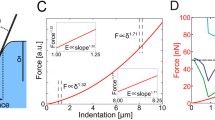
We demonstrate the relative roles of the cell cytoskeleton, and specific importance of actin in facilitating mechanical aspects of metastatic invasion. A crucial step in metastasis, the typically lethal spread of cancer to distant body-sites, is cell invasion through dense tissues composed of extracellular matrix and various non-cancerous cells. Cell invasion requires cell-cytoskeleton remodeling to facilitate dynamic morphological changes and force application. We have previously shown invasive cell subsets in heterogeneous samples can rapidly (2 h) and forcefully indent non-degradable, impenetrable, synthetic gels to cell-scale depths. The amounts of indenting cells and their attained depths provide the mechanical invasiveness of the sample, which as we have shown agrees with the in vitro metastatic potential and the in vivo metastatic risk in humans. To identify invasive force-application mechanisms, we evaluated changes in mechanical invasiveness following chemical perturbations targeting the structure and function of cytoskeleton elements and associated proteins. We evaluate effects on short-term (2-hr) indentations of single, well-spaced or closely situated cells as compared to long-time-scale Boyden chamber migration. We show that actomyosin inhibition may be used to reduce (mechanical) invasiveness of single or collectively invading cells, while actin-disruption may induce escape-response of treated single-cells, which may promote metastasis.





Similar content being viewed by others
References
Alvarez-Elizondo, M. B., C. W. Li, A. Marom, Y.-T. Tung, G. Drillich, Y. Horesh, S. C. Lin, G.-J. Wang, and D. Weihs. Micropatterned topographies reveal measurable differences between cancer and benign cells. Med. Eng. Phys. 75:5–12, 2020.
Alvarez-Elizondo, M. B., and D. Weihs. Cell-gel mechanical interactions as an approach to rapidly and quantitatively reveal invasive subpopulations of metastatic cancer cells. Tissue Eng. Part C Methods 23:180–187, 2017.
Blajeski, A. L., V. A. Phan, T. J. Kottke, and S. H. Kaufmann. G1 and G2 cell-cycle arrest following microtubule depolymerization in human breast cancer cells. J. Clin. Invest. 110:91–99, 2002.
Caswell, P. T., and T. Zech. Actin-based cell protrusion in a 3D matrix. Trends Cell Biol. 28:823–834, 2018.
Cheung, K. J., V. Padmanaban, V. Silvestri, K. Schipper, J. D. Cohen, A. N. Fairchild, M. A. Gorin, J. E. Verdone, K. J. Pienta, J. S. Bader, and A. J. Ewald. Polyclonal breast cancer metastases arise from collective dissemination of keratin 14-expressing tumor cell clusters. Proc. Natl. Acad. Sci. USA 113:E854–E863, 2016.
Clark, A. G. A. G., and D. M. D. M. Vignjevic. Modes of cancer cell invasion and the role of the microenvironment. Curr. Opin. Cell Biol. 36:13–22, 2015.
Dudaie, M., D. Weihs, F. J. Vermolen, and A. Gefen. Modeling migration in cell colonies in two and three dimensional substrates with varying stiffnesses. Silico Cell Tissue Sci. 2:1–14, 2015.
Dvir, L., R. Nissim, M. B. Alvarez-Elizondo, and D. Weihs. Quantitative measures to reveal coordinated cytoskeleton-nucleus reorganization during in vitro invasion of cancer cells. N. J. Phys. 17:043010, 2015.
Fidler, I. J. In vitro studies of cellular-mediated immunostimulation of tumor growth. J. Natl. Cancer Inst. 50:1307–1312, 1973.
Fife, C. M., J. A. McCarroll, and M. Kavallaris. Movers and shakers: cell cytoskeleton in cancer metastasis. Br. J. Pharmacol. 171:5507–5523, 2014.
Friedl, P., and S. Alexander. Cancer invasion and the microenvironment: plasticity and reciprocity. Cell 147:992–1009, 2011.
Friedl, P., J. Locker, E. Sahai, and J. E. Segall. Classifying collective cancer cell invasion. Nat. Cell Biol. 14:777–783, 2012.
Gal, N., and D. Weihs. Intracellular mechanics and activity of breast cancer cells correlate with metastatic potential. Cell Biochem. Biophys. 63:199–209, 2012.
Galbraith, C. G., K. M. Yamada, and M. P. Sheetz. The relationship between force and focal complex development. J. Cell Biol. 159:695–705, 2002.
Geiger, B., and K. M. Yamada. Molecular architecture and function of matrix adhesions. Cold Spring Harb. Perspect. Biol. 3:a005033, 2011.
Ghibaudo, M., A. Saez, L. Trichet, A. Xayaphoummine, J. Browaeys, P. Silberzan, A. Buguin, and B. Ladoux. Traction forces and rigidity sensing regulate cell functions. Soft Matter 4:1836–1843, 2008.
Gladilin, E., S. Ohse, M. Boerries, H. Busch, C. Xu, M. Schneider, M. Meister, and R. Eils. TGFβ-induced cytoskeletal remodeling mediates elevation of cell stiffness and invasiveness in NSCLC. Sci. Rep. 9:1–12, 2019.
Goldstein, D., T. Elhanan, M. Aronovitch, and D. Weihs. Origin of active transport in breast-cancer cells. Soft Matter 9:7167–7173, 2013.
Guck, J., S. Schinkinger, B. Lincoln, F. Wottawah, S. Ebert, M. Romeyke, D. Lenz, H. M. Erickson, R. Ananthakrishnan, D. Mitchell, J. Kas, S. Ulvick, and C. Bilby. Optical deformability as an inherent cell marker for testing malignant transformation and metastatic competence. Biophys. J. 88:3689–3698, 2005.
Guzman, A., M. J. Ziperstein, and L. J. Kaufman. The effect of fibrillar matrix architecture on tumor cell invasion of physically challenging environments. Biomaterials 35:6954–6963, 2014.
Haeger, A., S. Alexander, M. Vullings, F. M. P. Kaiser, C. Veelken, U. Flucke, G. E. Koehl, M. Hirschberg, M. Flentje, R. M. Hoffman, E. K. Geissler, S. Kissler, and P. Friedl. Collective cancer invasion forms an integrin-dependent radioresistant niche. J. Exp. Med. 217(1):e20181184, 2020.
Isogai, T., R. van der Kammen, and M. Innocenti. SMIFH2 has effects on formins and p53 that perturb the cell cytoskeleton. Sci. Rep. 5:9802, 2015.
Khalil, A. A., O. Ilina, P. G. Gritsenko, P. Bult, P. N. Span, and P. Friedl. Collective invasion in ductal and lobular breast cancer associates with distant metastasis. Clin. Exp. Metastasis 34:421–429, 2017.
Kraning-Rush, C. M., J. P. Califano, and C. A. Reinhart-King. Cellular traction stresses increase with increasing metastatic potential. PLoS ONE 7:e32572, 2012.
Kristal-Muscal, R., L. Dvir, and D. Weihs. Metastatic cancer cells tenaciously indent impenetrable, soft substrates. N. J. Phys. 15:035022, 2013.
Li, Q., Z. Ma, Y. Liu, X. Kan, C. Wang, B. Su, Y. Li, Y. Zhang, P. Wang, Y. Luo, D. Na, L. Wang, G. Zhang, X. Zhu, and L. Wang. Low doses of paclitaxel enhance liver metastasis of breast cancer cells in the mouse model. FEBS J. 283:2836–2852, 2016.
Mak, M., C. A. Reinhart-King, and D. Erickson. Elucidating mechanical transition effects of invading cancer cells with a subnucleus-scaled microfluidic serial dimensional modulation device. Lab Chip 13:340–348, 2013.
Massalha, S., and D. Weihs. Metastatic breast cancer cells adhere strongly on varying stiffness substrates, initially without adjusting their morphology. Biomech. Model. Mechanobiol. 16:961–970, 2017.
Merkher, Y., M. B. Alvarez-Elizondo, and D. Weihs. Taxol reduces synergistic, mechanobiological invasiveness of metastatic cells. Converg. Sci. Phys. Oncol. 3:044002, 2017.
Merkher, Y., Y. Horesh, Z. Abramov, G. Shleifer, O. Ben-Ishay, Y. Kluger, and D. Weihs. Rapid cancer diagnosis and early prognosis of metastatic risk based on mechanical invasiveness of sampled cells. Ann. Biomed. Eng. 2020. https://doi.org/10.1007/s10439-020-02547-4.
Merkher, Y., and D. Weihs. Proximity of metastatic cells enhances their mechanobiological invasiveness. Ann. Biomed. Eng. 45:1399–1406, 2017.
Mierke, C. T. The biomechanical properties of 3d extracellular matrices and embedded cells regulate the invasiveness of cancer cells. Cell Biochem. Biophys. 61:217–236, 2011.
Mierke, C. T., B. Frey, M. Fellner, M. Herrmann, and B. Fabry. Integrin alpha5beta1 facilitates cancer cell invasion through enhanced contractile forces. J. Cell Sci. 124:369–383, 2011.
Mohammadi, A., B. Mansoori, M. Aghapour, S. Shirjang, S. Nami, and B. Baradaran. The urtica dioica extract enhances sensitivity of paclitaxel drug to MDA-MB-468 breast cancer cells. Biomed. Pharmacother. 83:835–842, 2016.
O’Shaughnessy, J., W. J. Gradishar, P. Bhar, and J. Iglesias. nab-Paclitaxel for first-line treatment of patients with metastatic breast cancer and poor prognostic factors: a retrospective analysis. Breast Cancer Res. Treat. 138:829–837, 2013.
Patsialou, A., J. J. Bravo-Cordero, Y. Wang, D. Entenberg, H. Liu, M. Clarke, and J. S. Condeelis. Intravital multiphoton imaging reveals multicellular streaming as a crucial component of in vivo cell migration in human breast tumors. Intravital 2:e25294, 2013.
Peterson, J. A., B. Tian, A. D. Bershadsky, T. Volberg, R. E. Gangnon, I. Spector, B. Geiger, and P. L. Kaufman. Latrunculin-A increases outflow facility in the monkey. Investig. Ophthalmol. Vis. Sci. 40:931–941, 1999.
Poincloux, R., O. Collin, F. Lizarraga, M. Romao, M. Debray, M. Piel, and P. Chavrier. Contractility of the cell rear drives invasion of breast tumor cells in 3D matrigel. Proc. Natl. Acad. Sci. USA 108:1943–1948, 2011.
Seetharaman, S., and S. Etienne-Manneville. Cytoskeletal crosstalk in cell migration. Trends Cell Biol. 30:720–735, 2020.
Sen, S., A. J. Engler, and D. E. Discher. Matrix strains induced by cells: computing how far cells can feel. Cell. Mol. Bioeng. 2:39–48, 2009.
Stearns, M. E., and M. Wang. Taxol blocks processes essential for prostate tumor cell (PC-3 ML) invasion and metastases. Cancer Res. 52:3776–3781, 1992.
Stehn, J. R., N. K. Haass, T. Bonello, M. Desouza, G. Kottyan, H. Treutlein, J. Zeng, P. R. B. B. Nascimento, V. B. Sequeira, T. L. Butler, M. Allanson, T. Fath, T. A. Hill, A. McCluskey, G. Schevzov, S. J. Palmer, E. C. Hardeman, D. Winlaw, V. E. Reeve, I. Dixon, W. Weninger, T. P. Cripe, and P. W. Gunning. A novel class of anticancer compounds targets the actin cytoskeleton in tumor cells. Cancer Res. 73:5169–5182, 2013.
Swaminathan, V., K. Mythreye, E. T. O’Brien, A. Berchuck, G. C. Blobe, and R. Superfine. Mechanical stiffness grades metastatic potential in patient tumor cells and in cancer cell lines. Cancer Res. 71:5075–5080, 2011.
Tozluoglu, M., A. L. Tournier, R. P. Jenkins, S. Hooper, P. A. Bates, and E. Sahai. Matrix geometry determines optimal cancer cell migration strategy and modulates response to interventions. Nat. Cell Biol. 15:751–762, 2013.
Volk-Draper, L., K. Hall, C. Griggs, S. Rajput, P. Kohio, D. DeNardo, and S. Ran. Paclitaxel therapy promotes breast cancer metastasis in a TLR4-dependent manner. Cancer Res. 74:5421–5434, 2014.
Wang, N., and D. E. Ingber. Probing transmembrane mechanical coupling and cytomechanics using magnetic twisting cytometry. Biochem. Cell Biol. 73:327–335, 1995.
Weihs, D., Y. Merkher. A device and method for determining cell indention activity, Patent pending. Patent: PCT/IL2019/050463, 2019.
Weihs, D., T. G. Mason, and M. A. Teitell. Effects of cytoskeletal disruption on transport, structure, and rheology within mammalian cells. Phys. Fluids 19:103102, 2007.
Wyckoff, J. B., S. E. Pinner, S. Gschmeissner, J. S. Condeelis, and E. Sahai. ROCK- and myosin-dependent matrix deformation enables protease-independent tumor-cell invasion in vivo. Curr. Biol. 16:1515–1523, 2006.
Wyse, M. M., J. Lei, A. L. Nestor-Kalinoski, and K. M. Eisenmann. Dia-interacting protein (DIP) imposes migratory plasticity in mDia2-dependent tumor cells in three-dimensional matrices. PLoS ONE 7:e45085, 2012.
Yeung, T., P. C. Georges, L. A. Flanagan, B. Marg, M. Ortiz, M. Funaki, N. Zahir, W. Ming, V. Weaver, and P. A. Janmey. Effects of substrate stiffness on cell morphology, cytoskeletal structure, and adhesion. Cell Motil. Cytoskeleton 60:24–34, 2005.
Yilmaz, M., and G. Christofori. Mechanisms of motility in metastasizing cells. Mol. Cancer Res. 8:629–642, 2010.
Yizraeli, M. L., and D. Weihs. Time-dependent micromechanical responses of breast cancer cells and adjacent fibroblasts to electric treatment. Cell Biochem. Biophys. 61:605–618, 2011.
Zhang, Y., Y. Wang, and J. Xue. Paclitaxel inhibits breast cancer metastasis via suppression of aurora kinase-mediated cofilin-1 activity. Exp. Ther. Med. 15:1269–1276, 2018.
Acknowledgments
The authors thank Mr. Yam Horesh for his assistance with the cell viability determination. The work was partially funded by the Elias Fund for Medical Research, the Polak Fund for Applied Research, and the Samuel H. Born Fund for Biomedical Research.
Conflict of interest
No benefits in any form have been or will be received from a commercial party related directly or indirectly to the subject of this manuscript.
Author information
Authors and Affiliations
Corresponding author
Additional information
Associate Editor Aleksander S. Popel oversaw the review of this article.
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Electronic supplementary material
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Alvarez-Elizondo, M.B., Merkher, Y., Shleifer, G. et al. Actin as a Target to Reduce Cell Invasiveness in Initial Stages of Metastasis. Ann Biomed Eng 49, 1342–1352 (2021). https://doi.org/10.1007/s10439-020-02679-7
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10439-020-02679-7