Abstract
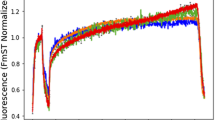
Multi-wavelength Chl a fluorometers are increasingly applied to assess phytoplankton photosynthetic capacity and composition, but their usefulness is limited by uncertainties in fluorescence excitation spectra (FES). We investigated this issue using the Phyto-PAM fluorometer to evaluate the effects of innate and irradiance-dependent variations in background (F) and variable (Fv) FES on analysis of three pigment groups (cyanobacteria, chlorophytes and chromophytes). The effects on group-specific estimates of minimum fluorescence (F0), a proxy for biomass, and Fv/Fm, the quantum yield of photochemistry, presented some challenges to the interpretation of group-specific results. F0 estimates usually had a 5–15% margin of error, even when measuring highly uneven mixtures, and applying imperfectly matched calibration FES or stressing samples with photosynthetically active and ultraviolet radiation; errors in Fv/Fm were commonly < 15%. Despite such relatively good accuracy, estimates for F0 and, especially, Fv/Fm are unreliable for groups at low relative abundance, and results can sometimes be reported for groups not actually present. We report margins of error for different levels of relative abundance to inform interpretation of measurements from natural communities and show that F and Fv spectra for some taxa can differ in ways that produce severe errors in F0 and Fv/Fm estimates if used uncritically.








Similar content being viewed by others
Availability of data
Data are available from authors on request.
Abbreviations
- Chl a :
-
Chlorophyll a
- F 0 :
-
Dark-adapted minimum fluorescence
- F :
-
Low-light adapted minimum fluorescence
- F m :
-
Maximum fluorescence
- F v :
-
Variable fluorescence
- F v/F m :
-
Maximum quantum yield of photochemistry (dark adapted)
- FES:
-
Fluorescence excitation spectrum/spectra
- LED:
-
Light emitting diode
- FP:
-
Fluoroprobe
- NMS:
-
Non-metric multidimensional scaling
- NPQ:
-
Non-photochemical quenching
- P:
-
PAR only treatment
- PA:
-
PAR + UV-A treatment
- PAB:
-
PAR + UV-A + UV-B treatment
- PAM:
-
Pulse amplitude modulation
- PAR:
-
Photosynthetically active radiation
- PC:
-
Phycocyanin
- PE:
-
Phycoerythrin
- PSII:
-
Photosystem II
- RS:
-
Reference Spectrum/Spectra
- UV(R):
-
Ultraviolet (radiation)
References
Aberle N, Beutler M, Moldaenke C, Wiltshire KH (2006) ‘Spectral fingerprinting’ for specific algal groups on sediments in situ: a new sensor. Arch Hydrobiol 167:575–592
Acuna AM, Snellenburg JJ, Gwizdala M, Kirilovsky D, van Grondelle R, van Stokkum IHM (2016) Resolving the contribution of the uncoupled phycobilisomes to cyanobacterial pulse-amplitude modulated (PAM) fluorometry signals. Photosynth Res 127:91–102
Beecraft L, Watson SB, Smith REH (2017) Multi-wavelength pulse amplitude modulated fluorometry (phyto-PAM) reveals differential effects of ultraviolet radiation on the photosynthetic physiology of phytoplankton pigment groups. Freshw Biol 62:72–86
Beecraft L, Watson SB, Smith REH (2019) Innate resistance of PSII efficiency to sunlight stress is not an advantage for cyanobacteria compared to eukaryotic phytoplankton. Aquat Ecol 53:347–364
Brunet C, Johnsen G, Lavaud J, Roy S (2011) Pigments and photoacclimation processes. In: Roy S, Llewellyn CA, Egeland ES, Johnsen G (eds) Phytoplankton pigments: characterization, chemotaxonomy and applications in oceanography. Cambridge University Press, Cambridge, pp 445–471
Catherine A, Escoffier N, Belhocine A, Nasri AB, Hamlaoui S, Yepremian C, Bernard C, Troussellier M (2012) On the use of the FluoroProbe (R), a phytoplankton quantification method based on fluorescence excitation spectra for large-scale surveys of lakes and reservoirs. Water Res 46:1771–1784
de Mendiburu F (2017) Agricolae: statistical procedures for agricultural research. R Foundation for Statistical Computing, Vienna
Escoffier N, Bernard C, Hamlaoui S, Groleau A, Catherine A (2015) Quantifying phytoplankton communities using spectral fluorescence: the effects of species composition and physiological state. J Plankton Res 37:233–247
Falkowski PG, Raven JA (2007) Aquatic photosynthesis, 2nd edn. Princeton University Press, Princeton
Fox J, Weisberg S (2011) An {R} companion to applied regression, 2nd edn. Sage Publications, London
Gaevsky N, Kolmakov V, Anishchenko O, Gorbaneva T (2005) Using DCMU-fluorescence method for the identification of dominant phytoplankton groups. J Appl Phycol 17:483–494
Garrido M, Cecchi P, Malet N, Bec B, Torre F, Pasqualini V (2019) Evaluation of FluoroProbe® performance for the phytoplankton-based assessment of the ecological status of Mediterranean coastal lagoons. Environ Monit Assess 191:204
Genty B, Briantais JM, Baker NR (1989) The relationship between the quantum yield of photosynthetic electron-transport and quenching of chlorophyll fluorescence. Biochim Biophys Acta 990:87–92
Goldman EA, Smith EM, Richardson TL (2013) Estimation of chromophoric dissolved organic matter (CDOM) and photosynthetic activity of estuarine phytoplankton using a multiple-fixed-wavelength spectral fluorometer. Water Res 47:1616–1630
Goss R, Jakob T (2010) Regulation and function of xanthophyll cycle-dependent photoprotection in algae. Photosynth Res 106:103–122
Gregor J, Geris R, Marsalek B, Hetesa J, Marvan P (2005) In situ quantification of phytoplankton in reservoirs using a submersible spectrofluorometer. Hydrobiologia 548:141–151
Guillard RR, Lorenzen C (1972) Yellow–green algae with chlorophyllide C. J Phycol 8:10–14
Harrison JW, Howell ET, Watson SB, Smith REH (2016) Improved estimates of phytoplankton community composition based on in situ spectral fluorescence: use of ordination and field-derived norm spectra for the bbe FluoroProbe. Can J Fish Aquat Sci 73:1472–1482
Harrison JW, Beecraft L, Smith REH (2018) Implications of irradiance exposure and non-photochemical quenching for multi-wavelength (bbe FluoroProbe) fluorometry. J Photochem Photobiol B Biol 189:36–48
Houliez E, Lizon F, Thyssen M, Artigas LF, Schmitt FG (2012) Spectral fluorometric characterization of haptophyte dynamics using the FluoroProbe: an application in the eastern english channel for monitoring Phaeocystis globosa. J Plankton Res 34:136–151
Huot Y, Babin M (2010) Overview of fluorescence protocols: theory, basic concepts, and practice. In: Suggett DJ, Prášil O, Borowitzka MA (eds) Chlorophyll a fluorescence in aquatic sciences methods and applications. Springer, Dordrecht, New York, p 31
Jakob T, Schreiber U, Kirchesch V, Langner U, Wilhelm C (2005) Estimation of chlorophyll content and daily primary production of the major algal groups by means of multiwavelength-excitation PAM chlorophyll fluorometry: performance and methodological limits. Photosynth Res 83:343–361
Kiefer D (1973) Fluorescence properties of natural phytoplankton populations. Mar Biol 22:263–269
Kirilovsky D (2015) Modulating energy arriving at photochemical reaction centers: orange carotenoid protein-related photoprotection and state transitions. Photosynth Res 126:3–17
Kirk JTO (1994) Light and photosynthesis in aquatic ecosystems. Cambridge University Press, Cambridge
Kolbowski J, Schreiber U (1995) Computer-controlled phytoplankton analyzer based on a 4-wavelengths PAM chlorophyll fluorometer. In: Mathis P (ed) Photosynthesis: from light to biosphere, vol V. Kluwer, Dordrecht, pp 825–828
Kring SA, Figary SE, Boyer GL, Watson SB, Twiss MR (2014) Rapid in situ measures of phytoplankton communities using the bbe FluoroProbe: evaluation of spectral calibration, instrument intercompatibility, and performance range. Can J Fish Aquat Sci 71:1087–1095
Kruskopf M, Flynn KJ (2006) Chlorophyll content and fluorescence responses cannot be used to gauge reliably phytoplankton biomass, nutrient status or growth rate. New Phytol 169(3):525–536
Leboulanger C, Dorigo U, Jacquet S, Le Berre B, Paolini G, Humbert J (2002) Application of a submersible spectrofluorometer for rapid monitoring of freshwater cyanobacterial blooms: a case study. Aquat Microb Ecol 30:83–89
Lorenzen C (1966) A method for the continuous measurement of in vivo chlorophyll concentration. Deep-Sea Res 13:223–227
MacDonald JH (2014) Handbook of biological statistics. Sparky House Publishing, Baltimore
MacIntyre HL, Lawrenz E, Richardson TL (2010) Taxonomic discrimination of phytoplankton by spectral fluorescence. In: Suggett DJ, Prášil O, Borowitzka MA (eds) Chlorophyll a fluorescence in aquatic sciences methods and applications. Springer, Dordrecht, New York, pp 129–169
Maxwell K, Johnson GN (2000) Chlorophyll fluorescence—a practical guide. J Exp Bot 51:659–668
Oksanen J, Blanchet FG, Friendly M, Kindt R, Legendre P, McGlinn D, Minchin PR, O’Hara RB, Simpson GL, Solymos P, Stevens MHH, Szoecs E, Wagner H (2016) Vegan: community ecology package. R package version 2.4-1
Papageorgiou GC, Govindjee (2014) The non-photochemical quenching of the electronically excited state of chlorophyll a in plants: definitions, timelines, viewpoints, open questions. In: Demmig-Adams B, Garab G, Adams W III, Govindjee (eds) Non-photochemical quenching and energy dissipation in plants, algae and cyanobacteria. Springer, Berlin, pp 1–33
Parsons TR, Strickland JDH (1963) Discussion of spectrophotometric determination of marine-plant pigments, with revised equations for ascertaining chlorophylls and carotenoids. J Mar Res 21(3):155–163
Phinney D, Yentsch C (1985) A novel phytoplankton chlorophyll technique—toward automated-analysis. J Plankton Res 7:633–642
R Core Team (2017) R: a language and environment for statistical computing. R Core Team, Vienna
Rastogi RP, Sonani RR, Madamwar D (2015) Effects of PAR and UV radiation on the structural and functional integrity of phycocyanin, phycoerythrin and allophycocyanin isolated from the marine cyanobacterium lyngbya sp A09DM. Photochem Photobiol 91:837–844
Rolland A, Rimet F, Jacquet S (2010) A 2-year survey of phytoplankton in the marne reservoir (France): a case study to validate the use of an in situ spectrofluorometer by comparison with algal taxonomy and chlorophyll a measurements. Knowl Manag Aquat Ecosyst 398:02
Schmitt-Jansen M, Altenburger R (2008) Community-level microalgal toxicity assessment by multiwavelength-excitation PAM fluorometry. Aquat Toxicol 86:49–58
Schreiber U (1998) Chlorophyll fluorescence: new instruments for special applications. In: Garab G (ed) Photosynthesis: mechanisms and effects, vol V. Springer, Berlin, pp 4253–4258
Schreiber U, Schliwa U, Bilger W (1986) Continuous recording of photochemical and nonphotochemical chlorophyll fluorescence quenching with a new type of modulation fluorometer. Photosynth Res 10:51–62
Seppala J, Olli K (2008) Multivariate analysis of phytoplankton spectral in vivo fluorescence: estimation of phytoplankton biomass during a mesocosm study in the baltic sea. Mar Ecol Prog Ser 370:69–85
Serra T, Borrego C, Quintana X, Calderer L, Lopez R, Colomer J (2009) Quantification of the effect of nonphotochemical quenching on the determination of in vivo Chl a from phytoplankton along the water column of a freshwater reservoir. Photochem Photobiol 85:321–331
Vuorio K, Lepistoe L, Holopainen A (2007) Intercalibrations of freshwater phytoplankton analyses. Boreal Environ Res 12:561–569
Walz GmbH H (2003) Phytoplankton analyzer PHYTO-PAM and Phyto-Win software V 1.45. In: Systems components and principles of operation. Effeltrich, Germany
Watson SB (1999) Outbreaks of taste/odour causing algal species: theoretical, mechanistic and applied approaches. University of Calgary, Calgary
Yentsch C, Phinney D (1985) Spectral fluorescence—an ataxonomic tool for studying the structure of phytoplankton populations. J Plankton Res 7:617–632
Yentsch C, Yentsch C (1979) Fluorescence spectral signatures—characterization of phytoplankton populations by the use of excitation and emission-spectra. J Mar Res 37:471–483
Acknowledgements
The work was supported by a Natural Sciences and Engineering Research Council of Canada Discovery Grant (R. Smith), with important additional support from Environment and Climate Change Canada (S. Watson). We thank two anonymous reviewers for their feedback that improved this manuscript.
Funding
Funding for this research was from a Natural Sciences and Engineering Research Council of Canada Discovery Grant (R. Smith), with important additional support from Environment and Climate Change Canada (S. Watson).
Author information
Authors and Affiliations
Contributions
All authors contributed to the study conception and design. Material preparation, data collection and analysis were performed by L. Beecraft. The first draft of the manuscript was written by L. Beecraft, and all authors participated in critical revision of the manuscript. All authors read and approved the final manuscript for submission.
Corresponding author
Ethics declarations
Conflict of interest
We have no conflicts of interest to declare.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Handling editor: Télesphore Sime-Ngando.
Supplementary Information
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Beecraft, L., Watson, S.B. & Smith, R.E.H. Quantifying the uncertainties in multi-wavelength PAM fluorometry due to innate and irradiance-induced variability of fluorescence spectra. Aquat Ecol 55, 169–186 (2021). https://doi.org/10.1007/s10452-020-09821-6
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10452-020-09821-6