Abstract
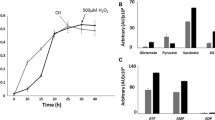
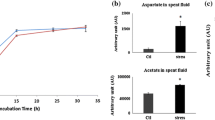
The interaction of keto-acids with reactive oxygen species (ROS) is known to produce the corresponding carboxylic acid with the concomitant formation of CO2. Formate is liberated when the keto-acid glyoxylate neutralizes ROS. Here we report on how formate is involved in combating oxidative stress in the nutritionally-versatile Pseudomonas fluorescens. When the microbe was subjected to hydrogen peroxide (H2O2), the levels of formate were 8 and two-fold higher in the spent fluid and the soluble cell-free extracts obtained in the stressed cultures compared to the controls respectively. Formate was subsequently utilized as a reducing force to generate NADPH and succinate. The former is mediated by formate dehydrogenase (FDH-NADP), whose activity was enhanced in the stressed cells. Fumarate reductase that catalyzes the conversion of fumarate into succinate was also markedly increased in the stressed cells. These enzymes were modulated by H2O2. While the stressed whole cells produced copious amounts of formate in the presence of glycine, the cell-free extracts synthesized ATP and succinate from formate. Although the exact role of formate in anti-oxidative defence has to await further investigation, the data in this report suggest that this carboxylic acid may be a potent reductive force against oxidative stress.




Similar content being viewed by others
Notes
(These values were compared to the formate levels in the respective control experiments where the cells were omitted respectively).
These are compared to the formate values in the reaction mixture in the control and stressed bands at time 0.
These are relative to succinate levels in the control and stress reaction bands at time 0 respectively.
These values are relative to the respective metabolite levels in the reaction mixtures at time 0.
References
Alekseeva AA, Savin SS, Tishkov VI (2011) NAD-dependent Formate dehydrogenase from plants. Acta Nat 3(4):38–54
Alhasawi A, Auger C, Appanna VP, Chahma MH, Appanna VD (2014) Zinc toxicity and ATP production in Pseudomonas fluorescens. J Appl Microbiol 117:65–73
Alhasawi A, Leblanc M, Appanna ND, Auger C, Appanna VD (2015a) Aspartate metabolism and pyruvate homeostasis triggered by oxidative stress in Pseudomonas fluorescens: a functional metabolomics study. Metabolomics. doi: 10.10007s11306-015-0841-4
Alhasawi A, Castonguay Z, Appanna ND, Auger C, Appanna VD (2015b) Glycine metabolism and anti-oxidative defence mechanisms in Pseudomonas fluorescens. Microbiol Res 171:26–31
Andreadeli A, Felmatakis E, Axarli I, Dimou M, Udvardi MK, Katinakis P, Labrou NE (2009) Cloning and characterization of Lotus japonicus formate dehydrogenase: a possible correlation with hypoxia. Biochim Biophys Acta 1794:976–984
Appanna VD, Preston CM (1987) Manganese elicits the synthesis of a novel exopolysaccharide in an arctic Rhizobium. FEBS Lett 215(1):79–82
Appanna VP, Auger C, Thomas SC, Omri A (2014) Fumarate metabolism and ATP production in Pseudomonas fluorescens exposed to nitrosative stress. Antonie Van Leeuwenhoek 106(3):431–438. doi:10.1007/s10482-014-0211-7
Auger C, Appanna ND, Alhasawi A, Appanna VD (2015) Deciphering metabolic networks by blue native polyacrylamide gel electrophoresis: a functional proteomic exploration. EuPa Open Proteom 7:64–72. doi:10.1016/j.euprot.2015.05.003
Bagramyan K, Trchounian A (2003) Structural and functional features of formate hydrogen lyase, an enzyme of mixed-acid fermentation from Escherichia coli. Biochemistry 68(11):1159–1170
Bagramyan K, Galstyan A, Trchounian A (2000) Redox potential is a determinant in the Escherichia coli anaerobic fermentative growth and survival effects of impermeable oxidant. Bioelectrochemistry 51(2):151–156
Beriault R, Chenier D, Singh R, Middaugh J, Mailloux R, Appanna VD (2005) Detection and purification of glucose 6-phosphate dehydrogenase, malic enzyme, and NADP-dependent isocitrate dehydrogenase by blue native polyacrylamide gel electrophoresis. Electrophoresis 26(15):2892–2897
Beriault R, Hamel R, Chenier D, Mailloux RJ, Joly H, Appanna VD (2007) The overexpression of NADPH-producing enzymes counters the oxidative stress evoked by gallium, an iron mimetic. Biometals 20(2):165–176
Bignucolo A, Appanna VP, Thomas SC, Auger C, Han S, Omri A et al (2013) Hydrogen peroxide stress provokes a metabolic reprogramming in Pseudomonas fluorescens enhanced production of pyruvate. J Biotechnol 167:309–315
Bradford MM (1976) A rapid and sensitive method for the quantitation of microgram quantities of protein utilizing the principle of protein-dye binding. Anal Biochem 72:248–254
Brioukhanov AL, Netrousov AI, Eggen RIL (2006) The catalase and superoxide dismutase genes are transcriptionally up-regulated upon oxidative stress in the strictly anaerobic archaeon Methanosarcina barkeri. Microbiology 152:1671–1677. doi:10.1099/mic.0.28542-0
Chenier D, Beriault R, Mailloux R, Baguie M, Abramia G, Lemire J (2008) Involvement of fumarase C and NADH oxidase in metabolic adaptation of Pseudomonas fluorescens cells evoked by aluminum and gallium toxicity. Appl Environ Microbiol 74(13):3977–3984
Gul-Karaguler N, Sessions RB, Clarke AR, Holbrook JJ (2001) A single mutation in the NAD-specific formate dehydrogenase from Candida methylica allows the enzyme to use NADP. Biotechnol Lett 23:283–287
Hamel R, Appanna VD, Viswanatha T, Puiseux-Dao S (2004) Overexpression of isocitrate lyase is an important strategy in the survival of Pseudomonas fluorescens exposed to aluminum. Biochem Biophys Res Commun. 217:1189–1194
Hoelsch K, Suhrer I, Heusel M, Weister-Botz D (2013) Engineering of formate dehydrogenase: synergistic effect of mutations affecting cofactor specificity and chemical stability. Appl Microbiol Biotechnol 97(6):2473–2481. doi:10.1007/s00253-012-4142-9
Hourton-Cabassa C, Ambard-Bretteville F, Moreau F, Davy de Virville J, Remy R, Colas des Franc-Small C (1998) Stress induction of mitochondrial formate dehydrogenase in potato leaves. Plant Physiol 116(2):627–635
Jormakka M, Tornroth S, Byrne B, Iwata S (2002) Molecular basis of proton motive force generation: structure of formate dehydrogenase-N. Science 295(5561):1863–1868
Jormakka M, Byrne B, Iwata S (2003) Formate dehydrogenase—a versatile enzyme in changing environments. Curr Opin Struct Biol 13(4):418–423
Lemire J, Appanna VD (2011) Aluminum toxicity and astrocyte dysfunction: a metabolic link to neurological disorders. J Inorg Biochem 105(11):1513–1517
Leonharsberger S, Korsa I, Bock A (2002) The molecular biology of formate metabolism in Enterobacteria. J Mol Micorbiol Biotechnol 4(3):269–276
Li S, Liu H, Zhang Y, Yan Y, Li Y (2009) The protective effects of α-ketoacids against oxidative stress on rat spermatozoa in vitro. Asian J Androl 12(2):247–256. doi:10.1038/aja.2009.78
Mailloux RJ, Beriault R, Lemire J, Singh R, Che´nier DR et al (2007) The tricarboxylic acid cycle, an ancient metabolic network with a novel twist. PLoS One 2(8):e690. doi:10.1371/journal.pone.0000690
Mailloux RJ, Lemire J, Kalyuzhnyi S, Appanna VD (2008) A novel metabolic network leads to enhanced citrate biogenesis in Pseudomonas fluorescens exposed to aluminum toxicity. Extremophiles 12:451–459
Mailloux RJ, Puiseux-Dao S, Appanaa VD (2009) α-ketoglutarate abrogates the nuclear localization of HIF-1α in aluminum-exposed hepatocytes. Biochimie 91:408–415
Mailloux RJ, Lemire J, Appanna VD (2011) Metabolic networks to combat oxidative stress in Pseudomonas fluorescens. Anton Leeuwen J Microbiol 99(3):433–442
Schagger H, von Jagow G (1991) Blue native electrophoresis for isolation of membrane protein complexes in enzymatically active form. Anal Biochem 199(2):223–231
Shinagawa E, Toyama H, Matsushita K, Tuitemwong P, Theeragool G, Adachi O (2008) Formaldehyde elimination with formaldehyde and formate oxidase in membrane of acetic acid bacteria. J Biosci Bioeng 105(3):292–295
Singh R, Lemire J, Mailloux RJ, Appanna VD (2008) A novel strategy involved anti-oxidative defense: the conversion of NADH into NADPH by a metabolic network. PLoS One 3:2682
Stubbe J, Nocera DG, Yee CS, Chang MCY (2003) Radical initiation in the class I ribonucleotide reductase: long-range proton-coupled electron transfer? Chem Rev 103(6):2167–2202
Su C, Puls RW (2004) Nitrate reduction by zero valent iron: effects of formate, oxalate, citrate, chloride, sulfate, borate, and phosphate. Environ Sci Technol 38(9):2715–2720
Suzuki K, Itai R, Suzuki K, Nakanishi H, Nishizawa N, Yoshimura E, Mori S (1998) Formate dehydrogenase, an enzyme of anaerobic metabolism, is induced by iron deficiency in barley roots. Plant Physiol 116(2):725–732
Thomas SC, Alhasawi A, Appanna VP, Auger C, Appanna VD (2015) Brain metabolism and Alzheimer’s disease the prospect for a metabolite-based therapy. J Nutr Health Aging 19(1):58–63. doi:10.1007/s12603-014-0511-7
Uchimura H, Enjoji H, Seki T, Taguchi A, Tsakaya N, Shoun H (2002) Nitrate reductase-formate dehydrogenase couple involved in the fungal denitrification by Fusarium oxysporum. J Biochem 131(4):579–586
Yammamoto I, Saiki T, Liu SM, Ljungdahl LG (1983) Purification and properties of NADP-dependent formate dehydrogenase from Clostridium thermoaceticum a tungsten-selenium-iron protein. J Biol Chem 258(3):1826–1832
Ying W (2008) NAD+/NADH and NADP+/NADPH in cellular functions and cell death: regulation and biological consequences. Antioxid Redox Signal 10:179–206
Yokota A, Kawabata A, Kitaoka S (1983) Mechanism of glyoxylate decarboxylation in the glycolate pathway in Euglena gracilis Z participation of Mn2+-dependent NADPH oxidase in chloroplasts. Plant Physiol 71:772–776
Yokota A, Koura H, Kitaoka S (1985) Refixation of photorespired CO2 during photosynthesis in Euglena gracilis Z. Agric Biol Chem 49:3309–3310
Yoshida A, Nishimura T, Kawaguchi H, Inui M, Yukawa H (2005) Enhanced hydrogen production from formic acid by formate hydrogen lyase-overexpressing Escherichia coli strains. Appl Environ Microbiol 71(11):6762–6768. doi:10.1128/AEM.71.11.6762-6768.2005
Zaunmuller T, Kelly DJ, Glockner FO, Unden G (2006) Succinate dehydrogenase functioning by a reverse redox loop mechanism and fumarate reductase in sulphate-reducing bacteria. Microbiology 152:2443–2453
Acknowledgments
This work was supported by Laurentian University and Northern Ontario Heritage Fund Corporation. Sean C. Thomas is a recipient of the Natural Sciences and Engineering Research Council Post Graduate Scholarship-Masters. Azhar Alhasawi is a recipient of a doctoral scholarship from the Ministry of Higher Education of Saudi Arabia.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of Interest
The authors declare that they have no conflict of interest.
Rights and permissions
About this article
Cite this article
Thomas, S.C., Alhasawi, A., Auger, C. et al. The role of formate in combatting oxidative stress. Antonie van Leeuwenhoek 109, 263–271 (2016). https://doi.org/10.1007/s10482-015-0629-6
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10482-015-0629-6