Abstract
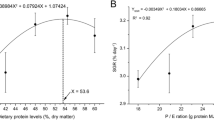
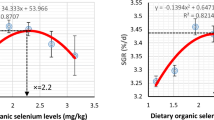
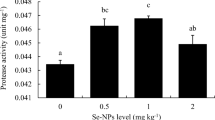
A 12-week feeding trial was conducted to determine the optimal dietary selenium requirement for coho salmon (Oncorhynchus kisutch W.) alevins. Six experimental diets (contained about 42.2% crude protein and 10.5% crude lipid) were formulated to contain selenium level of 0.07, 0.15, 0.24, 0.41, 0.75, and 1.38 mg/kg, respectively. Each diet was fed to triplicate groups of fish with initial mean body weight of 0.38 ± 0.01 g, which were randomly assigned to 18 plastic tanks (80 × 60 × 60 cm, length × width × height, water volume 240 L), and each tank was stocked initially with 50 fish. Fish were fed four times daily and used the method of overfeeding (15–20% body weight) to fish. The sampling procedure was implemented after 24 h of fasting. Results showed that specific growth rate (SGR) in 0.24, 0.41, and 0.75 mg Se/kg groups were significantly higher than those in 0.07 and 1.38 mg Se/kg groups (P < .05). There were no significant differences in hepatosomatic index (HIS), intestinal somatic index (ISI), condition factor (CF), and whole-body composition among dietary groups (P > .05). Hepatic malondialdehyde (MDA) content significantly declined with increasing dietary up to 0.41 mg Se/kg and increased as selenium further increased. The hepatic superoxide dismutase (SOD), catalase (CAT), and glutathione peroxidase (GPx) significantly increased with increasing dietary selenium up to 0.41 mg/kg and decreased significantly as selenium further increased (P < .05). Fish fed diet with 0.75 mg Se/kg had the highest glutathione reductase (GR), which did not differ significantly with 0.41 and 1.38 mg Se/kg groups (P > .05) but significantly higher than those in 0.07, 0.15, and 0.24 mg Se/kg groups (P < .05). Broken-line regression analyses based on SGR, hepatic, and whole-body selenium contents indicated that the optimum dietary selenium requirements were in the range of 0.39–0.43 mg/kg for coho salmon alevins.




Similar content being viewed by others
Data availability
All the data in the article are available from the corresponding author upon reasonable request.
References
Antony JP, Schrama JW, Kaushik SJ (2016) Mineral requirements of fish: a systematic review. Rev Aquac 8:172–219. https://doi.org/10.1111/raq.12090
AOAC (1995) Official Methods of Analysis of the Association of Analytical Chemistry, 16th edn. Association of Official Analytical Chemists, Washington
Ayhanci A, Günes S, Sahinturk V, Appak S, Uyar R, Cengiz M, Altuner Y, Yaman S (2010) Seleno L-methionine acts on cyclophosphamide-induced kidney toxicity. Biol Trace Elem Res 136(2):171–179. https://doi.org/10.1007/s12011-009-8535-2
Bell JG, Cowey CB, Adron JW, Shanks AM (1985) Some effects of vitamin E and selenium deprivation on tissue enzyme levels and indices of tissue peroxidation in rainbow trout (Salmo gairdneri). Br J Nutr 53:149–157. https://doi.org/10.1079/bjn19850019
Burk RF, Hill KE (1993) Regulation of selenoproteins. Annu Rev Nutr 13:65–81. https://doi.org/10.1146/annurev.nutr.13.1.65
Clark RF, Strukle E, Williams SR, Manoguerra AS (1996) Selenium poisoning from a nutritional supplement. J Am Med Assoc 275:1087–1088. https://doi.org/10.1001/jama.275.14.1087
Carlberg I, Mannervik B (1975) Purification and characterization of the flavoenzyme glutathione reductase from rat liver. J Biol Chem 250:5475–5480. https://doi.org/10.0000/PMID237922
Chen SJ, Liu P, Jia YM, Liao HP, Zhu SX, Zhou LL, Dan XM, Liu L, Li JH, Zheng SB, Yang JJ, Gan L (2020) Dietary lipid concentrations influence growth, body composition, morphology of the liver and mid-intestine, and antioxidant status of marbled eel (Anguilla marmorata). Aquac Int 28:2287–2302. https://doi.org/10.1007/s10499-020-00590-5
Dawood MAO, Zommara M, Eweedah NM, Helal AI (2019) Synergistic effects of selenium nanoparticles and vitamin E on growth, immune-related gene expression, and regulation of antioxidant status of Nile tilapia (Oreochromis niloticus). Biol Trace Elem Res 195:624–635. https://doi.org/10.1007/s12011-019-01857-6
De Riu N, Lee JW, SSY H, Moniello G, Hung SSO (2014) Effect of dietary selenomethionine on growth performance, tissue burden, and histopathology in green and white sturgeon. Aquat Toxicol 148:65–73. https://doi.org/10.1016/j.aquatox.2013.12.030
Diplock AT (1994) Antioxidants and disease prevention. Mol Asp Med 15:293–376. https://doi.org/10.1016/0098-2997(94)90005-1
FAO (Food and Agriculture Organization of the United Nations) (2020) The State of World Fisheries and Aquaculture 2020. In: Sustainability in action. FAO, Rome. https://doi.org/10.4060/ca9229en
Fattman CL, Schaefer LM, Oury TD (2003) Extracellular superoxide dismutase in biology. Free Radic Biol Med 35(3):236–256. https://doi.org/10.1016/s0891-5849(03)00275-2
Gaber MM (2007) Efficiency of selenium ion inclusion into common carp (Cyprinus carpio L.) diets. J Fish Int 2(3):250–254
Gao XJ, Tang B, Liang HH, Yi L, Wei ZG (2019a) Selenium deficiency induced an inflammatory response by the HSP60-TLR2-MAPKs signalling pathway in the liver of carp. Fish Shellfish Immunol 87:688–694. https://doi.org/10.1016/j.fsi.2019.02.017
Gao XJ, Tang B, Liang HH, Yi L, Wei ZG (2019b) Selenium deficiency inhibits micRNA-146a to promote ROS-induced inflammation via regulation of the MAPK pathway in the head kidney of carp. Fish Shellfish Immunol 91:284–292. https://doi.org/10.1016/j.fsi.2019.05.039
Garcia d l SD, Devlin RH, Johnston IA (2015) RNAseq analysis of fast skeletal muscle in restriction-fed transgenic coho salmon (Oncorhynchus kisutch): an experimental model uncoupling the growth hormone and nutritional signals regulating growth. BMC Genomics 16(1):564. https://doi.org/10.1186/s12864-015-1782-z
Gasmi A, Garnier R, Galliot-Guilley M, Gaudillat C, Quartenoud B, Buisine A, Djebbar D (1997) Acute selenium poisoning. Vet Hum Toxicol 39:304–308. https://doi.org/10.1016/S0093-691X(97)00309-9
Gatlin DM, Wilson RP (1984) Dietary selenium requirement of fingerling channel catfish. J Nutr 114(3):627–633. https://doi.org/10.1093/jn/114.3.627
Gobi N, Vaseeharan B, Rekha R, Vijayakumar S, Faggio C (2018) Bioaccumulation, cytotoxicity and oxidative stress of the acute exposure selenium in Oreochromis mossambicus. Ecotoxicol Environ Saf 162:147–159. https://doi.org/10.1016/j.ecoenv.2018.06.070
Hamilton SJ, Buhl KJ, Faerber NL (1990) Toxicity of organic selenium in the diet to Chinook salmon. Environ Toxicol Chem 9:347–358. https://doi.org/10.1002/etc.5620090310
Han D, Xie S, Liu M, Xiao X, Liu H, Zhu X, Yang Y (2011) The effects of dietary selenium on growth performances, oxidative stress and tissue selenium concentration of gibel carp (Carassius auratus gibelio). Aquac Nutr 17(3):e741–e749. https://doi.org/10.1111/j.1365-2095.2010.00841.x
Hao J, Lin Y, Pan W, Jiang W, Ge X (2020) Dietary selenium enhances the growth and anti-oxidant capacity of juvenile blunt snout bream (Megalobrama amblycephala). Fish Shellfish Immunol 101:115–125. https://doi.org/10.1016/j.fsi.2020.03.041
Hicks BD, Hilton JW, Ferguson HW (1984) Influence of dietary selenium on the occurrence of nephrocalcinosis in the rainbow trout, Salmo gairdneri Richardson. J Fish Dis 7:379–389. https://doi.org/10.1111/j.1365-2761.1984.tb01202.x
Hilton JW, Hodson PV, Slinger SJ (1980) The requirement and toxicity of selenium in rainbow trout (Salmo gairdneri). J Nutr 110(12):2527–2535. https://doi.org/10.1093/jn/110.12.2527
Jamil Z (2013) Effects of inorganic and nanoform of selenium on growth performance and biochemical indices of mahseer (Tor putitora). M.Phil. Thesis, Quaid-i-Azam University, Islamabad
Jovanovic A, Grubor-Lajsic G, Djukic N, Gardinovacki G, Matic A, Spasic M, Pritsos CA (1997) The effect of selenium on antioxidant system in erythrocytes and liver of the carp (Cyprinus carpio L.). Crit Rev Food Sci Nutr 37(5):443–448. https://doi.org/10.1080/10408399709527783
Julshamn K, Sandnes K, Lie O, Waagbo R (1990) Effects of dietary selenium supplementation on growth, blood chemistry and trace element levels in serum and liver of adult Atlantic salmon (Salmo salar). Fisk Dir Skr Ernaering III 2:47–58
Juniper DT, Phipps RH, Ramos-Morales E, Bertin G (2008) Effect of dietary supplementation with selenium-enriched yeast or sodium selenite on selenium tissue distribution and meat quality in beef cattle. J Anim Sci 86:3100–3109. https://doi.org/10.2527/jas.2006-067
Khalil HS, Mansour AT, Goda AMA, Omar EA (2019) Effect of selenium yeast supplementation on growth performance, feed utilization, lipid profile, liver and intestine histological changes, and economic benefit in meagre, Argyrosomus regius, fingerlings. Aquaculture 501(25):135–143. https://doi.org/10.1016/j.aquaculture.2018.11.018
Khan KU, Zuberi A, Nazir S, Fernandes JBK, Jamil Z, Sarwar H (2016) Effects of dietary selenium nanoparticles on physiological and biochemical aspects of juvenile Tor putitora. Turk J Zool 40:704–712. https://doi.org/10.3906/zoo-1510-5
Khan KU, Zuberi A, Fernandes JBK, Ullah I, Sarwar H (2017) An overview of the ongoing insights in selenium research and its role in fish nutrition and fish health. Fish Physiol Biochem 43:1689–1705. https://doi.org/10.1007/s10695-017-0402-z
Klein RG, Halver JE (1970) Nutrition of salmonoid fishes: arginine and histidine requirements of chinook and coho salmon. the. J Nutr 100(9):1105–1109. https://doi.org/10.1093/jn/100.9.1105
Koller LD, Exon JH (1986) The two faces of selenium deficiency and toxicity--are similar in animals and man. Can J Vet Res 50(3):297–306
Kong YQ, Ding ZL, Zhang YX, Ye JY, Du ZY (2017) Dietary selenium requirement of juvenile oriental river prawn Macrobrachium nipponense. Aquaculture 476:72–78. https://doi.org/10.1016/j.aquaculture.2017.04.010
Kosik-Bogacka DI, Baranowska-Bosiacka I, Noceń I, Jakubowska K, Chlubek D (2011) Hymenolepis diminuta: activity of anti-oxidant enzymes in different parts of rat gastrointestinal tract. Exp Parasitol 128(3):265–271. https://doi.org/10.1016/j.exppara.2011.02.026
Lee S, Nambi RW, Won S, Katya K, Bai SC (2016) Dietary selenium requirement and toxicity levels in juvenile Nile tilapia, Oreochromis niloticus. Aquaculture 464:153–158. https://doi.org/10.1016/j.aquaculture.2016.06.027
Le KT, Fotedar R (2014) Bioavailability of selenium from different dietary sources in yellowtail kingfish (Seriola lalandi). Aquaculture 420-421:57–62. https://doi.org/10.1016/j.aquaculture.2013.10.034
Li HC, Zhang JS, Wang T, Luo WR, Zhou QF, Jiang GB (2008) Elemental selenium particles at nano-size (Nano-Se) are more toxic to Medaka (Oryzias latipes) as a consequence of hyper-accumulation of selenium: a comparison with sodium selenite. Aquat Toxicol 89:251–256. https://doi.org/10.1016/j.aquatox.2008.07.008
Lin YH, Shiau SY (2005) Dietary selenium requirements of juvenile grouper, Epinephelus malabaricus. Aquaculture 250(1-2):356–363. https://doi.org/10.1016/j.aquaculture.2005.03.022
Little C, Olinescu R, Reid KG, O’Brien PJ (1970) Properties and regulation of glutathione peroxidase. J Biol Chem 245(14):3632–3636. https://doi.org/10.1016/S0021-9258(18)62973-4
Liu K, Wang XJ, Ai QH, Mai KS, Zhang WB (2010) Dietary selenium requirement for juvenile cobia, Rachycentron canadum L. Aquac Res 41(10):e594–e601. https://doi.org/10.1111/j.1365-2109.2010.02562.x
Lorentzen M, Maage A, Julshamn K (1994) Effects of dietary selenite or selenomethionine on tissue selenium levels of Atlantic salmon (Salmo salar). Aquaculture 121:359–367. https://doi.org/10.1016/0044-8486(94)90270-4
Maier KJ, Knight AW (1994) Ecotoxicology of selenium in freshwater systems. Rev Environ Contam Toxicol 134:31–48. https://doi.org/10.1007/978-1-4684-7068-0_2
Mateo RD, Spallholz JE, Elder R, Yoon I, Kim SW (2007) Efficacy of dietary selenium sources on growth and carcass characteristics of growing-finishing pig fed diets containing high endogenous selenium. J Anim Sci 85:1177–1183. https://doi.org/10.2527/jas.2006-067
Nordberg GF, Fowler BA, Nordberg M, Friberg LT (2007) Handbook on the toxicology of metals. Reprod Develop Toxic Metals 1:213–249. https://doi.org/10.1016/B978-0-12-369413-3.X5052-6
NRC (National Research Council) (2011) Nutrient requirements of fish and shrimp. The National Academy Press, Washington, DC
Orun I, Talas ZS, Ozdemir I, Alkan A, Erdogan K (2008) Antioxidative role of selenium on some tissues of (Cd2+, Cr3+)-induced rainbow trout. Ecotoxicol Environ Saf 71:71–75. https://doi.org/10.1016/j.ecoenv.2007.07.008
Poston HA, Combs GF, Leibovitz L (1976) Vitamin E and selenium interrelations in the diet of Atlantic salmon (Salmo salar): gross, histological and biochemical deficiency signs. J Nutr 106:892–904. https://doi.org/10.1093/jn/106.7.892
Prohaska JR, Ganther HE (1977) Glutathione peroxidase activity of glutathione-S-transferees purified from rat liver. Biochem Biophys Res Commun 76(2):437–445. https://doi.org/10.1016/0006-291X(77)90744-6
Raza A (2012) Effects of graded levels of dietary selenium supplementation on the growth of juvenile mahseer (Tor putitora). M.Phil. Thesis, Quaid-i-Azam University, Islamabad
Robbins KR, Norton HW, Baker DH (1979) Estimation of nutrient requirements from growth data. J Nutr 109(10):1710–1714. https://doi.org/10.1093/jn/109.10.1710
Rodríguez PH, Rojas MS (2014) Efecto de dietas enriquecidas con vitamina e y selenio orgánico en el comportamiento productivo y calidad funcional del filete de trucha arco iris (Oncorhynchus mykiss). Rev Inv Vet Perú 25(2):213–225
Schwarz K, Foltz CM (1999) Selenium as an integral part of factor 3 against dietary necrotic liver degeneration. Nutrition 36(11):255–3293. https://doi.org/10.1021/ja01569a087
Schirmer RH, Krauth-Siegel RL, Schulz GE (1989) Glutathione reductase. Wiley, New York
Sedlak J, Linday RH (1968) Estimation of total, protein-bound, and nonprotein sulfhydryl groups in tissue with Ellman’s reagent. Anal Biochem 25:192–205. https://doi.org/10.1016/0003-2697(68)90092-4
Tashjian DH, Teh SJ, Sogomonyan A, Hung SSO (2006) Bioaccumulation and chronic toxicity of dietary l-selenomethionine in juvenile white sturgeon (Acipenser transmontanus). Aquat Toxicol 79(4):401–409. https://doi.org/10.1016/j.aquatox.2006.07.008
Vignola G, Lambertini L, Mazzone G, Giammarco M, Tassinari M, Martelli G, Bertin G (2008) Effects of selenium source and level of supplementation on the performance and meat quality of lambs. Meat Sci 81:678–685. https://doi.org/10.1016/j.meatsci.2008.11.009
Wang C, Lovell RT (1997) Organic selenium sources, selenomethionine and selenoyeast, have higher bioavailability than an inorganic selenium source, sodium selenite, in diets for channel catfish (Ictalurus punctatus). Aquaculture 152(1-4):223–234. https://doi.org/10.1016/S0044-8486(96)01523-2
Wang L, Xiao JX, Hua Y, Xiang XW, Zhou YF, Ye L, Shao QJ (2019) Effects of dietary selenium polysaccharide on growth performance, oxidative stress and tissue selenium accumulation of juvenile black sea bream, Acanthopagrus schlegelii. Aquaculture 503(30):389–395. https://doi.org/10.1016/j.aquaculture.2019.01.033
Yao Z, Liu Y, Yang H, Liang G, Tian L (2012) Effect of dietary selenium level on growth performance, body composition and hepatic glutathione peroxidase activities of largemouth bass Micropterus salmoide. Aquac Res 43(11):1660–1668. https://doi.org/10.1111/j.1365-2109.2011.02972.x
Zhou F, Xiong W, Xiao JX, Shao QJ, Bergo ON, Hua Y, Chai X (2010) Optimum arginine requirement of juvenile black sea bream, Sparus macrocephalus. Aquac Res 41:e418–e430. https://doi.org/10.1111/j.1365-2109.2009.02474.x
Zhou X, Wang Y, Gu Q, Li W (2009) Effects of different dietary selenium sources (selenium nanoparticle and selenomethionine) on growth performance, muscle composition and glutathione peroxidase enzyme activity of crucian carp (Carassius auratus gibelio). Aquaculture 291(1):78–81. https://doi.org/10.1016/j.aquaculture.2009.03.007
Zhu Y, Chen Y, Liu Y, Yang H, Liang G, Tian L (2011) Effect of dietary selenium level on growth performance, body composition and hepatic glutathione peroxidase activities of largemouth bass (Micropterus salmoide). Aquac Res 43:1660–1668. https://doi.org/10.1111/j.1365-2109.2011.02972.x
Funding
This study was funded by the Shandong Provincial Key Research and Development Programs (Major Scientific and Technological Innovation Projects, MSTIP), Grant/Award Numbers, 2018CXGC0102 and 2019JZZY020710; Scientific and Technologic Development Program of Weifang, Grant/Award Number, 2019ZJ1046; and Innovation-Driven Development Special Project of Guangxi Science and Technology Major Project, Grant/Award Number, AA17204044
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Ethics approval
All procedures implemented for fish followed the handling guidelines of Weifang University.
Conflict of interest
The authors declare no competing interests.
Additional information
Handling Editor: Gavin Burnell
Publisher’s note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Du, Lc., Yu, Hr., Li, Ly. et al. Dietary selenium requirement of coho salmon (Oncorhynchus kisutch W.) alevins. Aquacult Int 29, 2291–2304 (2021). https://doi.org/10.1007/s10499-021-00749-8
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10499-021-00749-8