Abstract
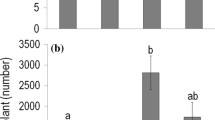
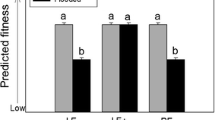
Exotic invasive plant species (EIPS) may succeed in part because they release allelochemicals that are novel to native plants (Novel Weapons Hypothesis). Plant allelopathic effects may increase under abiotic and biotic stresses based on The Stress Hypothesis of Allelopathy (TSHA). Lonicera maackii is an aggressive EIPS in the Midwestern USA, able to affect native flora by means of allelopathy. We aimed to test the role of intraspecific competition, as biotic stress, and expected future climate conditions (i.e. intense floods in spring; hotter and drier conditions in summer), as abiotic stress, on the performance and allelopathic potential of L. maackii. We grew L. maackii plants, collected from the field (field plants) or germinated from seeds in a greenhouse (seedlings), with or without intraspecific competition and under simulated present and expected future (1) spring (intense flooding vs no flooding) and (2) summer conditions. We subsequently measured plant performance variables, such as Fv/Fm (an indicator of plant stress), plant biomass, and concentration of pigments, carbon and nitrogen; and allelopathic potential (i.e. the effect of L. maackii leaf extracts and conditioned substrates on the germination of two herb species). In accordance with TSHA, intense flooding stressed L. maackii (Fv/Fm < 0.7), reduced its biomass, and increased its allelopathic potential. In summer, L. maackii plants were more stressed (lower average Fv/Fm) under future summer conditions and intraspecific competition than under present conditions without competitors, but they had similar allelopathic potential. This suggests the presence of a stress threshold from which allelopathic potential does not increase further. Intraspecific competition more negatively affected the performance of seedlings (e.g. plant mass reduction) than field plants, but only increased the allelopathic potential of the latter. Our results indicated that the negative effects of intense flooding on L. maackii performance could be counteracted by the benefit gained in plant–plant competition by means of increased allelopathic potential. The complex role of abiotic and biotic conditions on L. maackii performance and allelopathy are also discussed.







Similar content being viewed by others
References
Allen SE, Grimshaw HM, Parkinson JA, Quarnby C (1974) Chemical analysis of ecological materials. Blackwell, Oxford
Anderson LJ, Cipollini D (2013) Gas exchange, growth, and defense responses of invasive Alliaria petiolata (Brassicaceae) and native Geum vernum (Rosaceae) to elevated atmospheric CO2 and warm spring temperatures. Am J Bot 100:1544–1554. https://doi.org/10.3732/ajb.1300014
Barnes JD, Balaguer L, Manrique E et al (1992) A reappraisal of the use of DMSO for the extraction and determination of chlorophylls a and b in lichens and higher plants. Environ Exp Bot 32:85–100
Batra NG, Sharma V, Kumari N (2014) Drought-induced changes in chlorophyll fluorescence, photosynthetic pigments, and thylakoid membrane proteins of Vigna radiata. J Plant Interact 9:712–721. https://doi.org/10.1080/17429145.2014.905801
Bauer JT, Shannon SM, Stoops RE, Reynolds HL (2012) Context dependency of the allelopathic effects of Lonicera maackii on seed germination. Plant Ecol 213:1907–1916. https://doi.org/10.1007/s11258-012-0036-2
Bonilla D, Rodà F (1989) Dinámica del nitrógeno en el suelo de un encinar montano: respuesta a una perturbación experimental. Options Méditerranéennes Serie A 3:187–190
Boyce RL (2015) Recovery of native plant communities in southwest Ohio after Lonicera maackii removal. J Torrey Bot Soc 142:193–204. https://doi.org/10.3159/TORREY-D-14-00055
Bradley BA, Wilcove DS, Oppenheimer M (2010) Climate change increases risk of plant invasion in the Eastern United States. Biol Invas 12:1855–1872. https://doi.org/10.1007/s10530-009-9597-y
Callaway RM, Ridenour WM (2004) Novel weapons: invasive success and the evolution of increased competitive ability. Front Ecol Environ 2:436. https://doi.org/10.2307/3868432
Callaway RM, Walker LR (1997) Competition and facilitation: a synthetic approach to interactions in plant communities. Ecology 78:1958. https://doi.org/10.1002/j.1537-2197.1982.tb13318.x
Cappuccino N, Arnason JT (2006) Novel chemistry of invasive exotic plants. Biol Lett 2:189–193. https://doi.org/10.1098/rsbl.2005.0433
Chapin FS (1991) Integrated responses of plants to stress. Bioscience 41:29–36. https://doi.org/10.2307/1311538
Cipollini D, Dorning M (2008) Direct and indirect effects of conditioned soils and tissue extracts of the invasive shrub, Lonicera maackii, on target plant performance. Castanea 73:166–176. https://doi.org/10.2179/0008-7475-73.3.166
Cipollini D, Stevenson R, Enright S et al (2008a) Phenolic metabolites in leaves of the invasive shrub, Lonicera maackii, and their potential phytotoxic and anti-herbivore effects. J Chem Ecol 34:144–152. https://doi.org/10.1007/s10886-008-9426-2
Cipollini KA, McClain GY, Cipollini D (2008b) Separating above- and belowground effects of Alliaria petiolata and Lonicera maackii on the performance of Impatiens capensis. Am Midl Nat 160:117–128. https://doi.org/10.1674/0003-0031(2008)160%5b117:saabeo%5d2.0.co;2
Cipollini D, Rigsby CM, Barto EK (2012) Microbes as targets and mediators of allelopathy in plants. J Chem Ecol 38:714–727. https://doi.org/10.1007/s10886-012-0133-7
Das K, Roychoudhury A (2014) Reactive oxygen species (ROS) and response of antioxidants as ROS-scavengers during environmental stress in plants. Front Environ Sci 2:1–13. https://doi.org/10.3389/fenvs.2014.00053
Demmig-Adams B, Adams WW (1996) The role of xanthophyll cycle carotenoids in the protection of photosynthesis. Trends Plant Sci 1:21–26. https://doi.org/10.1016/S1360-1385(96)80019-7
Díaz-Mendoza M, Velasco-Arroyo B, Santamaria ME et al (2016) Plant senescence and proteolysis: two processes with one destiny. Genet Mol Biol 39:329–338. https://doi.org/10.1590/1678-4685-GMB-2016-0015
Didham RK, Tylianakis JM, Hutchison MA et al (2005) Are invasive species the drivers of ecological change? Trends Ecol Evol 20:470–474. https://doi.org/10.1016/j.tree.2005.07.006
Dorning M, Cipollini D (2006) Leaf and root extracts of the invasive shrub, Lonicera maackii, inhibit seed germination of three herbs with no autotoxic effects. Plant Ecol 184:287–296. https://doi.org/10.1007/s11258-005-9073-4
Dubois J-JB, Blazich FA (2008) Lonicera L.: honeysuckle. In: Bonner Franklin T, Karrfalt Robert P (eds) The woody plant seed manual. Agriculture handbook no. 727. U.S. Department of Agriculture, Forest Service, Washington, DC, p 1223
Dukes JS, Mooney HA (1999) Does global change increase the success of biological invaders? Trends Ecol Evol 14:135–139. https://doi.org/10.1016/S0169-5347(98)01554-7
Fernandez C, Monnier Y, Santonja M et al (2016) The impact of competition and allelopathy on the trade-off between plant defense and growth in two contrasting tree species. Front Plant Sci 7:1–14. https://doi.org/10.3389/fpls.2016.00594
Gilad E, Shachak M, Meron E (2007) Dynamics and spatial organization of plant communities in water-limited systems. Theor Popul Biol 72:214–230. https://doi.org/10.1016/j.tpb.2007.05.002
Global Invasive Species Database (2018) Species profile: Lonicera maackii. http://www.iucngisd.org/gisd/speciesname/Lonicera+maackii. Accessed 13 Apr 2018
Gorchov DL, Trisel DE (2003) Competitive effects of the invasive shrub, Lonicera maackii (Rupr.) Herder (Caprifoliaceae), on the growth and survival of native tree seedlings. Plant Ecol 166:13–24
Gould AMA, Gorchov DL (2000) Effects of the exotic invasive shrub Lonicera maackii on the survival and fecundity of three species of native annuals. Am Midl Nat 144:36–50. https://doi.org/10.1674/0003-0031(2000)144%5b0036:eoteis%5d2.0.co;2
Gray SB, Brady SM (2016) Plant developmental responses to climate change. Dev Biol 419:64–77. https://doi.org/10.1016/j.ydbio.2016.07.023
Hall AB, Blum U, Fites RC (1982) Stress modification of allelopathy of Helianthus annuus L. debris on seed germination. Am J Bot 69:776–783. https://doi.org/10.2307/2442968
Hanson AD, Ditz KM, Singletary GW, Leland TJ (1983) Gramine accumulation in leaves of barley grown under high-temperature stress. Plant Physiol 71:896–904. https://doi.org/10.1104/pp.71.4.896
Hatfield JL, Prueger JH (2015) Temperature extremes: effect on plant growth and development. Weather Clim Extrem 10:4–10. https://doi.org/10.1016/j.wace.2015.08.001
Hellmann JJ, Byers JE, Bierwagen BG, Dukes JS (2008) Five potential consequences of climate change for invasive species. Conserv Biol 22:534–543. https://doi.org/10.1111/j.1523-1739.2008.00951.x
Hidayati SN, Baskin JM, Baskin CC (2000) Dormancy-breaking and germination requirements of seeds of four Lonicera species (Caprifoliaceae) with underdeveloped spatulate embryos. Seed Sci Res 10:459–469. https://doi.org/10.1017/S0960258500000507
Hierro JL, Callaway RM (2003) Allelopathy and exotic plant invasion. Plant Soil 256:29–39. https://doi.org/10.1023/A:1026208327014
Hochman A, Harpaz T, Saaroni H, Alpert P (2018) The seasons’ length in 21st century CMIP5 projections over the eastern Mediterranean. Int J Climatol 38:2627–2637. https://doi.org/10.1002/joc.5448
IPCC (2013) Climate change 2013: the physical science basis. Contribution of working group I to the fifth assessment report of the intergovernmental panel on climate change, Cambridge University Press, New York, NY
Kobayashi K (2004) Factors affecting phytotoxic activity of allelochemicals in soil. Cheminform 35:1–7. https://doi.org/10.1002/chin.200435273
Körner C (2015) Paradigm shift in plant growth control. Curr Opin Plant Biol 25:107–114. https://doi.org/10.1016/j.pbi.2015.05.003
Kozlowski TT (1997) Responses of woody plants to flooding and salinity. Tree Physiol 17:490. https://doi.org/10.1093/treephys/17.7.490
Kunkel KE, Stevens LE, Stevens SE, Sun L, Janssen E, Wuebbles D, Hilberg SD, Timlin MS, Stoecker L, Westcott NE, Dobson JG (2013) Regional climate trends and scenarios for the US national climate assessment. Part 3. Climate of the Midwest US, NOAA Technical Report NESDIS 142-3, p 95
Lankau RA (2011) Intraspecific variation in allelochemistry determines an invasive species’ impact on soil microbial communities. Oecologia 165:453–463. https://doi.org/10.1007/s00442-010-1736-8
Lawrence JG, Colwell A, Sexton OJ (1991) The ecological impact of allelopathy in Ailanthus altissima (Simaroubaceae). Am J Bot 78:948–958. https://doi.org/10.2307/2445173
Li H, Syvertsen JP, Mccoy CW, Stuart RJ, Schumann AW (2006) Water stress and root injury from simulated. Soil Sci 171:138–151. https://doi.org/10.1097/01.ss.0000187367.31094.e5
Liu Q, Xu R, Yan Z et al (2013) Phytotoxic allelochemicals from roots and root exudates of Trifolium pratense. J Agric Food Chem 61:6321–6327. https://doi.org/10.1021/jf401241e
Luken JO, Thieret JW (1996) Amur Honeysuckle, its fall from grace. Bioscience 46:18–24. https://doi.org/10.2307/1312651
Maestre FT, Cortina J (2004) Do positive interactions increase with abiotic stress? A test from a semi-arid steppe. Proc R Soc B Biol Sci 271:S331–S333. https://doi.org/10.1098/rsbl.2004.0181
Maestre FT, Callaway RM, Valladares F, Lortie CJ (2009) Refining the stress-gradient hypothesis for competition and facilitation in plant communities. J Ecol 97:199–205
Malik AI, Colmer TD, Lambers H, Setter TL, Schortemeyer M (2002) Short-term waterlogging has long-term effects on the growth and physiology of wheat. New Phytol 153(2):225–236
Mangla S, Inderjit, Callaway RM (2008) Exotic invasive plant accumulates native soil pathogens which inhibit native plants. J Ecol 96:58–67. https://doi.org/10.1111/j.1365-2745.2007.01312.x
Mangla S, Sheley RL, James JJ, Radosevich SR (2011) Intra and interspecific competition among invasive and native species during early stages of plant growth. Plant Ecol 212:531–542. https://doi.org/10.1007/s11258-011-9909-z
Martínez-Vilalta J, Poyatos R, Aguadé D et al (2014) A new look at water transport regulation in plants. New Phytol 204:105–115. https://doi.org/10.1111/nph.12912
Matías L, Castro J, Villar-Salvador P et al (2017) Differential impact of hotter drought on seedling performance of five ecologically distinct pine species. Plant Ecol 218:201–212. https://doi.org/10.1007/s11258-016-0677-7
Maxwell K, Johnson GN (2000) Chlorophyll fluorescence—a practical guide. J Exp Bot 51:659–668. https://doi.org/10.1093/jxb/51.345.659
Medina CL, Sanches MC, Tucci MLS et al (2009) Erythrina speciosa (Leguminosae-Papilionoideae) under soil water saturation: morphophysiological and growth responses. Ann Bot 104:671–680. https://doi.org/10.1093/aob/mcp159
Medina-Villar S, Alonso Á, Castro-Díez P, Pérez-Corona ME (2017) Allelopathic potentials of exotic invasive and native trees over coexisting understory species: the soil as modulator. Plant Ecol 218:579–594. https://doi.org/10.1007/s11258-017-0713-2
Mommer L, Lenssen JPM, Huber H et al (2006) Ecophysiological determinants of plant performance under flooding: a comparative study of seven plant families. J Ecol 94:1117–1129. https://doi.org/10.1111/j.1365-2745.2006.01175.x
Owings CF, Jacobs DF, Shields JM et al (2017) Individual and interactive effects of white-tailed deer and an exotic shrub on artificial and natural regeneration in mixed hardwood forests. AoB Plants. https://doi.org/10.1093/aobpla/plx024
Pedrol MN, González L, Reigosa MJ (2006) Allelopathy and abiotic stress. In: Reigosa MJ, Pedrol N, González L (eds) Allelopathy: a physiological process with ecological implications. Kluwer Academic Publishers, Dordrecht
Prasch CM, Sonnewald U (2013) Simultaneous application of heat, drought, and virus to Arabidopsis plants reveals significant shifts in signaling networks. Plant Physiol 162:1849–1866. https://doi.org/10.1104/pp.113.221044
Pugnaire FI, Luque MT (2001) Changes in plant interactions along a gradient of environmental stress. Oikos 93:42–49. https://doi.org/10.1034/j.1600-0706.2001.930104.x
Qi M, Sun T, Xue S et al (2018) Competitive ability, stress tolerance and plant interactions along stress gradients. Ecology 99:848–857. https://doi.org/10.1002/ecy.2147
R Core Team (2017) R: a language and environment for statistical computing. https://www.R-project.org/. Accessed Oct 2019
Reigosa MJ, Pedrol N, Sánchez-Moreiras A, González L (2002) Stress and allelopathy. In: Reigosa MJ, Pedrol N (eds) Allelopathy from molecules to ecosystems. Science Publisher Inc., Enfield, pp 231–256
Rice EL (1984) Allelopathy, 2nd edn. Academic Press, New York, p 422
Ruan X, De Pan C, Liu R et al (2016) Effects of climate warming on plant autotoxicity in forest evolution: a case simulation analysis for Picea schrenkiana regeneration. Ecol Evol 6:5854–5866. https://doi.org/10.1002/ece3.2315
Schuetzenmeister A, Dufey F (2017) VCA: variance component analysis. R package version 1.3.3. https://CRAN.R-project.org/package=VCA. Accessed Oct 2019
Selmar D, Kleinwächter M (2013) Stress enhances the synthesis of secondary plant products: the impact of stress-related over-reduction on the accumulation of natural products. Plant Cell Physiol 54:817–826. https://doi.org/10.1093/pcp/pct054
Skurski TC, Rew LJ, Maxwell BD (2014) Mechanisms underlying nonindigenous plant impacts: a review of recent experimental research. Invas Plant Sci Manag 7:432–444. https://doi.org/10.1614/IPSM-D-13-00099.1
Tet-Vun C, Ismail BS (2006) Field evidence of the allelopathic properties of Dicranopteris linearis. Weed Biol Manag 6:59–67. https://doi.org/10.1111/j.1445-6664.2006.00203.x
Tharayil N, Bhowmik PC, Xing BS (2006) Preferential sorption of phenolic phytotoxins to soil: implications for altering the availability of allelochemicals. J Agric Food Chem 54:3033–3040. https://doi.org/10.1021/jf053167q
Tharayil N, Suseela V, Triebwasser DJ et al (2011) Changes in the structural composition and reactivity of Acer rubrum leaf litter tannins exposed to warming and altered precipitation: climatic stress-induced tannins are more reactive. New Phytol 191:132–145. https://doi.org/10.1111/j.1469-8137.2011.03667.x
Thelen GC, Vivanco JM, Newingham B et al (2005) Insect herbivory stimulates allelopathic exudation by an invasive plant and the suppression of natives. Ecol Lett 8:209–217. https://doi.org/10.1111/j.1461-0248.2004.00713.x
Tongma S, Kobayashi K, Usui K (2001) Allelopathic activity of Mexican sunflower [Tithonia diversifolia (Hemsl.) A. Gray] in soil under natural field conditions and different moisture conditions. Weed Biol Manag 1:115–119. https://doi.org/10.1046/j.1445-6664.2001.00020.x
Top SM, Preston CM, Dukes JS, Tharayil N (2017) Climate influences the content and chemical composition of foliar tannins in green and senesced tissues of Quercus rubra. Front Plant Sci 8:1–12. https://doi.org/10.3389/fpls.2017.00423
van Kooten O, Snell JFH (1990) The use of chlorophyll fluorescence nomenclature in plant stress physiology. Photosynth Res 25:147–150
Vartapetian BB, Jackson MB (1997) Plant adaptations to anaerobic stress. Ann Bot 79:3–20. https://doi.org/10.1006/anbo.1996.0295
Verdú M, Traveset A (2005) Early emergence enhances plant fitness: a phylogenetically controlled meta-analysis. Ecology 86:1385–1394. https://doi.org/10.1890/04-1647
Wardle DA, Ahmed M, Nicholson KS (1991) Allelopathic influence of nodding thistle (Carduus nutans L.) seeds on germination and radicle growth of pasture plants. New Zeal J Agric Res 34:185–191. https://doi.org/10.1080/00288233.1991.10423358
Wellburn AR (1994) The spectral determination of chlorophylls a and b, total carotenoids using various solvents with spectrophotometers of different resolution. J Plant Physiol 144:307–313. https://doi.org/10.1016/S0176-1617(11)81192-2
Acknowledgements
The authors gratefully acknowledge the supports of the REMEDINAL3-CM S2013/MAE-2719 network (Comunidad de Madrid), UCM Research Groups Program (2018. Research group 91034. Plant Evolutionary Ecology and Restoration Ecology) and the project FONDECYT-CONICYT No 3180289 (Chile). The authors also thank the help of Jose Sigala with the lab work and Emily C. Thyroff with field plant collections.
Author information
Authors and Affiliations
Corresponding author
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Electronic supplementary material
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Medina-Villar, S., Uscola, M., Pérez-Corona, M.E. et al. Environmental stress under climate change reduces plant performance, yet increases allelopathic potential of an invasive shrub. Biol Invasions 22, 2859–2881 (2020). https://doi.org/10.1007/s10530-020-02286-6
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10530-020-02286-6