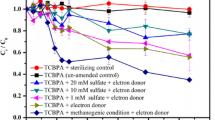
Abstract
Experiments with Fe(III)-rich, chloroethene-contaminated sediment demonstrated that trichloroethylene (TCE) and vinyl chloride (VC) were completely reduced to ethene regardless of whether electron donor(s) were added at 1 × stoichiometry or 10 × stoichiometry relative to all-electron acceptors. Unamended controls uniformly reduced TCE to ethene with a mean time to complete dechlorination (operationally defined as the presence of stoichiometric ethene production) of 79 days. Adding 1 × and 10 × acetate hindered the rate and extent of TCE and VC reduction relative to unamended controls, with several only partially reduced when the experiments were terminated. Adding high molecular mass (soybean oil derivative) substrates did not increase microbial reductive dechlorination relative to unamended incubations, and in many cases, hindered microbial dechlorination in favor of methanogenesis. The mean time to complete dechlorination was comparable between low (× 1) and high (× 10) electron donor concentration for all lipid-based electron donors tested. Those tested included Newman Zone® Standard without sodium lactate (96 vs. 75 days, respectively), CAP 18 ME (85 vs. 94 days, respectively), EOS 598B42 (68 vs. 72 days, respectively), and acetate (134 vs. 125 days, respectively). These data suggest that the addition of an electron donor does not always increase the rate and extent of reductive dechlorination but will increase costs. In particular, increasing the concentration of electron donors higher than the stoichiometric demand only decreased complete microbial reductive dechlorination, which is the opposite of most standard “more time and more electrons” approaches. These data argue that site-specific electron donor demands must be evaluated, and in some cases, a monitored natural attenuation (MNA) approach is most favorable.





Similar content being viewed by others
References
Adamson DT, McDade JM, Hughes JB (2003) Inoculation of a DNAPL source zone to initiate reductive dechlorination of PCE. Environ Sci Technol 37:2525–2533
Adetutu EM, Gundry TD, Patil SS, Golneshin A, Adigun J, Bhaskarla V, Aleer S, Shahsavari E, Ross E, Ball AS (2015) Exploiting the intrinsic microbial degradative potential for field-based in situ reductive dechlorination of trichloroethene contaminated groundwater. J Hazard Mater 300:48–57. https://doi.org/10.1016/j.jhazmat.2015.06.055
Aulenta F, Potalivo M, Majone M, Papini MP, Tandoi V (2006) Anaerobic bioremediation of groundwater containing a mixture of 1,1,2,2-tetrachloroethane and chloroethenes. Biodegradation 17:193–206. https://doi.org/10.1007/s10532-005-4218-7
Aulenta F, Canosa A, Leccese M, Papini MP, Majone M, Viottit P (2007) Field study of in situ anaerobic bioremediation of a chlorinated solvent source zone. Industrial Eng Chem Res 46:6812–6819. https://doi.org/10.1021/ie070048m
Azam HM, Finneran KT (2013) Ferric iron amendment increases Fe(III)-reducing microbial diversity and carbon oxidation in on-site wastewater systems. Chemosphere 90:1435–1443. https://doi.org/10.1016/j.chemosphere.2012.09.002
Basu NB, Fure AD, Jawitz JW (2008) Predicting dense nonaqueous phase liquid dissolution using a simplified source depletion model parameterized with partitioning tracers. Water Resour Res 44:W07414. https://doi.org/10.1029/2007wr006008
Bouwer EJ, McCarty PL (1983) Transformations of 1-carbon and 2-carbon halogenated aliphatic organic-compounds under methanogenic conditions. Appl Environ Microbiol 45:1286–1294. https://doi.org/10.1128/AEM.45.4.1286-1294.1983
Bryant D, Van Vooren K, Haghebaert S (2005) CAP18TM: properties and mechanisms. European Remediation Technologies, Knokke Heist
Capiro NL, Granbery EK, Lebron CA, Major DW, McMaster ML, Pound MJ, Loeffler FE, Pennell KD (2011) Liquid-liquid mass transfer of partitioning electron donors in chlorinated solvent source Zones. Environ Sci Technol 45:1547–1554. https://doi.org/10.1021/es102249x
Conrad R (1996) Soil microorganisms as controllers of atmospheric trace gases (H-2, CO, CH4, OCS, N2O, and NO). Microbiol Rev 60:609–640
Cope N, Hughes JB (2001) Biologically-enhanced removal of PCE from NAPL source zones. Environ Sci Technol 35:2014–2021. https://doi.org/10.1021/es0017357
Cordruwisch R, Seitz HJ, Conrad R (1988) The capacity of hydrogenotrophic anaerobic-bacteria to compete for traces of hydrogen depends on the redox potential of the terminal electron-acceptor. Arch Microbiol 149:350–357
Cupples AM, Spormann AM, McCarty PL (2004) Vinyl chloride and cis-dichloroethene reductive dechlorination kinetics and microorganism growth under substrate limiting conditions. Environ Sci Technol 38:1102–1107. https://doi.org/10.1021/es0348647
Da Silva MLB, Daprato RC, Gomez DE, Hughes JB, Ward CH, Alvarez PJJ (2006) Comparison of bioaugmentation and biostimulation for the enhancement of dense nonaqueous phase liquid source zone bioremediation. Water Environ Res 78:2456–2465. https://doi.org/10.2175/106143006x123111
Ding C, Alvarez-Cohen L, He J (2018) Growth of Dehalococcoides mccartyi species in an autotrophic consortium producing limited acetate. Biodegradation 29:487–498. https://doi.org/10.1007/s10532-018-9846-9
Eaddy A (2008) Development and evaluation of an enrichment culture for bioaugmentation of the p-area chlorinated ethene plume at the savannah river national laboratory. Clemson University, Clemson
EOS Remediation (2013) EOS PRO product information sheet. EOS Remediation LCC, Raleigh
Gossett JM (1987) Measurement of Henrys law constants for c1 and c2 chlorinated hydrocarbons. Environ Sci Technol 21:202–208. https://doi.org/10.1021/es00156a012
Grady CPL, Daigger G, Love N, Filipe C (2011) Biological wastewater treatment, 3rd edn. CRC Press Inc, Boca Raton
Haluska A, Schaefer C, Cho J, Lavorgna G, Annable M (2019) Long-term mass flux assessment of a DNAPL source area treated using bioremediation. J Contam Hydrol 277:103516. https://doi.org/10.1016/j.jconhyd.2019.103516
Harkness MR, Bracco AA, Brennan MJ, Deweerd KA, Spivack JL (1999) Use of bioaugmentation to stimulate complete reductive dechlorination of trichloroethene in dover soil columns. Environ Sci Technol 33:1100–1109. https://doi.org/10.1021/es9807690
He JZ, Sung Y, Dollhopf ME, Fathepure BZ, Tiedje JM, Loffler FE (2002) Acetate versus hydrogen as direct electron donors to stimulate the microbial reductive dechlorination process at chloroethene-contaminated sites. Environ Sci Technol 36:3945–3952. https://doi.org/10.1021/es025528d
He B, He J, Wang F, Lian Y, Zhao Y (2018) Migration, clogging, and carbon source release of nano emulsified vegetable oil in porous media, evaluated by column experiments. Bioremediat J 22:33–42. https://doi.org/10.1080/10889868.2018.1476451
Hood ED, Major DW, Quinn JW, Yoon WS, Gavaskar A, Edwards EA (2008) Demonstration of enhanced bioremediation in a TCE source area at launch complex 34, cape canaveral air force station. Ground Water Monit Remediat 28:98–107. https://doi.org/10.1111/j.1745-6592.2008.00197.x
Imfeld G, Nijenhuis I, Nikolausz M, Zeiger S, Paschke H, Drangmeister J, Grossmann J, Richnow HH, Weber S (2008) Assessment of in situ degradation of chlorinated ethenes and bacterial community structure in a complex contaminated groundwater system. Water Res 42:871–882. https://doi.org/10.1016/j.watres.2007.08.035
Krug T, Cox E, Major D, Harkness M (2013) Economics and valuation of bioaugmentation. In: Stroo H, Leeson A, Hard C (eds) Bioaugmentation for groundwater remediation. Springer, New York, pp 313–332
Kwon MJ, Finneran KT (2008) Biotransformation products and mineralization potential for hexahydro-1,3,5-trinitro-1,3,5-triazine (RDX) in abiotic versus biological degradation pathways with anthraquinone-2,6-disulfonate (AQDS) and Geobacter metallireducens. Biodegradation 19:705–715. https://doi.org/10.1007/s10532-008-9175-5
Leahy JG, Shreve GS (2000) The effect of organic carbon on the sequential reductive dehalogenation of tetrachloroethylene in landfill leachates. Water Res 34:2390–2396. https://doi.org/10.1016/S0043-1354(99)00389-9
Lee MD, Odom JM, Buchanan RJ (1998) New perspectives on microbial dehalogenation of chlorinated solvents: Insights from the field. Annu Rev Microbiol 52:423–452. https://doi.org/10.1146/annurev.micro.52.1.423
Leeson A, Becvar E, Henry B, Fortenberry J, Coyle C (2004) Technical report TR-2250-ENV: principles and practices of enhanced anaerobic bioremediation of chlorinated solvents. ESTCP Program Office, Arlington
Lendvay JM, Loffler FE, Dollhopf M, Aiello MR, Daniels G, Fathepure BZ, Gebhard M, Heine R, Helton R, Shi J, Krajmalnik-Brown R, Major CL, Barcelona MJ, Petrovskis E, Hickey R, Tiedje JM, Adriaens P (2003) Bioreactive barriers: a comparison of bioaugmentation and biostimulation for chlorinated solvent remediation. Environ Sci Technol 37:1422–1431. https://doi.org/10.1021/es025985u
Liang SH, Kuo YC, Chen SH, Chen CY, Kao CM (2013) Development of a slow polycolloid-releasing substrate (SPRS) biobarrier to remediate TCE-contaminated aquifers. J Hazard Mater 254:107–115. https://doi.org/10.1016/j.jhazmat.2013.03.047
Löffler FE, Tiedje JM, Sanford RA (1999) Fraction of electrons consumed in electron acceptor reduction and hydrogen thresholds as indicators of halorespiratory physiology. Appl Environ Microbiol 65:4049–4056
Lorah MM, Olsen LD (1999) Natural attenuation of chlorinated volatile organic compounds in a freshwater tidal wetland: field evidence of anaerobic biodegradation. Water Resour Res 35:3811–3827. https://doi.org/10.1029/1999wr900116
Lovley DR (1985) Minimum threshold for hydrogen metabolism in methanogenic bacteria. Appl Environ Microbiol 49:1530–1531
Lovley DR (1997a) Microbial Fe(III) reduction in subsurface environments. Fems Microbiol Rev 20:305–313. https://doi.org/10.1016/s0168-6445(97)00013-2
Lovley DR (1997b) Potential for anaerobic bioremediation of BTEX in petroleum-contaminated aquifers. J Indus Microbiol Biotechnol 18:75–81. https://doi.org/10.1038/sj.jim.2900246
Lovley DR, Phillips EJP (1986) Organic-matter mineralization with reduction of ferric iron in anaerobic sediments. Appl Environ Microbiol 51:683–689. https://doi.org/10.1128/AEM.51.4.683-689.1986
Lu XX, Tao S, Bosma T, Gerritse J (2001) Characteristic hydrogen concentrations for various redox processes in batch study. J Environ Sci Health Part A 36:1725–1734. https://doi.org/10.1081/ese-100106254
Lyon D, Vogel T (2013) Chapter 1: bioaugmentation for groundwater remediation: an overview. In: Stroo H, Leeson A, Ward C (eds) Bioaugmentation for groundwater remediation. Springer, New York
Major DW, McMaster ML, Cox EE, Edwards EA, Dworatzek SM, Hendrickson ER, Starr MG, Payne JA, Buonamici LW (2002) Field demonstration of successful bioaugmentation to achieve reductive dechlorination of tetrachloroethene to ethene. Environ Sci Technol 36:5106–5116. https://doi.org/10.1021/es0255711
McLean JE, Ervin J, Zhou J, Sorensen DL, Dupont RR (2015) Biostimulation and bioaugmentation to enhance reductive dechlorination of tce in a long-term flow through column study. Ground Water Monit Remediat 35:76–88. https://doi.org/10.1111/gwmr.12113
Millerick KA, Johnston JT, Finneran KT (2016) Photobiological transformation of hexahydro-1,3,5-trinitro-1,3,5-triazine (RDX) using Rhodobacter sphaeroides. Chemosphere 159:138–144. https://doi.org/10.1016/j.chemosphere.2016.05.056
Mills JC, Wilson JT, Wilson BH, Wiedemeier TH, Freedman DL (2018) Quantification of TCE co-oxidation in groundwater using a c-14-assay. Ground Water Monit Remediat 38:57–67. https://doi.org/10.1111/gwmr.12266
Mirza BS, Sorensen DL, Dupont RR, McLean JE (2016) Dehalococcoides abundance and alternate electron acceptor effects on large, flow-through trichloroethene dechlorinating columns. Appl Microbio Biotechnol 100:2367–2379. https://doi.org/10.1007/s00253-015-7112-1
Naidu R, Nandy S, Megharaj M, Kumar RP, Chadalavada S, Chen Z, Bowman M (2012) Monitored natural attenuation of a long-term petroleum hydrocarbon contaminated sites: a case study. Biodegradation 23(6):881–895. https://doi.org/10.1007/s10532-012-9580-7
Perez-de-Mora A, Zila A, McMaster ML, Edwards EA (2014) Bioremediation of chlorinated ethenes in fractured bedrock and associated changes in dechlorinating and nondechlorinating microbial populations. Environ Sci Technol 48:5770–5779. https://doi.org/10.1021/es404122y
Prommer H, Aziz LH, Bolano N, Taubald H, Schueth C (2008) Modelling of geochemical and isotopic changes in a column experiment for degradation of TCE by zero-valent iron. J Contam Hydrol 97:13–26. https://doi.org/10.1016/j.jconhyd.2007.11.003
Remediation and Natural Attenuation Services Inc (2013) Newman zone emulsified vegetable oil product ingredients. Remediation and Natural Attenuation Services Inc. http://www.rnasinc.com/newman-zone/product-description/product-ingredients/more. accessed Jan 2014
Rodriguez E, McGuinness KA, Ophori DU (2004) A field evaluation of enhanced reductive dechlorination of chlorinated solvents in groundwater, New York Metropolitan. Area Environ Geol 45(5):623–632
Schaefer CE, Condee CW, Vainberg S, Steffan RJ (2009) Bioaugmentation for chlorinated ethenes using Dehalococcoides sp.: comparison between batch and column experiments. Chemosphere 75:141–148. https://doi.org/10.1016/j.chemosphere.2008.12.041
Schaefer CE, Lippincott DR, Steffan RJ (2010) Field-scale evaluation of bioaugmentation dosage for treating chlorinated ethenes. Ground Water Monit Remediat 30:113–124. https://doi.org/10.1111/j1745-6592.2010.01297.x
Schaefer CE, Lavorgna GM, Haluska AA, Annable MD (2018) Long-term impacts on groundwater and reductive dechlorination following bioremediation in a highly characterized trichloroethene DNAPL source area. Ground Water Monit Remediat 38:65–74. https://doi.org/10.1111/gwmr.12294
Scheutz C, Durant ND, Dennis P, Hansen MH, Jorgensen T, Jakobsen R, Cox EE, Bjerg PL (2008) Concurrent ethene generation and growth of dehalococcoides containing vinyl chloride reductive dehalogenase genes during an enhanced reductive dechlorination field demonstration. Environ Sci Technol 42:9302–9309. https://doi.org/10.1021/es800764t
Scheutz C, Broholm MM, Durant ND, Weeth EB, Jorgensen TH, Dennis P, Jacobsen CS, Cox EE, Chambon JC, Bjerg PL (2010) Field evaluation of biological enhanced reductive dechlorination of chloroethenes in clayey till. Environ Sci Technol 44:5134–5141. https://doi.org/10.1021/es1003044
Schink B (1997) Energetics of syntrophic cooperation in methanogenic degradation. Microbiol Mol Biol Rev 61:262–280. https://doi.org/10.1128/.61.2.262-280.1997
Semkiw ES, Dybas MJ, Barcelona MJ (2009) Evaluation of three electron-donor permeable reactive barrier materials for enhanced reductive dechlorination of trichloroethene. Bioremediation J 13:7–20. https://doi.org/10.1080/10889860802690489
Smith S, Dupont RR, McLean JE (2019) Arsenic release and attenuation processes in a groundwater aquifer during anaerobic remediation of tce with biostimulation. Ground Water Monit Remediat 39:61–70. https://doi.org/10.1111/gwmr.12340
Soga K, Page JWE, Illangasekare TH (2004) A review of NAPL source zone remediation efficiency and the mass flux approach. J Hazard Mater 110:13–27. https://doi.org/10.1016/j.jhazmat.2004.02.034
Song DL, Conrad ME, Sorenson KS, Alvarez-Cohen L (2002) Stable carbon isotope fractionation during enhanced in situ bioremediation of trichloroethene. Environ Sci Technol 36:2262–2268. https://doi.org/10.1021/es011162d
van Eekert MHA, Schroder TJ, van Rhee A, Stams AJM, Schraa G, Field JA (2001) Constitutive reductive dechlorination of chlorinated ethenes by a methanol degrading methanogenic consortium. Bioresource Technol 77:163–170. https://doi.org/10.1016/s0960-8524(00)00149-8
Weatherill JJ, Atashgahi S, Schneidewind U, Krause S, Ullah S, Cassidy N, Rivett MO (2018) Natural attenuation of chlorinated ethenes in hyporheic zones: a review of key biogeochemical processes and in-situ transformation potential. Water Res 128:362–382. https://doi.org/10.1016/j.watres.2017.10.059
Wei N, Finneran KT (2009) Microbial community analyses of three distinct, liquid cultures that degrade methyl tert-butyl ether using anaerobic metabolism. Biodegradation 20:695–707. https://doi.org/10.1007/s10532-009-9257-z
Wei N, Finneran KT (2011) Influence of ferric iron on complete reductive dechlorination of trichloroethylene (TCE) to ethene: Fe(III) reduction does not always inhibit complete reductive dechlorination. Environ Sci Technol 45(17):7422–7430. https://doi.org/10.1021/es201501a
Wei N, Finneran KT (2013) Low and high acetate amendments are equally as effective at promoting complete reductive dechlorination of trichloroethylene (TCE). Biodegradation 24:413–425. https://doi.org/10.1007/s10532-012-9598-x
Yang Y, McCarty PL (1998) Competition for hydrogen within a chlorinated solvent dehalogenating anaerobic mixed culture. Environ Sci Technol 32:3591–3597. https://doi.org/10.1021/es980363n
Yang Y, McCarty PL (2000) Biologically enhanced dissolution of tetrachloroethene DNAPL. Environ Sci Technol 34:2979–2984. https://doi.org/10.1021/es991410u
Yang MJE, Annable MD, Jawitz JW (2015) Back diffusion from thin low permeability zones. Environ Sci Technol 49:415–422. https://doi.org/10.1021/es5045634
Yang Yi, Higgins SA, Yan J, Simsir B, Chourney K, Iyer R, Hettich RL, Baldwin B, Ogles DM, Loeffler FE (2017) Grape pomace compost harbors organohalide-respiring Dehalaogenimonas species with novel reductive dehaloganse genes. ISME J 11:2767–2780
Yu S, Semprini L (2009) Enhanced reductive dechlorination of PCE DNAPL with TBOS as a slow-release electron donor. J Contam Hydrol 167:97–104. https://doi.org/10.1016/j.jhazmat.2008.12.087
Zhuang P, Pavlostathis SG (1995) Effect of temperature, pH and electron donor on the microbial reductive dechlorination of chloroalkenes. Chemosphere 31:3537–3548
Acknowledgements
This project was funded by Clemson University external faculty grants. The authors want to specifically acknowledge Bob Borden (EOS), Matthew Dingens (CAP 18), and William Newman (Newman Zone) for providing the vegetable oils used in this research. The results and conclusions presented herein are those of the authors and do not necessarily represent those of Clemson University, and no endorsement of the described technology is implied. This paper has not been subjected to peer review within Clemson University or by any of the Vendors who provided materials for this study, and the conclusions stated here do not necessarily reflect the official views of the University or the Vendors, nor does this document constitute an official endorsement by the University or the Vendors. The authors thank the anonymous reviewers for their feedback.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
The authors declare that they have no conflict of interest.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Haluska, A.A., Finneran, K.T. Increasing electron donor concentration does not accelerate complete microbial reductive dechlorination in contaminated sediment with native organic carbon. Biodegradation 32, 577–593 (2021). https://doi.org/10.1007/s10532-021-09953-y
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10532-021-09953-y