Abstract
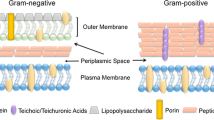
Iron-starved Mycobacterium tuberculosis utilises the carboxymycobactin-mycobactin siderophore machinery to acquire iron. These two siderophores have high affinity for ferric iron and can withdraw the metal ion from insoluble iron hydroxides and iron-binding proteins. We first reported HupB, a multi-functional mycobacterial protein to be associated with iron acquisition in M. tuberculosis. This 28 kDa cell wall protein, up regulated upon iron limitation functions as a transcriptional activator of mycobactin biosynthesis and is essential for the pathogen to survive inside macrophages. The focus of this study is to understand the role of HupB in iron uptake and transport by the carboxmycobactin-mycobactin siderophore machinery in M. tuberculosis. Experimental approaches included radiolabelled iron uptake studies by viable organisms and protein–ligand binding studies using the purified HupB and the two siderophores. Uptake of 55Fe-carboxymycobactin by wild type M. tuberculosis (WT M.tb.H37Rv) and not by the hupB KO mutant (M.tb.ΔhupB) showed that HupB is necessary for the uptake of ferri-carboxymycobactin. Additionally, the radiolabel recovery was high in HupB-incorporated liposomes upon addition of the labelled siderophore. Bioinformatic and experimental studies using spectrofluorimetry, CD analysis and surface plasmon resonance not only confirmed the binding of HupB with ferri-carboxymycobactin and ferri-mycobactin but also with free iron. In conclusion, HupB is established as a ferri- carboxymycobactin receptor and by virtue of its property to bind ferric iron, functions as a transporter of the ferric iron from the extracellular siderophore to mycobactin within the cell envelope.






taken from 250 to 200 nm. Panels A, B and C show the spectral changes in HupB upon addition of ferri-carboxymycobactin, ferri-mycobactin and desferri-carboxymycobactin respectively. The upward arrow indicates addition of aliquots of ferri-carboxymycobactin and ferri-mycobactin to a maximal concentration of 3 µM (Panels A and B). In Panel C, a maximal concentration of 50 µM of desferri- carboxymycobactin was added to HupB (Panel C). The spectra in grey colour are control data of buffer (solid lines) and buffer + acetonitrile (dashed lines)



Similar content being viewed by others
References
Anthis NJ, Clore GM (2013) Sequence-specific determination of protein and peptide concentrations by absorbance at 205 nm. Protein Sci. https://doi.org/10.1002/pro.2253
Barclay R, Ewing DF, Ratledge C (1985) Isolation, identification, and structural analysis of the mycobactins of Mycobacterium avium, Mycobacterium intracellulare, Mycobacterium scrofulaceum, and Mycobacterium paratuberculosis. J Bacteriol. https://doi.org/10.1128/jb.164.2.896-903.1985
Braun V, Hantke K (2003) Mechanisms of bacterial iron transport. In: Winkelmann G (ed) Microbial transport systems. Wiley, Hoboken
Daylight Chemical Information Systems (2011) 3. SMILES—A simplified chemical language. Daylight Theory Manual, Version 49
Engh RA, Huber R (1991) Accurate bond and angle parameters for X-ray protein structure refinement. Acta Crystallogr Sect A. https://doi.org/10.1107/S0108767391001071
Geourjon C, Deléage G (1995) Sopma: significant improvements in protein secondary structure prediction by consensus prediction from multiple alignments. Bioinformatics. https://doi.org/10.1093/bioinformatics/11.6.681
Gobin J, Horwitz MA (1996) Exochelins of Mycobacterium tuberculosis remove iron from human iron-binding proteins and donate iron to mycobactins in the M. tuberculosis cell wall. J Exp Med. https://doi.org/10.1084/jem.183.4.1527
Gobin J, Moore CH, Reeve JR et al (1995) Iron acquisition by Mycobacterium tuberculosis: isolation and characterization of a family of iron-binding exochelins. Proc Natl Acad Sci USA. https://doi.org/10.1073/pnas.92.11.5189
Holmes MA, Paulsene W, Jide X et al (2005) Siderocalin (Lcn 2) Also binds carboxymycobactins, potentially defending against mycobacterial infections through iron sequestration. Structure 13:29–41. https://doi.org/10.1016/j.str.2004.10.009
Hołówka J, Trojanowski D, Gind K et al (2017) HupB is a bacterial nucleoid-associated protein with an indispensable eukaryotic-like tail. MBio. https://doi.org/10.1128/mBio.01272-17
Hooft RWW, Vriend G, Sander C, Abola EE (1996) Errors in protein structures. Nature 381:272
Kalra P, Mishra SK, Kaur S, Kumar A, Prasad HK, Sharma TK, Tyagi J (2018) G-quadruplex-forming DNA aptamers inhibit the DNA-binding function of HupB and Mycobacterium tuberculosis entry into host cells. Mol Ther Nucleic Acids 13:99–109
Kochan I (1973) The role of iron in bacterial infections, with special consideration of host-tubercle bacillus interaction. Curr Top Microbiol Immunol 60:1–30
Kumar N, Sritharan M (2020) Role of a 21 kDa iron-regulated protein IrpA in the uptake of ferri-exochelin by Mycobacterium smegmatis. J Appl Microbiol. https://doi.org/10.1111/jam.14728
Laskowski RA, Swindells MB (2011) LigPlot+: multiple ligand-protein interaction diagrams for drug discovery. J Chem Inf Model. https://doi.org/10.1021/ci200227u
Lovell SC, Davis IW, Arendall WB et al (2003) Structure validation by Cα geometry: φ, ψ and Cβ deviation. Proteins Struct Funct Genet. https://doi.org/10.1002/prot.10286
Matsumoto S, Yukitake H, Furugen M, Matsuo T, Mineta TYT (1999) Identification of a novel DNA-binding protein from Mycobacterium bovis bacillus Calmette-Guerin. Microbiol Immunol 43:1027–1036
Pandey SD, Choudhury M, Yousuf S et al (2014) Iron-regulated protein HupB of Mycobacterium tuberculosis positively regulates siderophore biosynthesis and is essential for growth in macrophages. J Bacteriol. https://doi.org/10.1128/JB.01483-13
Ratledge C (2004) Iron, mycobacteria and tuberculosis. Tuberculosis 84:110–130
Ratledge C, Ewing M (1996) The occurrence of carboxymycobactin, the siderophore of pathogenic mycobacteria, as a second extracellular siderophore in Mycobacterium smegmatis. Microbiology. https://doi.org/10.1099/13500872-142-8-2207
Ryndak MB, Wang S, Smith I, Rodriguez GM (2010) The Mycobacterium tuberculosis high-affinity iron importer, IrtA, contains an FAD-binding domain. J Bacteriol. https://doi.org/10.1128/JB.00223-09
Sali A, Blundell T (1994) Comparative modelling by satisfaction of spatial restraints. J Mol Biol 234:779–815
Shimoji Y, Vincent NG, Matsumura K et al (1999) A 21-kDa surface protein of Mycobacterium leprae binds peripheral nerve laminin-2 and mediates Schwann cell invasion. Proc Natl Acad Sci USA. https://doi.org/10.1073/pnas.96.17.9857
Sritharan M (2016) Iron homeostasis in Mycobacterium tuberculosis: mechanistic insights into siderophore-mediated Iron uptake. J Bacteriol. https://doi.org/10.1128/JB.00359-16
Sritharan N, Choudhury M, Sivakolundu S et al (2014) Highly immunoreactive antibodies against the rHup-F2 fragment (aa 63–161) of the iron-regulated HupB protein of Mycobacterium tuberculosis and its potential for the serodiagnosis of extrapulmonary and recurrent tuberculosis. Eur J Clin Microbiol Infect Dis. https://doi.org/10.1007/s10096-014-2203-y
Takatsuka M, Osada-Oka M, Satoh EF et al (2011) A histone-like protein of mycobacteria possesses ferritin superfamily protein-like activity and protects against DNA damage by Fenton reaction. PLoS ONE. https://doi.org/10.1371/journal.pone.0020985
Trott O, Olson AJ (2010) AutoDock Vina: improving the speed and accuracy of docking with a new scoring function, efficient optimization, and multithreading. J Comput Chem 31:455–461. https://doi.org/10.1002/jcc.21334
Vriend G (1990) WHAT IF: a molecular modeling and drug design program. J Mol Graph. https://doi.org/10.1016/0263-7855(90)80070-V
WHO (2020) WHO | Global tuberculosis report 2019
Yaseen I, Choudhury M, Sritharan M, Khosla S (2017) Histone methyltransferase SUV39H1 participates in host defense by methylating mycobacterial histone-like protein HupB. EMBO J. https://doi.org/10.15252/embj.201796918
Yeruva VC, Duggirala S, Lakshmi V et al (2006) Identification and characterization of a major cell wall-associated iron-regulated envelope protein (Irep-28) in Mycobacterium tuberculosis. Clin Vaccine Immunol. https://doi.org/10.1128/CVI.00125-06
Acknowledgements
MC, TNK and NK thank the University Grants Commission for providing fellowship during the doctoral study. MS acknowledges the financial assistance by UPE-II program of University of Hyderabad. KD acknowledges the financial assistance for doctoral studies by the University of Hyderabad.
Author information
Authors and Affiliations
Corresponding author
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Choudhury, M., Koduru, T.N., Kumar, N. et al. Iron uptake and transport by the carboxymycobactin-mycobactin siderophore machinery of Mycobacterium tuberculosis is dependent on the iron-regulated protein HupB. Biometals 34, 511–528 (2021). https://doi.org/10.1007/s10534-021-00292-2
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10534-021-00292-2