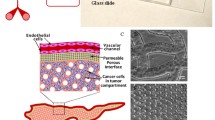
Abstract
The advent of microfluidic technologies has enabled a better recapitulation of in vitro tumor model with higher biological relevance over conventional monolayer assays. This work built upon a microfluidic system that supported the spontaneous aggregate formation of tumoral cells under flow-induced dynamic physical forces in a confined microchamber without additional matrix materials. Our findings indicated that fluidic streams significantly modulated the biological and architectural features of human breast adenocarcinoma cell (MCF-7), human hepatocarcinoma cell (HepG2), and human cervix adenocarcinoma cell (HeLa) with cell-type-dependent variation. The microfluidic platform was further integrated with a fluorescence detection and imaging system, allowing for non-invasive monitoring of cellular accumulation and spatial distribution of a chemotherapeutic agent, doxorubicin (DOX). The cytotoxic effects of DOX of various concentrations were determined and compared in MCF-7 cells in conventional two-dimensional (2D) static and microfluidic culture conditions. Dose-dependent response to DOX was noticed in both cultures, whereas tumor micronodules grown in microfluidic devices demonstrated significantly lower sensitivity to DOX at increased concentration. Our platform owns promising potentials as a universal modality for bridging traditional 2D cell cultures and in vivo experimentation for preclinical anticancer drug screening.







Similar content being viewed by others
References
A. Agosti, S. Marchesi, G. Scita, P. Ciarletta, ArXiv, 1905.08074 1 (2019)
N.H. Baek, O.W. Seo, M.S. Kim, J. Hulme, S.S.A. An, Onco. Targets. Ther. 9, 7207 (2016)
A. Casey, M. Gargotti, F. Bonnier, H.J. Byrne, Toxicol. Vitr. 33, 99 (2016)
N. Chaicharoenaudomrung, P. Kunhorm, P. Noisa, World J. Stem Cells 11, 1065 (2019)
R. Edmondson, J.J. Broglie, A.F. Adcock, L. Yang, Assay Drug Dev. Technol. 12, 207 (2014)
Z. Farhane, F. Bonnier, A. Casey, H.J. Byrne, Analyst 140, 4212 (2015)
P. Gunness, D. Mueller, V. Shevchenko, E. Heinzle, M. Ingelman-Sundberg, F. Noor, Toxicol. Sci. 133, 67 (2013)
B. Han, C. Qu, K. Park, S.F. Konieczny, M. Korc, Cancer Lett. 380, 319 (2016)
S. Hehlgans, N. Cordes, Am. J. Cancer Res. 1, 521 (2011)
C.K.M. Ip, S.S. Li, M.Y.H. Tang, S.K.H. Sy, Y. Ren, H.C. Shum, A.S.T. Wong, Sci. Rep. 6, 1 (2016)
Y. Jo, N. Choi, K. Kim, H.J. Koo, J. Choi, H.N. Kim, Theranostics 8, 5259 (2018)
Y. Li, E. Kumacheva, Sci. Adv. 4, eaas8998 (2018)
W. Li, Y. Liu, Z. Qian, Y. Yang, Sci. Rep. 7, 1 (2017)
C.J. Lovitt, T.B. Shelper, V.M. Avery, BMC Cancer 18, 1 (2018)
R. Lüpertz, W. Wätjen, R. Kahl, Y. Chovolou, Toxicology 271, 115 (2010)
M.J. Mitchell, M.R. King, Front. Oncol. 3, 1 (2013a)
M.J. Mitchell, M.R. King, New J. Phys. 18, 015008 (2013b)
M.V. Monteiro, V.M. Gaspar, L.P. Ferreira, J.F. Mano, Biomater. Sci. (2020)
B. Nath, A. Raza, V. Sethi, A. Dalal, S.S. Ghosh, G. Biswas, Sci. Rep. 8, 1 (2018)
C. Novak, E. Horst, G. Mehta, APL Bioeng. 2, 031701 (2018)
A.S. Nunes, A.S. Barros, E.C. Costa, A.F. Moreira, I.J. Correia, Biotechnol. Bioeng. 116, 206 (2019)
Z.C. Nwosu, W. Piorońska, N. Battello, A.D. Zimmer, B. Dewidar, M. Han, S. Pereira, B. Blagojevic, D. Castven, V. Charlestin, P. Holenya, J. Lochead, C. De La Torre, N. Gretz, P. Sajjakulnukit, L. Zhang, M.H. Ward, J.U. Marquardt, M.P. di Magliano, C.A. Lyssiotis, J. Sleeman, S. Wölfl, M.P. Ebert, C. Meyer, U. Hofmann, S. Dooley, EBioMedicine 54, 102699 (2020)
A. Ozcelikkale, K. Shin, V. Noe-Kim, B.D. Elzey, Z. Dong, J.T. Zhang, K. Kim, I.C. Kwon, K. Park, B. Han, J. Control. Release 266, 129 (2017)
X.L. Qian, Y.H. Pan, Q.Y. Huang, Y.B. Shi, Q.Y. Huang, Z.Z. Hu, L.X. Xiong, Onco. Targets Ther. 12, 1539 (2019)
N. Raghunand, R.J. Gillies, Drug Resist. Updat. 3, 39 (2000)
R. Ravizza, M.B. Gariboldi, L. Passarelli, E. Monti, BMC Cancer 4, 1 (2004)
I. Rizvi, U.A. Gurkan, S. Tasoglu, N. Alagic, J.P. Celli, L.B. Mensah, Z. Mai, U. Demirci, T. Hasan, Proc. Natl. Acad. Sci. U. S. A. 110, E1974 (2013)
M. Santoro, S.E. Lamhamedi-Cherradi, B.A. Menegaz, J.A. Ludwig, A.G. Mikos, Proc. Natl. Acad. Sci. U. S. A. 112, 10304 (2015)
D.A. Senthebane, A. Rowe, N.E. Thomford, H. Shipanga, D. Munro, M.A.M. Al Mazeedi, H.A.M. Almazyadi, K. Kallmeyer, C. Dandara, M.S. Pepper, M.I. Parker, K. Dzobo, Int. J. Mol. Sci. 18 (2017)
M. Shang, R.H. Soon, C.T. Lim, B.L. Khoo, J. Han, Lab Chip 19, 369 (2019)
S. Shuford, C. Wilhelm, M. Rayner, A. Elrod, M. Millard, C. Mattingly, A. Lotstein, A.M. Smith, Q.J. Guo, L. O’Donnell, J. Elder, L. Puls, S.J. Weroha, X. Hou, V. Zanfagnin, A. Nick, M.P. Stany, G.L. Maxwell, T. Conrads, A.K. Sood, D. Orr, L.M. Holmes, M. Gevaert, H.E. Crosswell, T.M. DesRochers, Sci. Rep. 9, 1 (2019)
G. Trujillo-de Santiago, B.G. Flores-Garza, J.A. Tavares-Negrete, I.M. Lara-Mayorga, I. González-Gamboa, Y.S. Zhang, A. Rojas-Martínez, R. Ortiz-López, M.M. Álvarez, Materials (Basel) 12 (2019)
K.P. Valente, S. Khetani, A.R. Kolahchi, A. Sanati-Nezhad, A. Suleman, M. Akbari, Drug Discov. Today 22, 1654 (2017)
M. Verheijen, Y. Schrooders, H. Gmuender, R. Nudischer, O. Clayton, J. Hynes, S. Niederer, H. Cordes, L. Kuepfer, J. Kleinjans, F. Caiment, Toxicol. Lett. 294, 184 (2018)
E.T. Verjans, J. Doijen, W. Luyten, B. Landuyt, L. Schoofs, J. Cell. Physiol. 233, 2993 (2018)
P.R. Wachsberger, J. Landry, C. Storck, K. Davis, M.D. O’Hara, C.S. Owen, D.B. Leeper, R.A. Coss, Int. J. Hyperth. 13, 251 (1997)
L.Y. Wu, D. Di Carlo, L.P. Lee, Biomed. Microdevices 10, 197 (2008)
Y. Yang, X. Yang, J. Zou, C. Jia, Y. Hu, H. Du, H. Wang, Lab a Chip - Miniaturisation Chem. Biol. 15, 735 (2015)
Y. Yang, A. Lü, W. Li, Z. Qian, AIP Adv. 9 (2019)
T. Yokochi, K.D. Robertson, Mol. Pharmacol. 66, 1415 (2004)
H. Zhang, L. Xiao, Q. Li, X. Qi, A. Zhou, Biomicrofluidics 12, 024119 (2018)
Acknowledgments
This work was supported by the National Natural Science Foundation of China (61875085, 81727804, 81827803). The authors thank Dr. Yangyang Liu for her technical assistance in manuscript revision.
Author information
Authors and Affiliations
Corresponding author
Additional information
Publisher’s note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Electronic supplementary material
ESM 1
(DOCX 1293 kb)
Rights and permissions
About this article
Cite this article
Yang, Y., Liu, S., Chen, C. et al. Microfluidic-enabled self-organized tumor model for in vitro cytotoxicity assessment of doxorubicin. Biomed Microdevices 22, 70 (2020). https://doi.org/10.1007/s10544-020-00523-2
Accepted:
Published:
DOI: https://doi.org/10.1007/s10544-020-00523-2