Abstract
Purpose
The higher prevalence of cognitive impairment/ dementia among cancer survivors is likely multifactorial. Since both exposures to cytomegalovirus (CMV) and inflammation are common among elderly cancer survivors, we evaluated their contribution towards dementia.
Methods
Data from 1387 cancer survivors and 7004 participants without cancer in the 2016 wave of the Health and Retirement Study (HRS) was used in this study. Two inflammatory biomarkers, C-reactive protein (CRP) and neutrophil–lymphocyte ratio (NLR), were used to create an inflammation score. We used survey logistic regression adjusted for survey design parameters.
Results
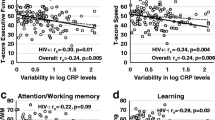
CMV seropositivity was not associated with cognitive impairment among cancer survivors (p = 0.2). In addition, inflammation was associated with elevated odds of cognitive impairment (OR = 2.2, 95% CI [1.2, 4.2]). Cancer survivors who were both CMV seropositive and had increased inflammation had the highest odds of cognitive impairment compared to those who were CMV seronegative and had low inflammation (OR = 3.8, 95% CI [1.5, 9.4]). The stratified analysis among cancer survivors showed this association was seen only among cancer survivors in whom the cancer was diagnosed within three years of measurement of inflammation score and CMV serostatus (OR = 18.5; 95% CI [6.1, 56.1]).
Conclusion
The CMV seropositivity and high inflammation was associated with higher cognitive impairment among cancer survivors. The stronger associations seen among cancer survivors diagnosed within the last three years suggest that strategies to reduce CMV activation and inflammation during or immediately after cancer treatment may be important in reducing the prevalence of cognitive impairment/ dementia among cancer survivors.
Similar content being viewed by others
Data availability
Data used in this manuscript are accessed from Health and Retirement Study website (https://hrs.isr.umich.edu/data-products?_ga=2.8747955.705712069.1606796306-411200660.1601442014). Cognition and basic demographic data used in this study is publicly available to registered members of Health and Retirement Study. The venous blood biomarker data is under sensitive health data and can be accessed by submitting sensitive health data order form (https://hrsdata.isr.umich.edu/data-products/sensitive-health/order-form).
References
Hardy SJ, Krull KR, Wefel JS, Janelsins M (2018) Cognitive changes in cancer survivors. Am Soc Clini Oncol Educational Book Am Soc Clinic Oncol Annual Meeting 38:795–806. https://doi.org/10.1200/edbk_201179
Vardy JL, Dhillon HM, Pond GR et al (2015) Cognitive function in patients with colorectal cancer who do and do not receive chemotherapy: a prospective, longitudinal, controlled study. J Clin Oncol 33(34):4085–4092. https://doi.org/10.1200/jco.2015.63.0905
Pendergrass JC, Targum SD, Harrison JE (2018) Cognitive impairment associated with cancer: a brief review. Innovations in Clin Neurosci 15(1–2):36–44
Bettcher BM, Kramer JH (2014) Longitudinal inflammation, cognitive decline, and alzheimer’s disease: a mini-review. Clin Pharmacol Ther 96(4):464–469. https://doi.org/10.1038/clpt.2014.147
Yaffe K, Kanaya A, Lindquist K et al (2004) The metabolic syndrome, inflammation, and risk of cognitive decline. JAMA 292(18):2237–2242. https://doi.org/10.1001/jama.292.18.2237
Yaffe K, Haan M, Blackwell T, Cherkasova E, Whitmer RA, West N (2007) Metabolic syndrome and cognitive decline in elderly Latinos: findings from the Sacramento area latino study of aging study. J Am Geriatr Soc 55(5):758–762. https://doi.org/10.1111/j.1532-5415.2007.01139.x
Engelhart MJ, Geerlings MI, Meijer J et al (2004) Inflammatory proteins in plasma and the risk of dementia: the rotterdam study. Arch Neurol 61(5):668–672. https://doi.org/10.1001/archneur.61.5.668
Weaver JD, Huang MH, Albert M, Harris T, Rowe JW, Seeman TE (2002) Interleukin-6 and risk of cognitive decline: MacArthur studies of successful aging. Neurology 59(3):371–378. https://doi.org/10.1212/wnl.59.3.371
Tarkowski E, Andreasen N, Tarkowski A, Blennow K (2003) Intrathecal inflammation precedes development of Alzheimer’s disease. J Neurol Neurosurg Psychiatry 74(9):1200–1205. https://doi.org/10.1136/jnnp.74.9.1200
Schmidt R, Schmidt H, Curb JD, Masaki K, White LR, Launer LJ (2002) Early inflammation and dementia: a 25-year follow-up of the Honolulu-Asia Aging Study. Ann Neurol 52(2):168–174. https://doi.org/10.1002/ana.10265
Tan ZS, Beiser AS, Vasan RS et al (2007) Inflammatory markers and the risk of Alzheimer disease: the Framingham Study. Neurology 68(22):1902–1908. https://doi.org/10.1212/01.wnl.0000263217.36439.da
Panova-Noeva M, Schulz A, Arnold N et al (2018) Coagulation and inflammation in long-term cancer survivors: results from the adult population. J Thrombosis Haemostasis 16(4):699–708. https://doi.org/10.1111/jth.13975
van der Willik KD, Koppelmans V, Hauptmann M, Compter A, Ikram MA, Schagen SB (2018) Inflammation markers and cognitive performance in breast cancer survivors 20 years after completion of chemotherapy: a cohort study. Breast Cancer Res 20(1):135. https://doi.org/10.1186/s13058-018-1062-3
Merani S, Pawelec G, Kuchel GA, McElhaney JE (2017) Impact of aging and cytomegalovirus on immunological response to influenza vaccination and infection. Front Immunol 8:784. https://doi.org/10.3389/fimmu.2017.00784
Weiskopf D, Weinberger B, Grubeck-Loebenstein B (2009) The aging of the immune system. Transplant Inter 22(11):1041–1050. https://doi.org/10.1111/j.1432-2277.2009.00927.x
Aiello AE, Haan M, Blythe L, Moore K, Gonzalez JM, Jagust W (2006) The influence of latent viral infection on rate of cognitive decline over 4 years. J Am Geriatr Soc 54(7):1046–1054. https://doi.org/10.1111/j.1532-5415.2006.00796.x
George KM, Folsom AR, Norby FL, Lutsey PL (2020) No association found between midlife seropositivity for infection and subsequent cognitive decline: the atherosclerosis risk in communities neurocognitive study (ARIC-NCS). J Geriatr Psychiatry Neurol 33(1):15–21. https://doi.org/10.1177/0891988719856692
Gow AJ, Firth CM, Harrison R, Starr JM, Moss P, Deary IJ (2013) Cytomegalovirus infection and cognitive abilities in old age. Neurobiol Aging 34(7):1846–1852. https://doi.org/10.1016/j.neurobiolaging.2013.01.011
Mathei C, Vaes B, Wallemacq P, Degryse J (2011) Associations between cytomegalovirus infection and functional impairment and frailty in the BELFRAIL Cohort. J Am Geriatr Soc 59(12):2201–2208. https://doi.org/10.1111/j.1532-5415.2011.03719.x
Stebbins RC, Noppert GA, Yang YC, Dowd JB, Simanek A, Aiello AE (2020) Immune response to cytomegalovirus and cognition in the health and retirement study. Am J Epidemiol. https://doi.org/10.1093/aje/kwaa238
Torniainen-Holm M, Suvisaari J, Lindgren M, Härkänen T, Dickerson F, Yolken RH (2018) Association of cytomegalovirus and Epstein-Barr virus with cognitive functioning and risk of dementia in the general population: 11-year follow-up study. Brain Behav Immun 69:480–485. https://doi.org/10.1016/j.bbi.2018.01.006
Wills MR, Poole E, Lau B, Krishna B, Sinclair JH (2015) The immunology of human cytomegalovirus latency: could latent infection be cleared by novel immunotherapeutic strategies? Cell Mol Immunol 12(2):128–138. https://doi.org/10.1038/cmi.2014.75
Leng SX, Li H, Xue Q-L et al (2011) Association of detectable cytomegalovirus (CMV) DNA in monocytes rather than positive CMV IgG serology with elevated neopterin levels in community-dwelling older adults. Exp Gerontol 46(8):679–684. https://doi.org/10.1016/j.exger.2011.04.002
Bayer C, Varani S, Wang L et al (2013) Human cytomegalovirus infection of M1 and M2 macrophages triggers inflammation and autologous T-cell proliferation. J Virol 87(1):67–79. https://doi.org/10.1128/JVI.01585-12
Söderberg-Nauclér C, Fish KN, Nelson JA (1997) Interferon-gamma and tumor necrosis factor-alpha specifically induce formation of cytomegalovirus-permissive monocyte-derived macrophages that are refractory to the antiviral activity of these cytokines. J Clin Investig 100(12):3154–3163. https://doi.org/10.1172/jci119871
Mao M, Wei X, Sheng H et al (2017) C-reactive protein/albumin and neutrophil/lymphocyte ratios and their combination predict overall survival in patients with gastric cancer. Oncol Lett 14(6):7417–7424. https://doi.org/10.3892/ol.2017.7179
Guo J, Chen S, Chen Y, Li S, Xu D (2018) Combination of CRP and NLR: a better predictor of postoperative survival in patients with gastric cancer. Cancer Manag Res 10:315–321. https://doi.org/10.2147/CMAR.S156071
Crimmins EM, Kim JK, Langa KM, Weir DR (2011) Assessment of cognition using surveys and neuropsychological assessment: the health and retirement study and the aging, demographics, and memory study. J Gerontol B Psychol Sci Soc Sci 66(Suppl 1):i162–i171. https://doi.org/10.1093/geronb/gbr048
Gianattasio KZ, Wu Q, Glymour MM, Power MC (2019) Comparison of methods for algorithmic classification of dementia status in the health and retirement study. Epidemiology 30(2):291–302. https://doi.org/10.1097/EDE.0000000000000945
Hurd MD, Martorell P, Delavande A, Mullen KJ, Langa KM (2013) Monetary costs of dementia in the United States. N Engl J Med 368(14):1326–1334. https://doi.org/10.1056/NEJMsa1204629
https://hrsdata.isr.umich.edu/sites/default/files/documentation/data-descriptions/HRS2016VBSDD.pdf.
Williams AM, Shah R, Shayne M et al (2018) Associations between inflammatory markers and cognitive function in breast cancer patients receiving chemotherapy. J Neuroimmunol 314:17–23. https://doi.org/10.1016/j.jneuroim.2017.10.005
Pusztai L, Mendoza TR, Reuben JM et al (2004) Changes in plasma levels of inflammatory cytokines in response to paclitaxel chemotherapy. Cytokine 25(3):94–102. https://doi.org/10.1016/j.cyto.2003.10.004
Pomykala KL, Ganz PA, Bower JE et al (2013) The association between pro-inflammatory cytokines, regional cerebral metabolism, and cognitive complaints following adjuvant chemotherapy for breast cancer. Brain Imaging Behav 7(4):511–523. https://doi.org/10.1007/s11682-013-9243-2
Kuo CP, Wu CL, Ho HT, Chen CG, Liu SI, Lu YT (2008) Detection of cytomegalovirus reactivation in cancer patients receiving chemotherapy. Clin Microbiol Infec 14(3):221–227. https://doi.org/10.1111/j.1469-0691.2007.01895.x
Azanan MS, Abdullah NK, Chua LL et al (2016) Immunity in young adult survivors of childhood leukemia is similar to the elderly rather than age-matched controls: Role of cytomegalovirus. Eur J Immunol 46(7):1715–1726. https://doi.org/10.1002/eji.201646356
Dik MG, Jonker C, Hack CE, Smit JH, Comijs HC, Eikelenboom P (2005) Serum inflammatory proteins and cognitive decline in older persons. Neurology 64(8):1371–1377. https://doi.org/10.1212/01.Wnl.0000158281.08946.68
Ravaglia G, Forti P, Maioli F et al (2007) Blood inflammatory markers and risk of dementia: the conselice study of brain aging. Neurobiol Aging 28(12):1810–1820. https://doi.org/10.1016/j.neurobiolaging.2006.08.012
Sundelöf J, Kilander L, Helmersson J et al (2009) Systemic inflammation and the risk of Alzheimer’s disease and dementia: a prospective population-based study. J Alzheimers Dis 18(1):79–87. https://doi.org/10.3233/jad-2009-1126
Teunissen CE, van Boxtel MP, Bosma H et al (2003) Inflammation markers in relation to cognition in a healthy aging population. J Neuroimmunol 134(1–2):142–150. https://doi.org/10.1016/s0165-5728(02)00398-3
Jenny NS, French B, Arnold AM et al (2012) Long-term assessment of inflammation and healthy aging in late life: the cardiovascular health study all stars. J Gerontol A Biol Sci Med Sci 67(9):970–976. https://doi.org/10.1093/gerona/glr261
Metti AL, Yaffe K, Boudreau RM et al (2014) Change in inflammatory markers and cognitive status in the oldest-old women from the Study of Osteoporotic Fractures. J Am Geriatr Soc 62(4):662–666. https://doi.org/10.1111/jgs.12739
Li H, Weng P, Najarro K et al (2014) Chronic CMV infection in older women: longitudinal comparisons of CMV DNA in peripheral monocytes, anti-CMV IgG titers, serum IL-6 levels, and CMV pp65 (NLV)-specific CD8(+) T-cell frequencies with twelve year follow-up. Exp Gerontol 54:84–89. https://doi.org/10.1016/j.exger.2014.01.010
Di Benedetto S, Gaetjen M, Muller L (2019) The modulatory effect of gender and cytomegalovirus-seropositivity on circulating inflammatory factors and cognitive performance in elderly individuals. Inter J Mol Sci. https://doi.org/10.3390/ijms20040990
Okedele OO, Nelson HH, Oyenuga ML, Thyagarajan B, Prizment A (2020) Cytomegalovirus and cancer-related mortality in the national health and nutritional examination survey. Cancer Causes Control 31(6):541–547. https://doi.org/10.1007/s10552-020-01296-y
Funding
This work was supported by the National Institute on Aging (R01 AG AG060110). The Health and Retirement Study is supported by NIA U01 AG009740.
Author information
Authors and Affiliations
Contributions
SV, BT and HHN designed the study. SV completed the statistical analysis and wrote the manuscript. BT developed the hypothesis, oversaw statistical analysis and manuscript preparation. HHN also provided critical feedback to the analysis strategy and comments to the manuscript. AEP, JF and EC reviewed the manuscript and suggested specific analysis for this project.
Corresponding author
Ethics declarations
Conflict of interest
There is no competing financial or non-financial interests in relation to this manuscript. The authors declare no conflict of interest.
Ethical approval
All study participants in HRS were consented for study participation and the study was approved by the Institutional Review Board at the University of Michigan, Ann Arbor.
Consent for publication
There is no individually identifiable data in this manuscript. All HRS participants consented to study participation and to use their data and samples for additional analysis.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Vivek, S., Nelson, H.H., Prizment, A.E. et al. Cross sectional association between cytomegalovirus seropositivity, inflammation and cognitive impairment in elderly cancer survivors. Cancer Causes Control 33, 81–90 (2022). https://doi.org/10.1007/s10552-021-01504-3
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10552-021-01504-3