Abstract
Mitochondrial metabolism and function are modulated by changes in matrix Ca2+. Small increases in the matrix Ca2+ stimulate mitochondrial bioenergetics, whereas excessive Ca2+ leads to cell death by causing massive matrix swelling and impairing the structural and functional integrity of mitochondria. Sustained opening of the non-selective mitochondrial permeability transition pores (PTP) is the main mechanism responsible for mitochondrial Ca2+ overload that leads to mitochondrial dysfunction and cell death. Recent studies suggest the existence of two or more types of PTP, and adenine nucleotide translocator (ANT) and FOF1-ATP synthase were proposed to form the PTP independent of each other. Here, we elucidated the role of ANT in PTP opening by applying both experimental and computational approaches. We first developed and corroborated a detailed model of the ANT transport mechanism including the matrix (ANTM), cytosolic (ANTC), and pore (ANTP) states of the transporter. Then, the ANT model was incorporated into a simple, yet effective, empirical model of mitochondrial bioenergetics to ascertain the point when Ca2+ overload initiates PTP opening via an ANT switch-like mechanism activated by matrix Ca2+ and is inhibited by extra-mitochondrial ADP. We found that encoding a heterogeneous Ca2+ response of at least three types of PTPs, weakly, moderately, and strongly sensitive to Ca2+, enabled the model to simulate Ca2+ release dynamics observed after large boluses were administered to a population of energized cardiac mitochondria. Thus, this study demonstrates the potential role of ANT in PTP gating and proposes a novel mechanism governing the cryptic nature of the PTP phenomenon.
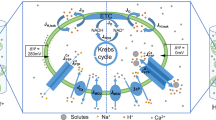
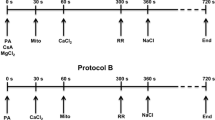
Graphical abstract







Similar content being viewed by others
Data availability
The datasets generated during and/or analyzed during the current study are available from the corresponding author on reasonable request. Modeling codes are available in Supplementary Information.
Abbreviations
- ANT:
-
Adenine nucleotide translocator
- ANTC :
-
C-state, ANT cytosolic state
- ANTM :
-
M-state, ANT matrix state
- ANTP :
-
P-state, ANT pore state
- CRC:
-
Calcium retention capacity
- ETC:
-
Electron transport chain
- IMM:
-
Inner mitochondrial membrane;
- MCU:
-
Mitochondrial Ca2+ uniporter
- OPA1:
-
Optic atrophy 1
- PTP:
-
Permeability transition pores
- PTPm :
-
Moderately stimulated PTP
- PTPs :
-
Strongly stimulated PTP
- PTPw :
-
Weakly stimulated PTP
- ΔΨ m :
-
Mitochondrial membrane potential
References
Alvarez-Illera P, Garcia-Casas P, Fonteriz RI, Montero M, and Alvarez J. Mitochondrial Ca(2+) dynamics in MCU knockout C. elegans Worms. Int J Mol Sci. 2020 21 https://doi.org/10.3390/ijms21228622.
Austin S, Mekis R, Mohammed SEM, Scalise M, Pfeiffer C, Galluccio M, Borovec T, et al. MICS1 is the Ca2+/H+ antiporter of mammalian mitochondria. bioRxiv. 2021;https://doi.org/10.1101/2021.11.11.468204:2021.2011.2011.468204, https://doi.org/10.1101/2021.11.11.468204.
Barbour RL, Chan SH. Characterization of the kinetics and mechanism of the mitochondrial ADP-atp carrier. J Biol Chem. 1981;256:1940–8.
Bazil JN, Buzzard GT, Rundell AE. Modeling mitochondrial bioenergetics with integrated volume dynamics. PLoS Comput Biol. 2010;6:e1000632. https://doi.org/10.1371/journal.pcbi.1000632.
Bazil JN, Blomeyer CA, Pradhan RK, Camara AK, Dash RK. Modeling the calcium sequestration system in isolated guinea pig cardiac mitochondria. J Bioenerg Biomembr. 2013;45:177–88. https://doi.org/10.1007/s10863-012-9488-2.
Bazil JN, Beard DA, Vinnakota KC. Catalytic coupling of oxidative phosphorylation, ATP demand, and reactive oxygen species generation. Biophys J. 2016;110:962–71. https://doi.org/10.1016/j.bpj.2015.09.036.
Bernardi P, Di Lisa F. The mitochondrial permeability transition pore: molecular nature and role as a target in cardioprotection. J Mol Cell Cardiol. 2015;78:100–6. https://doi.org/10.1016/j.yjmcc.2014.09.023.
Bernardi P, Carraro M, Lippe G. The mitochondrial permeability transition: recent progress and open questions. FEBS J. 2021. https://doi.org/10.1111/febs.16254,10.1111/febs.16254.
Bondarenko AI, Jean-Quartier C, Parichatikanond W, Alam MR, Waldeck-Weiermair M, Malli R, Graier WF. Mitochondrial Ca(2+) uniporter (MCU)-dependent and MCU-independent Ca(2+) channels coexist in the inner mitochondrial membrane. Pflugers Arch. 2014;466:1411–20. https://doi.org/10.1007/s00424-013-1383-0.
Brenner C, Subramaniam K, Pertuiset C, Pervaiz S. Adenine nucleotide translocase family: four isoforms for apoptosis modulation in cancer. Oncogene. 2011;30:883–95. https://doi.org/10.1038/onc.2010.501.
Bround MJ, Bers DM, Molkentin JD. A 20/20 view of ANT function in mitochondrial biology and necrotic cell death. J Mol Cell Cardiol. 2020;144:A3–13. https://doi.org/10.1016/j.yjmcc.2020.05.012.
Brustovetsky N, Tropschug M, Heimpel S, Heidkamper D, Klingenberg M. A large Ca2+-dependent channel formed by recombinant ADP/ATP carrier from Neurospora crassa resembles the mitochondrial permeability transition pore. Biochemistry. 2002;41:11804–11. https://doi.org/10.1021/bi0200110.
Brustovetsky N. The role of adenine nucleotide translocase in the mitochondrial permeability transition. Cells. 2020;9, https://doi.org/10.3390/cells9122686.
Buchanan BB, Eiermann W, Riccio P, Aquila H, Klingenberg M. Antibody evidence for different conformational states of ADP, ATP translocator protein isolated from mitochondria. Proc Natl Acad Sci U S A. 1976;73:2280–4. https://doi.org/10.1073/pnas.73.7.2280.
Carrer A, Tommasin L, Sileikyte J, Ciscato F, Filadi R, Urbani A, Forte M, et al. Defining the molecular mechanisms of the mitochondrial permeability transition through genetic manipulation of F-ATP synthase. Nat Commun. 2021;12:4835. https://doi.org/10.1038/s41467-021-25161-x.
Chapa-Dubocq X, Makarov V, Javadov S. Simple kinetic model of mitochondrial swelling in cardiac cells. J Cell Physiol. 2018;233:5310–21. https://doi.org/10.1002/jcp.26335.
Chapa-Dubocq XR, Rodriguez-Graciani KM, Guzman-Hernandez RA, Jang S, Brookes PS, and Javadov S. Cardiac function is not susceptible to moderate disassembly of mitochondrial respiratory supercomplexes. Int J Mol Sci. 2020;21, https://doi.org/10.3390/ijms21051555.
Chinopoulos C. Mitochondrial permeability transition pore: back to the drawing board. Neurochem Int. 2018;117:49–54. https://doi.org/10.1016/j.neuint.2017.06.010.
Chinopoulos C, Vajda S, Csanady L, Mandi M, Mathe K, Adam-Vizi V. A novel kinetic assay of mitochondrial ATP-ADP exchange rate mediated by the ANT. Biophys J. 2009;96:2490–504. https://doi.org/10.1016/j.bpj.2008.12.3915.
Davidson AM, Halestrap AP. Liver mitochondrial pyrophosphate concentration is increased by Ca2+ and regulates the intramitochondrial volume and adenine nucleotide content. Biochem J. 1987;246:715–23. https://doi.org/10.1042/bj2460715.
Duong QV, Hoffman A, Zhong K, Dessinger MJ, Zhang Y, Bazil JN. Calcium overload decreases net free radical emission in cardiac mitochondria. Mitochondrion. 2020;51:126–39. https://doi.org/10.1016/j.mito.2020.01.005.
Duyckaerts C, Sluse-Goffart CM, Fux JP, Sluse FE, Liebecq C. Kinetic mechanism of the exchanges catalysed by the adenine-nucleotide carrier. Eur J Biochem. 1980;106:1–6. https://doi.org/10.1111/j.1432-1033.1980.tb05990.x.
Garcia N, Martinez-Abundis E, Pavon N, Chavez E. On the opening of an insensitive cyclosporin A non-specific pore by phenylarsine plus mersalyl. Cell Biochem Biophys. 2007;49:84–90. https://doi.org/10.1007/s12013-007-0047-0.
Garlid KD. Cation transport in mitochondria–the potassium cycle. Biochim Biophys Acta. 1996;1275:123–6. https://doi.org/10.1016/0005-2728(96)00061-8.
Guan L, Che Z, Meng X, Yu Y, Li M, Yu Z, Shi H, et al. MCU up-regulation contributes to myocardial ischemia-reperfusion injury through calpain/OPA-1-mediated mitochondrial fusion/mitophagy inhibition. J Cell Mol Med. 2019;23:7830–43. https://doi.org/10.1111/jcmm.14662.
Halestrap AP. The regulation of the matrix volume of mammalian mitochondria in vivo and in vitro and its role in the control of mitochondrial metabolism. Biochim Biophys Acta. 1989;973:355–82. https://doi.org/10.1016/s0005-2728(89)80378-0.
Halestrap AP. A pore way to die: the role of mitochondria in reperfusion injury and cardioprotection. Biochem Soc Trans. 2010;38:841–60. https://doi.org/10.1042/BST0380841.
Halestrap AP, Brenner C. The adenine nucleotide translocase: a central component of the mitochondrial permeability transition pore and key player in cell death. Curr Med Chem. 2003;10:1507–25. https://doi.org/10.2174/0929867033457278.
Halestrap AP, Davidson AM. Inhibition of Ca2(+)-induced large-amplitude swelling of liver and heart mitochondria by cyclosporin is probably caused by the inhibitor binding to mitochondrial-matrix peptidyl-prolyl cis-trans isomerase and preventing it interacting with the adenine nucleotide translocase. Biochem J. 1990;268:153–60. https://doi.org/10.1042/bj2680153.
Halestrap AP, Woodfield KY, Connern CP. Oxidative stress, thiol reagents, and membrane potential modulate the mitochondrial permeability transition by affecting nucleotide binding to the adenine nucleotide translocase. J Biol Chem. 1997;272:3346–54. https://doi.org/10.1074/jbc.272.6.3346.
Halestrap AP, Kerr PM, Javadov S, Woodfield KY. Elucidating the molecular mechanism of the permeability transition pore and its role in reperfusion injury of the heart. Biochim Biophys Acta. 1998;1366:79–94. https://doi.org/10.1016/s0005-2728(98)00122-4.
Hamilton J, Brustovetsky T, Rysted JE, Lin Z, Usachev YM, Brustovetsky N. Deletion of mitochondrial calcium uniporter incompletely inhibits calcium uptake and induction of the permeability transition pore in brain mitochondria. J Biol Chem. 2018;293:15652–63. https://doi.org/10.1074/jbc.RA118.002926.
He L, Lemasters JJ. Regulated and unregulated mitochondrial permeability transition pores: a new paradigm of pore structure and function? FEBS Lett. 2002;512:1–7. https://doi.org/10.1016/s0014-5793(01)03314-2.
Hollander JM, Thapa D, Shepherd DL. Physiological and structural differences in spatially distinct subpopulations of cardiac mitochondria: influence of cardiac pathologies. Am J Physiol Heart Circ Physiol. 2014;307:H1-14. https://doi.org/10.1152/ajpheart.00747.2013.
Hunter DR, Haworth RA, Southard JH. Relationship between configuration, function, and permeability in calcium-treated mitochondria. J Biol Chem. 1976;251:5069–77.
Jang S, Javadov S. OPA1 regulates respiratory supercomplexes assembly: the role of mitochondrial swelling. Mitochondrion. 2020;51:30–9. https://doi.org/10.1016/j.mito.2019.11.006.
Javadov S, Jang S, Parodi-Rullan R, Khuchua Z, Kuznetsov AV. Mitochondrial permeability transition in cardiac ischemia-reperfusion: whether cyclophilin D is a viable target for cardioprotection? Cell Mol Life Sci. 2017;74:2795–813. https://doi.org/10.1007/s00018-017-2502-4.
Javadov S, Chapa-Dubocq X, Makarov V. Different approaches to modeling analysis of mitochondrial swelling. Mitochondrion. 2018;38:58–70. https://doi.org/10.1016/j.mito.2017.08.004.
Karch J, Bround MJ, Khalil H, Sargent MA, Latchman N, Terada N, Peixoto PM, et al. Inhibition of mitochondrial permeability transition by deletion of the ANT family and CypD. Sci Adv. 2019;5:eaaw4597. https://doi.org/10.1126/sciadv.aaw4597.
Korotkov SM, Konovalova SA, Brailovskaya IV, Saris NE. To involvement the conformation of the adenine nucleotide translocase in opening the Tl(+)-induced permeability transition pore in Ca(2+)-loaded rat liver mitochondria. Toxicol in Vitro. 2016;32:320–32. https://doi.org/10.1016/j.tiv.2016.01.015.
Kosmach A, Roman B, Sun J, Femnou A, Zhang F, Liu C, Combs CA, et al. Monitoring mitochondrial calcium and metabolism in the beating MCU-KO heart. Cell Rep. 2021;37:109846. https://doi.org/10.1016/j.celrep.2021.109846.
Kowaltowski AJ, Castilho RF. Ca2+ acting at the external side of the inner mitochondrial membrane can stimulate mitochondrial permeability transition induced by phenylarsine oxide. Biochim Biophys Acta. 1997;1322:221–9. https://doi.org/10.1016/s0005-2728(97)00078-9.
Kramer R, Klingenberg M. Electrophoretic control of reconstituted adenine nucleotide translocation. Biochemistry. 1982;21:1082–9. https://doi.org/10.1021/bi00534a040.
Kristian T, Weatherby TM, Bates TE, Fiskum G. Heterogeneity of the calcium-induced permeability transition in isolated non-synaptic brain mitochondria. J Neurochem. 2002;83:1297–308. https://doi.org/10.1046/j.1471-4159.2002.01238.x.
Kunji ERS, Ruprecht JJ. The mitochondrial ADP/ATP carrier exists and functions as a monomer. Biochem Soc Trans. 2020;48:1419–32. https://doi.org/10.1042/BST20190933.
Kushnareva YE, Wiley SE, Ward MW, Andreyev AY, Murphy AN. Excitotoxic injury to mitochondria isolated from cultured neurons. J Biol Chem. 2005;280:28894–902. https://doi.org/10.1074/jbc.M503090200.
Kushnareva YE, Gerencser AA, Bossy B, Ju WK, White AD, Waggoner J, Ellisman MH, et al. Loss of OPA1 disturbs cellular calcium homeostasis and sensitizes for excitotoxicity. Cell Death Differ. 2013;20:353–65. https://doi.org/10.1038/cdd.2012.128.
Leung AW, Varanyuwatana P, Halestrap AP. The mitochondrial phosphate carrier interacts with cyclophilin D and may play a key role in the permeability transition. J Biol Chem. 2008;283:26312–23. https://doi.org/10.1074/jbc.M805235200.
Litsky ML, Pfeiffer DR. Regulation of the mitochondrial Ca2+ uniporter by external adenine nucleotides: the uniporter behaves like a gated channel which is regulated by nucleotides and divalent cations. Biochemistry. 1997;36:7071–80. https://doi.org/10.1021/bi970180y.
Liu G, Wang ZK, Wang ZY, Yang DB, Liu ZP, Wang L. Mitochondrial permeability transition and its regulatory components are implicated in apoptosis of primary cultures of rat proximal tubular cells exposed to lead. Arch Toxicol. 2016;90:1193–209. https://doi.org/10.1007/s00204-015-1547-0.
Lu X, Thai PN, Lu S, Pu J, Bers DM. Intrafibrillar and perinuclear mitochondrial heterogeneity in adult cardiac myocytes. J Mol Cell Cardiol. 2019;136:72–84. https://doi.org/10.1016/j.yjmcc.2019.08.013.
Malyala S, Zhang Y, Strubbe JO, Bazil JN. Calcium phosphate precipitation inhibits mitochondrial energy metabolism. PLoS Comput Biol. 2019;15:e1006719. https://doi.org/10.1371/journal.pcbi.1006719.
Metelkin E, Goryanin I, Demin O. Mathematical modeling of mitochondrial adenine nucleotide translocase. Biophys J. 2006;90:423–32. https://doi.org/10.1529/biophysj.105.061986.
Mielke C, Lefort N, McLean CG, Cordova JM, Langlais PR, Bordner AJ, Te JA, et al. Adenine nucleotide translocase is acetylated in vivo in human muscle: modeling predicts a decreased ADP affinity and altered control of oxidative phosphorylation. Biochemistry. 2014;53:3817–29. https://doi.org/10.1021/bi401651e.
Morciano G, Naumova N, Koprowski P, Valente S, Sardao VA, Potes Y, Rimessi A, et al. The mitochondrial permeability transition pore: an evolving concept critical for cell life and death. Biol Rev Camb Philos Soc. 2021. https://doi.org/10.1111/brv.12764,10.1111/brv.12764.
Nguyen MH, Jafri MS. Mitochondrial calcium signaling and energy metabolism. Ann N Y Acad Sci. 2005;1047:127–37. https://doi.org/10.1196/annals.1341.012.
Pandya JD, Nukala VN, Sullivan PG. Concentration dependent effect of calcium on brain mitochondrial bioenergetics and oxidative stress parameters. Front Neuroenergetics. 2013;5:10. https://doi.org/10.3389/fnene.2013.00010.
Pradhan RK, Qi F, Beard DA, Dash RK. Characterization of Mg2+ inhibition of mitochondrial Ca2+ uptake by a mechanistic model of mitochondrial Ca2+ uniporter. Biophys J. 2011;101:2071–81. https://doi.org/10.1016/j.bpj.2011.09.029.
Rodriguez-Graciani KM, Chapa-Dubocq XR, MacMillan-Crow LA, Javadov S. Association between L-OPA1 cleavage and cardiac dysfunction during ischemia-reperfusion injury in rats. Cell Physiol Biochem. 2020;54:1101–14. https://doi.org/10.33594/000000303.
Rossi A, Pizzo P, Filadi R. Calcium, mitochondria and cell metabolism: a functional triangle in bioenergetics. Biochim Biophys Acta Mol Cell Res. 2019;1866:1068–78. https://doi.org/10.1016/j.bbamcr.2018.10.016.
Squire W, Trapp G. Using complex variables to estimate derivatives of real functions. SIAM Rev. 1998;40:110–2. https://doi.org/10.1137/s003614459631241x.
Strubbe-Rivera JO, Schrad JR, Pavlov EV, Conway JF, Parent KN, Bazil JN. The mitochondrial permeability transition phenomenon elucidated by cryo-EM reveals the genuine impact of calcium overload on mitochondrial structure and function. Sci Rep. 2021;11:1037. https://doi.org/10.1038/s41598-020-80398-8.
Strubbe-Rivera JO, Chen J, West BA, Parent KN, Wei GW, and Bazil JN. Modeling the effects of calcium overload on mitochondrial ultrastructural remodeling. Appl Sci (Basel). 2021b 11, https://doi.org/10.3390/app11052071.
Vinnakota KC, Bazil JN, Van den Bergh F, Wiseman RW, Beard DA. Feedback regulation and time hierarchy of oxidative phosphorylation in cardiac mitochondria. Biophys J. 2016;110:972–80. https://doi.org/10.1016/j.bpj.2016.01.003.
Wacquier B, Combettes L, Dupont G. Dual dynamics of mitochondrial permeability transition pore opening. Sci Rep. 2020;10:3924. https://doi.org/10.1038/s41598-020-60177-1.
Woodfield K, Ruck A, Brdiczka D, Halestrap AP. Direct demonstration of a specific interaction between cyclophilin-D and the adenine nucleotide translocase confirms their role in the mitochondrial permeability transition. Biochem J. 1998;336(Pt 2):287–90. https://doi.org/10.1042/bj3360287.
Wu F, Yang F, Vinnakota KC, Beard DA. Computer modeling of mitochondrial tricarboxylic acid cycle, oxidative phosphorylation, metabolite transport, and electrophysiology. J Biol Chem. 2007;282:24525–37. https://doi.org/10.1074/jbc.M701024200.
Wu F, Zhang J, Beard DA. Experimentally observed phenomena on cardiac energetics in heart failure emerge from simulations of cardiac metabolism. Proc Natl Acad Sci U S A. 2009;106:7143–8. https://doi.org/10.1073/pnas.0812768106.
Zoratti M, Szabo I. The mitochondrial permeability transition. Biochim Biophys Acta. 1995;1241:139–76. https://doi.org/10.1016/0304-4157(95)00003-a.
Zydowo MM, Swierczynski J, Nagel G, Wrzolkowa T. The respiration and calcium content of heart mitochondria from rats with vitamin D-induced cardionecrosis. Biochem J. 1985;226:155–61. https://doi.org/10.1042/bj2260155.
Funding
This study was supported by the National Science Foundation (Award 2006477 to S.J.) and the National Institutes of Health (Grants R25GM061838 and U54MD007600).
Author information
Authors and Affiliations
Contributions
SJ and JNB conceived and designed the study. XRC-D, JG-B, and JNB performed experiments and interpreted the results. Modeling analysis was performed by XRC-D and JNB. XRC-D and SJ wrote the first draft of the manuscript and all authors commented on the manuscript. SJ supervised the project and is responsible for its integrity. All authors read and approved the final manuscript.
Corresponding author
Ethics declarations
Ethics approval
The manuscript does not contain clinical studies or patient data.
Conflict of interest
The authors declare no competing interests.
Additional information
Publisher's note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Headlights
• We developed a detailed biophysical model of ANT kinetics that was verified using ATP/ADP exchange rates for parameter estimation.
• The model was successful in simulating ANT pore state transition and clarified a vital role of ANT in Ca2 + -induced PTP opening.
• The model provided insight into the nature of PTP gating in the presence of respiratory substrates and ADP.
Supplementary Information
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Chapa-Dubocq, X.R., Garcia-Baez, J.F., Bazil, J.N. et al. Crosstalk between adenine nucleotide transporter and mitochondrial swelling: experimental and computational approaches. Cell Biol Toxicol 39, 435–450 (2023). https://doi.org/10.1007/s10565-022-09724-2
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10565-022-09724-2