Abstract
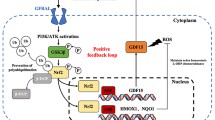
Gemcitabine resistance is a frequently occurring and intractable obstacle in pancreatic cancer treatment. However, the underlying mechanisms require further investigation. Adaptive regulation of oxidative stress and aberrant activation of the NF-κB signaling pathway are associated with resistance to chemotherapy. Here, we found that gemcitabine upregulated the expression of CASC9 in a dose-dependent manner, partially via induction of reactive oxygen species, whereas inhibition of CASC9 expression enhanced gemcitabine-induced oxidative stress and apoptosis in pancreatic cancer cells. Furthermore, suppression of CASC9 level inhibited the expression of NRF2 and the downstream genes NQO1 and HO-1, and vice versa, indicating that CASC9 forms a positive feedback loop with NRF2 signaling and modulates the level of oxidative stress. Silencing CASC9 attenuated NF-κB pathway activation in pancreatic cancer cells and synergistically enhanced the cytotoxic effect of gemcitabine chemotherapy in vivo. In conclusion, our findings suggest that CASC9 plays a key role in driving resistance to gemcitabine through a reciprocal loop with the NRF2-antioxidant signaling pathway and by activating NF-κB signaling. Our study reveals potential targets that can effectively reverse resistance to gemcitabine chemotherapy.







Similar content being viewed by others
Data availability
The datasets presented in the current study are available from the corresponding author on reasonable request.
Abbreviations
- CASC9:
-
LncRNA cancer susceptibility candidate 9
- GEM:
-
Gemcitabine
- NF-κB:
-
Nuclear factor-kappa B
- LncRNAs:
-
Long noncoding RNAs
- NAC:
-
N-acetyl-L-cysteine
- EMSA:
-
Electrophoretic mobility shift assay
- HO-1:
-
Heme oxygenase-1
- NQO1:
-
NAD(P)H quinone oxidoreductase 1
- NRF2:
-
Nuclear factor erythroid 2-related factor 2
- ROS:
-
Reactive oxygen species
References
Abdel Hadi N, Reyes-Castellanos G, Carrier A. Targeting redox metabolism in pancreatic cancer. Int J Mol Sci. 2021;22(4).
Arlt A, Gehrz A, Muerkoster S, Vorndamm J, Kruse ML, Folsch UR, et al. Role of NF-kappaB and Akt/PI3K in the resistance of pancreatic carcinoma cell lines against gemcitabine-induced cell death. Oncogene. 2003;22(21):3243–51.
Arlt A, Schafer H, Kalthoff H. The “N-factors” in pancreatic cancer: functional relevance of NF-kappaB, NFAT and Nrf2 in pancreatic cancer. Oncogenesis. 2012;1:e35.
Arora S, Bhardwaj A, Singh S, Srivastava SK, McClellan S, Nirodi CS, et al. An undesired effect of chemotherapy: gemcitabine promotes pancreatic cancer cell invasiveness through reactive oxygen species-dependent, nuclear factor kappaB- and hypoxia-inducible factor 1alpha-mediated up-regulation of CXCR4. J Biol Chem. 2013;288(29):21197–207.
Berlin J, Benson AB 3rd. Chemotherapy: gemcitabine remains the standard of care for pancreatic cancer. Nat Rev Clin Oncol. 2010;7(3):135–7.
Binenbaum Y, Na’ara S, Gil Z. Gemcitabine resistance in pancreatic ductal adenocarcinoma. Drug Resist Updat. 2015;23:55–68.
Chang J, Zhang Y, Ye X, Guo H, Lu K, Liu Q, et al. Long non-coding RNA (LncRNA) CASC9/microRNA(miR)-590-3p/sine oculis homeobox 1 (SIX1)/NF-kappaB axis promotes proliferation and migration in breast cancer. Bioengineered. 2021;12(1):8709–23.
Gibb EA, Brown CJ, Lam WL. The functional role of long non-coding RNA in human carcinomas. Mol Cancer. 2011;10:38.
Guan C, Wang Y. LncRNA CASC9 attenuates lactate dehydrogenase-mediated oxidative stress and inflammation in spinal cord injury via sponging miR-383-5p. Inflammation. 2021;44(3):923–33.
Ju HQ, Gocho T, Aguilar M, Wu M, Zhuang ZN, Fu J, et al. Mechanisms of overcoming intrinsic resistance to gemcitabine in pancreatic ductal adenocarcinoma through the redox modulation. Mol Cancer Ther. 2015;14(3):788–98.
Kabacaoglu D, Ciecielski KJ, Ruess DA, Algul H. Immune checkpoint inhibition for pancreatic ductal adenocarcinoma: current limitations and future options. Front Immunol. 2018;9:1878.
Leinonen HM, Kansanen E, Polonen P, Heinaniemi M, Levonen AL. Role of the Keap1-Nrf2 pathway in cancer. Adv Cancer Res. 2014;122:281–320.
Li D, He J, Qian X, Xu P, Wang X, Li Z, et al. The involvement of lncRNAs in the development and progression of pancreatic cancer. Cancer Biol Ther. 2017;18(12):927–36.
Li Q, Yang G, Feng M, Zheng S, Cao Z, Qiu J, et al. NF-kappaB in pancreatic cancer: Its key role in chemoresistance. Cancer Lett. 2018;421:127–34.
Li Y, Yang X, Kang X, Liu S. The regulatory roles of long noncoding RNAs in the biological behavior of pancreatic cancer. Saudi J Gastroenterol. 2019;25(3):145–51.
Liang Y, Chen X, Wu Y, Li J, Zhang S, Wang K, et al. LncRNA CASC9 promotes esophageal squamous cell carcinoma metastasis through upregulating LAMC2 expression by interacting with the CREB-binding protein. Cell Death Differ. 2018;25(11):1980–95.
Liptay S, Weber CK, Ludwig L, Wagner M, Adler G, Schmid RM. Mitogenic and antiapoptotic role of constitutive NF-kappaB/Rel activity in pancreatic cancer. Int J Cancer. 2003;105(6):735–46.
Lister A, Nedjadi T, Kitteringham NR, Campbell F, Costello E, Lloyd B, et al. Nrf2 is overexpressed in pancreatic cancer: implications for cell proliferation and therapy. Mol Cancer. 2011;10:37.
Luo K, Geng J, Zhang Q, Xu Y, Zhou X, Huang Z, et al. LncRNA CASC9 interacts with CPSF3 to regulate TGF-beta signaling in colorectal cancer. J Exp Clin Cancer Res. 2019;38(1):249.
Matsumoto R, Hamada S, Tanaka Y, Taguchi K, Yamamoto M, Masamune A. Nuclear factor erythroid 2-related factor 2 depletion sensitizes pancreatic cancer cells to gemcitabine via aldehyde dehydrogenase 3a1 repression. J Pharmacol Exp Ther. 2021;379(1):33–40.
Pramanik KC, Makena MR, Bhowmick K, Pandey MK. Advancement of NF-kappaB signaling pathway: a novel target in pancreatic cancer. Int J Mol Sci. 2018;19(12).
Rojo de la Vega M, Chapman E, Zhang DD. NRF2 and the hallmarks of cancer. Cancer Cell. 2018;34(1):21–43.
Sarvepalli D, Rashid MU, Rahman AU, Ullah W, Hussain I, Hasan B, et al. Gemcitabine: a review of chemoresistance in pancreatic cancer. Crit Rev Oncog. 2019;24(2):199–212.
Schmidlin CJ, Dodson MB, Madhavan L, Zhang DD. Redox regulation by NRF2 in aging and disease. Free Radic Biol Med. 2019;134:702–7.
Siegel RL, Miller KD, Jemal A. Cancer statistics, 2019. CA Cancer J Clin. 2019;69(1):7–34.
Silvestris N, Gnoni A, Brunetti AE, Vincenti L, Santini D, Tonini G, et al. Target therapies in pancreatic carcinoma. Curr Med Chem. 2014;21(8):948–65.
Soini Y, Eskelinen M, Juvonen P, Karja V, Haapasaari KM, Saarela A, et al. Nuclear Nrf2 expression is related to a poor survival in pancreatic adenocarcinoma. Pathol Res Pract. 2014;210(1):35–9.
Trachootham D, Lu W, Ogasawara MA, Nilsa RD, Huang P. Redox regulation of cell survival. Antioxid Redox Signal. 2008;10(8):1343–74.
Ushio-Fukai M, Nakamura Y. Reactive oxygen species and angiogenesis: NADPH oxidase as target for cancer therapy. Cancer Lett. 2008;266(1):37–52.
Wakabayashi N, Slocum SL, Skoko JJ, Shin S, Kensler TW. When NRF2 talks, who’s listening? Antioxid Redox Signal. 2010;13(11):1649–63.
Xiang Y, Ye W, Huang C, Yu D, Chen H, Deng T, et al. Brusatol enhances the chemotherapy efficacy of gemcitabine in pancreatic cancer via the Nrf2 signalling pathway. Oxid Med Cell Longev. 2018;2018:2360427.
Xie W, Chu M, Song G, Zuo Z, Han Z, Chen C, et al. Emerging roles of long noncoding RNAs in chemoresistance of pancreatic cancer. Semin Cancer Biol. 2020.
Xiong G, Feng M, Yang G, Zheng S, Song X, Cao Z, et al. The underlying mechanisms of non-coding RNAs in the chemoresistance of pancreatic cancer. Cancer Lett. 2017;397:94–102.
Yang G, Guan W, Cao Z, Guo W, Xiong G, Zhao F, et al. Integrative genomic analysis of gemcitabine resistance in pancreatic cancer by patient-derived xenograft models. Clin Cancer Res. 2021.
Yu X, Lin Y, Sui W, Zou Y, Lv Z. Analysis of distinct long noncoding RNA transcriptional fingerprints in pancreatic ductal adenocarcinoma. Cancer Med. 2017;6(3):673–80.
Yue J, Wu Y, Qiu L, Zhao R, Jiang M, Zhang H. LncRNAs link cancer stemness to therapy resistance. Am J Cancer Res. 2021;11(4):1051–68.
Zhang L, Li J, Zong L, Chen X, Chen K, Jiang Z, et al. Reactive oxygen species and targeted therapy for pancreatic cancer. Oxid Med Cell Longev. 2016;2016:1616781.
Zhang Y, Du Y, Le W, Wang K, Kieffer N, Zhang J. Redox control of the survival of healthy and diseased cells. Antioxid Redox Signal. 2011;15(11):2867–908.
Zhang Z, Duan Q, Zhao H, Liu T, Wu H, Shen Q, et al. Gemcitabine treatment promotes pancreatic cancer stemness through the Nox/ROS/NF-kappaB/STAT3 signaling cascade. Cancer Lett. 2016;382(1):53–63.
Funding
The study was supported by the National Natural Science Foundation of China (grant number: 81803030), and National Natural Science Foundation of China (grant number: 82172570).
Author information
Authors and Affiliations
Contributions
Hanjun Li and Jing Tao were responsible for the experimental concept and research supervision. Zhengle Zhang, Longjiang Chen, and Chuanbing Zhao carried out the main research, analyzed the data, and wrote the paper. Qiong Gong participated in data analysis. Zhigang Tang performed revising the manuscript. All authors reviewed and approved the final version of manuscript.
Corresponding authors
Ethics declarations
Ethical approval
The study was approved by the ethical review board of Renmin Hospital of Wuhan University (Wuhan, China).
Competing interests
The authors declare no competing interests.
Additional information
Publisher's note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Zhang, Z., Chen, L., Zhao, C. et al. CASC9 potentiates gemcitabine resistance in pancreatic cancer by reciprocally activating NRF2 and the NF-κB signaling pathway. Cell Biol Toxicol 39, 1549–1560 (2023). https://doi.org/10.1007/s10565-022-09746-w
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10565-022-09746-w