Abstract
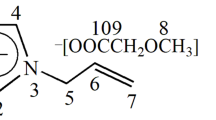
Organic solvents with low Henry's constants, such as propylene carbonate (PC) and sulfolane, were proposed to be used as CO2 absorbents to increase CO2 absorption capacity in the CO2 switchable solvent, so that the dissolution of cellulose can be effectively enhanced. With the addition of PC, the maximum concentration achieved for the dissolution of paper cellulose can be largely increased from 1.0 to 5.0 wt%, while those for microcrystalline cellulose and corncob cellulose were increased from 5.0 to 10.0 and 7.0 wt%, respectively. Moreover, the chemical and crystalline structure of the regenerated cellulose was not affected by the addition of CO2 absorbents. This study illustrated a simple and effective way to modify the CO2 switchable solvent, which would be helpful for the application of the complex solvent system for the enhanced dissolution, derivatization, and processing of cellulose in the future.








Similar content being viewed by others
Data availability
All data generated or analyzed during this study are included in this published article.
References
AlfredMichael DF et al (2013) Cellulose polymorphy, crystallite size, and the Segal Crystallinity Index. Cellulose 20:583–588. https://doi.org/10.1007/s10570-012-9833-y
Andanson JM, Bordes E, Devémy J et al (2014) Understanding the role of co-solvents in the dissolution of cellulose in ionic liquids. Green Chem 16:2528–2538. https://doi.org/10.1039/c3gc42244e
Bao X, Wang C, Zhang Z et al (2021) Wet spinning to prepare filaments from three cellulose carbonated derivatives: Synthesis, characterization and filament properties. Carbohydr Polym Technol Appl 2:100099. https://doi.org/10.1016/j.carpta.2021.100099
Borhani TN, Wang M (2019) Role of solvents in CO2 capture processes: the review of selection and design methods. Renew Sustain Energy Rev 114:109299. https://doi.org/10.1016/j.rser.2019.109299
Cai J, Zhang L (2005) Rapid dissolution of cellulose in LiOH/urea and NaOH/urea aqueous solutions. Macromol Biosci 5:539–548. https://doi.org/10.1002/mabi.200400222
Cai J, Zhang L (2006) Unique gelation behavior of cellulose in NaOH/urea aqueous solution. Biomacromol 7:183–189. https://doi.org/10.1021/bm0505585
Cai J, Zhang L, Zhou J et al (2004) Novel fibers prepared from cellulose in NaOH/urea aqueous solution. Macromol Rapid Commun 25:1558–1562. https://doi.org/10.1002/marc.200400172
Chen H, Yang F, Du J et al (2018) Efficient transesterification reaction of cellulose with vinyl esters in DBU/DMSO/CO2 solvent system at low temperature. Cellulose 25:6935–6945. https://doi.org/10.1007/s10570-018-2078-7
Cho HM, Gross AS, Chu JW (2011) Dissecting force interactions in cellulose deconstruction reveals the required solvent versatility for overcoming biomass recalcitrance. J Am Chem Soc 133:14033–14041. https://doi.org/10.1021/ja2046155
Fink HP, Weigel P, Purz HJ, Ganster J (2001) Structure formation of regenerated cellulose materials from NMMO-solutions. Prog Polym Sci 26:1473–1524. https://doi.org/10.1016/S0079-6700(01)00025-9
Gan J, Peng Y, Chen Q et al (2020) Reversible covalent chemistry of carbon dioxide unlocks the recalcitrance of cellulose for its enzymatic saccharification. Bioresour Technol 295:122230. https://doi.org/10.1016/j.biortech.2019.122230
Guo Y, Cai L, Guo G et al (2021a) Cellulose membranes from cellulose CO2—Based reversible ionic liquid solutions. ACS Sustain Chem Eng. https://doi.org/10.1021/acssuschemeng.1c03609
Guo Y, Li L, Guo G et al (2021b) Synthesis of a fully biobased cellulose-3-(2-hydroxyphenyl) propionate ester with antioxidant activity and UV-resistant properties by the DBU/CO2/DMSO solvent system. Green Chem 23:2352–2361. https://doi.org/10.1039/d0gc03478a
Halliday C, Hatton TA (2021) Sorbents for the capture of CO2 and other acid gases : a review. Ind Eng Chem Res 60:9313–9346. https://doi.org/10.1021/acs.iecr.1c00597
Heinze T, Dicke R, Koschella A et al (2000) Effective preparation of cellulose derivatives in a new simple cellulose solvent. Macromol Chem Phys 201:627–631. https://doi.org/10.1002/(SICI)1521-3935(20000301)201:6%3c627::AID-MACP627%3e3.0.CO;2-Y
Heldebrant DJ, Yonker CR, Jessop PG, Phan L (2008) Organic liquid CO2 capture agents with high gravimetric CO2 capacity. Energy Environ Sci 1:487–493. https://doi.org/10.1039/b809533g
Higgins HG, Stewart CM, Harrington KJ (1961) Infrared spectra of cellulose and related polysaccharides. J Polym Sci 51:59–84. https://doi.org/10.1002/pol.1961.1205115505
Hua L, Wanren C (2005) Solubility of dilute SO2 and CO2 in dimethyl sulfoxide. Phys Chem Liq 43:289–298. https://doi.org/10.1080/00319100500083583
Huang K, Chen Y Le, Zhang XM, et al (2014) Experimental study and thermodynamical modelling of the solubilities of SO2, H2S and CO2 in N-dodecylimidazole and 1,1’-[oxybis(2,1-ethanediyloxy-2,1-ethanediyl)]bis(imidazole): an evaluation of their potential application in the separation of acidic gase. Elsevier B.V. https://doi.org/10.1016/j.fluid.2014.06.026
Idström A, Schantz S, Sundberg J et al (2016) 13C NMR assignments of regenerated cellulose from solid-state 2D NMR spectroscopy. Carbohydr Polym 151:480–487. https://doi.org/10.1016/j.carbpol.2016.05.107
Jalili AH, Shokouhi M, Samani F, Hosseini-jenab M (2015) Measuring the solubility of CO2 and H2S in sulfolane and the density and viscosity of saturated liquid binary mixtures of (sulfolane + CO2) and (sulfolane + H2S) Amir. J Chem Thermodyn. https://doi.org/10.1016/j.jct.2015.01.001
Jessop PG, Heldebrant DJ, Li X et al (2005) Virology: independent virus development outside a host. Nature 436:1101–1102. https://doi.org/10.1038/nature4361101a
Jin L, Gan J, Hu G et al (2019) Preparation of cellulose films from sustainable CO2/DBU/DMSO system. Polymers (baseL). https://doi.org/10.3390/polym11060994
Kataoka Y, Kondo T (1998) FT-IR microscopic analysis of changing cellulose crystalline structure during wood cell wall formation. Macromolecules 31:760–764. https://doi.org/10.1021/ma970768c
Kirchberg A, Meier MAR (2021) Regeneration of cellulose from a switchable ionic liquid: toward more sustainable cellulose fibers. Macromol Chem Phys 222:2000433. https://doi.org/10.1002/macp.202000433
Klemm D, Heublein B, Fink HP, Bohn A (2005) Cellulose: fascinating biopolymer and sustainable raw material. Angew Chemie - Int Ed 44:3358–3393. https://doi.org/10.1002/anie.200460587
Lenoir JY, Renault P, Renon H (1971) Gas chromatographic determination of Henry’s constants of 12 gases in 19 solvents. J Chem Eng Data 16:340–342. https://doi.org/10.1021/je60050a014
Li H, Liu D, Wang F (2002) Solubility of dilute SO2 in dimethyl sulfoxide. J Chem Eng Data 43:772–775. https://doi.org/10.1080/00319100500083583
Li J, Lu S, Liu F et al (2021) Structure and properties of regenerated cellulose fibers based on dissolution of cellulose in a CO2 switchable solvent. ACS Sustain Chem Eng 9:4744–4754. https://doi.org/10.1021/acssuschemeng.0c08907
Li X, Dong F, Zhang L et al (2019) Cellulosic protic ionic liquids hydrogel: a green and efficient catalyst carrier for Pd nanoparticles in reduction of 4-nitrophenol in water. Chem Eng J 372:516–525. https://doi.org/10.1016/j.cej.2019.04.123
Liu X, Pang J, Zhang X (2013) Regenerated cellulose film with enhanced tensile strength prepared with ionic liquid 1-ethyl-3-methylimidazolium acetate ( EMIMAc ). Cellulose. https://doi.org/10.1007/s10570-013-9925-3
Lu S, Li J, Liu F et al (2021) Impact of DBU on the synthesis of cellulose-graft-poly(l-lactide) copolymer in CO2 switchable solvent with different grafting strategies. Polymer (guildf). https://doi.org/10.1016/j.polymer.2021.124020
Mérièm A, Yvon RD, Cécile T, Loubna El O, Johan J (2012) Low pressure carbon dioxide solubility in pure electrolyte solvents for lithium-ion batteries as a function of temperature. measurement and prediction. J Chem Thermodyn J 50:71–79. https://doi.org/10.1016/j.jct.2012.01.027
Murrieta-Guevara F, Romero-Martinez A, Trejo A (1988) Solubilities of carbon dioxide and hydrogen sulfide in propylene carbonate, N-methylpyrrolidone and sulfolane. Fluid Phase Equilib 44:105–115. https://doi.org/10.1016/0378-3812(88)80106-7
Nanta P, Skolpap W, Kasemwong K, Shimoyama Y (2017) Dissolution and modification of cellulose using high-pressure carbon dioxide switchable solution. J Supercrit Fluids 130:84–90. https://doi.org/10.1016/j.supflu.2017.07.019
Onwukamike KN, Grelier S, Grau E et al (2018a) Sustainable transesterification of cellulose with high oleic sunflower oil in a DBU-CO2 switchable solvent. ACS Sustain Chem Eng. https://doi.org/10.1021/acssuschemeng.8b01186
Onwukamike KN, Lapuyade L, Maille L et al (2019) Sustainable approach for cellulose aerogel preparation from the DBU-CO2 switchable solvent. ACS Sustain Chem Eng. https://doi.org/10.1021/acssuschemeng.8b05427
Onwukamike KN, Tassaing T, Grelier S et al (2018b) Detailed understanding of the DBU/CO2 switchable solvent system for cellulose solubilization and derivatization. ACS Sustain Chem Eng 6:1496–1503. https://doi.org/10.1021/acssuschemeng.7b04053
Padhye MR, Deshpande MM (1998) J Appl Polymer Sci 36:1475–1478
Pei M, Peng X, Shen Y et al (2020) Synthesis of water-soluble, fully biobased cellulose levulinate esters through the reaction of cellulose and alpha-angelica lactone in a DBU/CO2/DMSO solvent system. Green Chem 22:707–717. https://doi.org/10.1039/c9gc03149a
Pinkert A, Marsh KN, Pang S (2010) Pang S (2010) Reflections on the Solubility of Cellulose. Ind Eng Chem Res 49:11121–11130. https://doi.org/10.1021/ie1006596
Rabek J (1980) Applications of wide-angle X-ray diffraction (WAXD) to the study of the structure of polymers. Wiley, Chichester, p 505
Song L, Yang Y, Xie H, Liu E (2015) Cellulose dissolution and in situ grafting in a reversible system using an organocatalyst and carbon dioxide. Chemsuschem 8:3217–3221. https://doi.org/10.1002/cssc.201500378
Strlič M, Kolenc J, Kolar J, Pihlar B (2002) Enthalpic interactions in size exclusion chromatography of pullulan and cellulose in LiCl - N, N-dimethylacetamide. J Chromatogr A 964:47–54. https://doi.org/10.1016/S0021-9673(02)00591-5
Sun H, Miao J, Yu Y (2015) Dissolution of cellulose with a novel solvent and formation of regenerated cellulose fiber. Mater Sci Process. https://doi.org/10.1007/s00339-015-8986-6
Sun J, Cheng W, Yang Z et al (2014) Superbase/cellulose: an environmentally benign catalyst for chemical fixation of carbon dioxide into cyclic carbonates. Green Chem 16:3071–3078. https://doi.org/10.1039/c3gc41850b
Wang H, Gurau G, Rogers RD (2012) Ionic liquid processing of cellulose. Chem Soc Rev 41:1519–1537. https://doi.org/10.1039/c2cs15311d
Wang J, Xue Z, Yan C et al (2016) Fine regulation of cellulose dissolution and regeneration by low pressure CO2 in DMSO/organic base: dissolution behavior and mechanism. Phys Chem Chem Phys 18:32772–32779. https://doi.org/10.1039/c6cp05541a
Wolfs J, Meier MAR (2021) A more sustainable synthesis approach for solvent system. Green Chem 23:4410–4420. https://doi.org/10.1039/d1gc01508g
Xie H, Yu X, Yang Y, Zhao ZK (2014) Capturing CO2 for cellulose dissolution. Green Chem 16:2422–2427. https://doi.org/10.1039/c3gc42395f
Xu Q, Song L, Zhang L et al (2017) Organocatalytic cellulose dissolution and in situ grafting of ϵ-caprolactone via ROP in a reversible DBU/DMSO/CO2 system. ChemistrySelect 2:7128–7134. https://doi.org/10.1002/slct.201701639
Xu et al (2019) Carbohyd Polym 209:382–388
Yang Y, Song L, Peng C et al (2015) Activating cellulose via its reversible reaction with CO2 in the presence of 1,8-diazabicyclo[5.4.0]undec-7-ene for the efficient synthesis of cellulose acetate. Green Chem 17:2758–2763. https://doi.org/10.1039/c5gc00115c
Yuanlong G, Li L, Gu G et al (2021) Synthesis of a fully biobased cellulose- 3-(2-hydroxyphenyl) propionate ester with antioxidant activity and UV-resistant properties by the DBU/CO2/DMSO solvent system. Green Chem 23:2352. https://doi.org/10.1039/d0gc03478a
Zhang H, Wu J, Zhang J, He J (2005) 1-Allyl-3-methylimidazolium chloride room temperature ionic liquid: a new and powerful nonderivatizing solvent for cellulose. Macromolecules 38:8272–8277. https://doi.org/10.1021/ma0505676
Zhang Q, Oztekin NS, Barrault J et al (2013) Activation of microcrystalline cellulose in a CO2-based switchable system. Chemsuschem 6:593–596. https://doi.org/10.1002/cssc.201200815
Zhou S, Tashiro K, Hongo T et al (2001) Influence of water on structure and mechanical properties of regenerated cellulose studied by an organized combination of infrared spectra, X-ray diffraction, and dynamic viscoelastic data measured as functions of temperature and humidity. Macromolecules 34:1274–1280. https://doi.org/10.1021/ma001507x
Acknowledgments
We are grateful for the financial support by Ningbo Natural Science Foundation (2021J196), Key Research and Development Program of Shandong Province (2019JZZY020217), and the National Natural Science Foundation of China (21978310). We also gratefully acknowledge Prof. Xinghong Zhang for his help with the solid state 13C CP/MAS NMR.
Funding
Ningbo Natural Science Foundation (2021J196), Key Research and Development Program of Shandong Province (2019JZZY020217), and the National Natural Science Foundation of China (21978310).
Author information
Authors and Affiliations
Contributions
JZ carried out the majority parts of experiments, analysis of experimental results, and writing of manuscript. SM carried out part of experiments and analysis of experimental results. FL guided the analysis of experimental data and the writing of this manuscript. QQ gave constructive discussion during experiment and manuscript preparation. HN helped performing the analysis of partially experimental data. JZ guided the general conception of this study.
Corresponding authors
Ethics declarations
Competing interests
There is no competing interest to declare.
Ethics approval
Not applicable.
Animal research
Not applicable.
Consent to participate
Not applicable.
Consent to publish
Not applicable.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Zhang, J., Mi, S., Liu, F. et al. Enhanced dissolution of cellulose in CO2 switchable system by solvent with low Henry's constants as CO2 absorbent. Cellulose 29, 6745–6758 (2022). https://doi.org/10.1007/s10570-022-04670-y
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10570-022-04670-y