Abstract
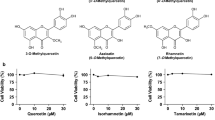
Hyperuricemia, the high uric acid (UA) state in blood, has been accepted as an important risk factor for gout. The liver is a main factory of UA production. In the present study, we have examined the effects of three kinds of flavonol and flavones as typical aglycons, i.e., quercetin, luteolin, apigenin, their glycosides and related compounds, on UA productivity in cultured hepatocytes, adopting allopurinol as the positive control drug. Quercetin, luteolin, diosmetin (4′-O-methylluteolin) and apigenin at 10, 30 and 100 μM as well as allopurinol at 0.1, 0.3 and 1 μM dose-dependently and significantly decreased UA production in the hepatocytes, when compared with 0 μM (control). Both rutin (quercetin-3-O-rutinoside) and quercitrin (quercetin-3-O-ramnoside) significantly reduced UA production in the hepatocytes at 100 μM. Luteolin glycosides such as orientin (luteolin-8-C-glucoside) and isoorientin (luteolin-6-C-glucoside) exerted no influences on it even at 100 μM. Likewise, apigenin glycosides such as vitexin (apigenin-8-C-glucoside) and isovitexin (apigenin-6-C-glucoside) showed no inhibitory effect on it, while apigetrin (apigenin-7-O-glucoside) significantly reduced it at 100 μM. In model mice with purine bodies-induced hyperuricemia, allopurinol completely suppressed the hyperuricemia at a dose of 10 mg/kg body weight. Rutin suppressed significantly the hyperuricemia at a dose of 300 mg/kg body weight, while vitexin showed no significant effect up to 300 mg/kg body weight. Thus, rutin (O-glycoside) is demonstrated to be hypouricemic in both cultured hepatocytes and model mice with recently contrived purine bodies-induced hyperuricemia.





Similar content being viewed by others
Abbreviations
- BSS:
-
Balanced salt solution
- GMP:
-
Guanosine-5′-monophosphate
- IMP:
-
Inosine-5′-monophosphate
- IC50:
-
50% Inhibitory concentration
- PBS (−):
-
Calcium- and magnesium-free phosphate buffered saline
- UA:
-
Uric acid
- XO:
-
Xanthine oxidase
References
Abeles AM (2015) Hyperuricemia, gout, and cardiovascular disease: an update. Curr Rheumatol Rep 17:13. https://doi.org/10.1007/s11926-015-0495-2
Adachi SI, Yoshizawa F, Yagasaki K (2017a) Hyperuricemia in type 2 diabetic model KK-Ay/Ta mice: a potent animal model with positive correlation between insulin resistance and plasma high uric acid levels. BMC Res Notes 10:577. https://doi.org/10.1186/s13104-017-2897-x
Adachi SI, Yoshizawa F, Yagasaki K (2017b) Assay systems for screening food and natural substances that have anti-hyperuricemic activity: uric acid production in cultured hepatocytes and purine bodies-induced hyperuricemic model mice. Cytotechnology 69:435–442. https://doi.org/10.1007/s10616-016-0005-z
Adachi SI, Nihei KI, Ishihara Y, Yoshizawa F, Yagasaki K (2017c) Anti-hyperuricemic effect of taxifolin in cultured hepatocytes and model mice. Cytotechnology 69:329–336. https://doi.org/10.1007/s10616-016-0061-4
Adachi SI, Kondo S, Sato Y, Yoshizawa F, Yagasaki K (2019) Anti-hyperuricemic effect of isorhamnetin in cultured hepatocytes and model mice: structure-activity relationships of methylquercetins as inhibitors of uric acid production. Cytotechnology 71:181–192. https://doi.org/10.1007/s10616-018-0275-8
Babio N, Martínez-González MA, Estruch R, Wärnberg J, Recondo J, Ortega-Calvo M, Serra-Majem L, Corella D, Fitó M, Ros E, Becerra-Tomás N, Basora J, Salas-Salvadó J (2015) Associations between serum uric acid concentrations and metabolic syndrome and its components in the PREDIMED study. Nutr Metab Cardiovasc Dis 25:173–180. https://doi.org/10.1016/j.numecd.2014.10.006
Bardin T, Richette P (2014) Definition of hyperuricemia and gouty conditions. Curr Opin Rheumatol 26:186–191. https://doi.org/10.1097/BOR.0000000000000028
Chen YS, Hu QH, Zhang X, Zhu Q, Kong LD (2013) Beneficial effect of rutin on oxonate-induced hyperuricemia and renal dysfunction in mice. Pharmacology 92:75–83. https://doi.org/10.1159/000351703
Choi HK, Atkinson K, Karlson EW, Willett W, Curhan G (2004) Purine-rich foods, dairy and protein intake, and the risk of gout in men. N Engl J Med 350:1093–1103. https://doi.org/10.1056/NEJMoa035700
Choi HK, Mount DB, Reginato AM (2005) American college of physicians; American Physiological Society. Pathogenesis of gout. Ann Intern Med 143:499–516. https://doi.org/10.7326/0003-4819-143-7-200510040-00009
de Araújo ME, Moreira Franco YE, Alberto TG, Sobreiro MA, Conrado MA, Priolli DG, Frankland Sawaya AC, Ruiz AL, de Carvalho JE, de Oliveira CP (2013) Enzymatic de-glycosylation of rutin improves its antioxidant and antiproriferative activities. Food Chem 141:266–273. https://doi.org/10.1016/j.foodchem.2013.02.127
Ishikawa T, Aw W, Kaneko K (2013) Metabolic interactions of purine derivatives with human ABC transporter ABCG2: genetic testing to assess gout risk. Pharmaceuticals (Basel) 6:1347–1360. https://doi.org/10.3390/ph6111347
Kondo M, Hirano Y, Nishio M, Furuya Y, Nakamura H, Watanabe T (2013) Xanthine oxidase inhibitory activity and hypouricemic effect of aspalathin from unfermented rooibos. J Food Sci 78:H1935-1939. https://doi.org/10.1111/1750-3841.12304
Nguyen MT, Awale S, Tezuka Y, Tran QL, Watanabe H, Kadota S (2004) Xanthine oxidase inhibitory activity of Vietnamese medicinal plants. Biol Pharm Bull 27:1414–1421. https://doi.org/10.1248/bpb.27.1414
Petrie JL, Patman GL, Sinha I, Alexander TD, Reeves HL, Agius L (2013) The rate of production of uric acid by hepatocytes is a sensitive index of compromised cell ATP homeostasis. Am J Physiol Endocrinol Metab 305:E1255–E1265. https://doi.org/10.1152/ajpendo.00214.2013
Richette P, Bardin T (2012) Purine-rich foods: an innocent bystander of gout attacks? Ann Rheum Dis 71:1435–1436. https://doi.org/10.1136/annrheumdis-2012-201838
Shin NR, Moon JS, Shin SY, Li L, Lee YB, Kim TJ, Han NS (2016) Isolation and characterization of human intestinal Enterococcus avium EFEL009 converting rutin to quercetin. Lett Appl Microbiol 62:68–74. https://doi.org/10.1111/lam.12512
Son MJ, Minakawa M, Miura Y, Yagasaki K (2013) Aspalathin improves hyperglycemia and glucose intolerance in obese diabetic ob/ob mice. Eur J Nutr 52:1607–1619. https://doi.org/10.1007/s00394-012-0466-6
Wang Y, Qin S, Jia J, Huang L, Li F, Jin F, Ren Z, Wang Y (2019) Intestinal microbiota-associated metabolites: crucial factors in the effectiveness of herbal medicines and diet therapies. Front Physiol 10:1343. https://doi.org/10.3389/fphys.2019.01343
Yang J, Lee H, Sung J, Kim Y, Jeong HS, Lee J (2019) Conversion of rutin to quercetin by acid treatment in relation to biological activities. Prev Nutr Food Sci 24:313–320. https://doi.org/10.3746/pnf.2019.24.3.313
Zhang G, Xu Z, Gao Y, Huang X, Zou Y, Yang T (2015) Effects of germination on the nutritional properties, phenolic profiles, and antioxidant activities of buckwheat. J Food Sci 80:H1111–H1119. https://doi.org/10.1111/1750-3841.12830
Zheng S, Geng D, Liu S, Wang Q, Liu S, Wang R (2019) A newly isolated human intestinal bacterium strain capable of deglycosylating flavone C-glycosides and its functional properties. Microb Cell Fact 18:94. https://doi.org/10.1186/s12934-019-1144-7
Zhu JX, Wang Y, Kong LD, Yang C, Zhang X (2004) Effects of Biota orientalis extract and its flavonoid constituents, quercetin and rutin on serum uric acid levels in oxonate-induced mice and xanthine dehydrogenase and xanthine oxidase activities in mouse liver. J Ethnopharmacol 93:133–140. https://doi.org/10.1016/j.jep.2004.03.037
Zhu Y, Hu Y, Huang T, Zhang Y, Li Z, Luo C, Luo Y, Yuan H, Hisatome I, Yamamoto T, Cheng J (2014) High uric acid directly inhibits insulin signalling and induces insulin resistance. Biochem Biophys Res Commun 447:707–714. https://doi.org/10.1016/j.bbrc.2014.04.080
Acknowledgements
Authors are grateful to Dr. Fumiaki Yoshizawa, Utsunomiya University, for providing animal experiment and feeding facility.
Author information
Authors and Affiliations
Contributions
KY supervised the study and wrote the manuscript in collaboration with SA. SA, MO and SK participated in the experimental works, and collected samples and data. SA conducted all statistical analyses. All authors have read and approved the final version of the manuscript.
Corresponding author
Ethics declarations
Conflict of interest
This study was funded by Suntory Malting Ltd., Utsunomiya, Tochigi, Japan. MO is the employee for this company.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Adachi, Si., Oyama, M., Kondo, S. et al. Comparative effects of quercetin, luteolin, apigenin and their related polyphenols on uric acid production in cultured hepatocytes and suppression of purine bodies‐induced hyperuricemia by rutin in mice. Cytotechnology 73, 343–351 (2021). https://doi.org/10.1007/s10616-021-00452-9
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10616-021-00452-9