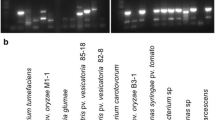
Abstract
Clavibacter michiganensis subsp. sepedonicus and Ralstonia solanacearum (Smith) Yabuuchi et al. race 3 are the causal agents of ring-rot and brown-rot of potato respectively. These diseases represent a serious threat to potato production in temperate climates. Both bacteria are listed as A2 pests in the EPPO region and as zero-tolerance quarantine organisms in the European Union. All the detection tests developed so far were only focused on the detection of a single pathogen while the absence of both bacteria has to be certified in the seed tubers. We have therefore developed a new multiplex real-time PCR assay to simultaneously detect both bacteria in a single assay. Additionally, the reliability of this molecular diagnostic test has been improved by the simultaneous amplification of an internal control, corresponding to a potato gene co-extracted from the sample. The polyvalence and the specificity of each set of bacterial primers and probes were evaluated on more than 90 bacterial strains. The limit of detection of this triplex real-time protocol was similar to those observed with other molecular protocols previously developed for the individual detection of one of these bacteria. A concordance of 100 % was obtained in a blind test mimicking the routine application of the technology. In conclusion, this new protocol represents a straightforward and convenient method potentially adapted to primary screening of potato tubers.
Similar content being viewed by others
References
Abu Al-Soud, W., & Rådström, P. (1998). Capacity of nine thermostable DNA polymerases to mediate DNA amplification in the presence of PCR-inhibiting samples. Applied and Environmental Microbiology, 64(10), 3748–3753.
Arulappan, F. X., van Beuningen, A. R., Derks, J. H. J., Wullings, B., Tjou-Tam-Sin, N. N. A., & Janse, J. D. (1996). Comparison of the indirect and direct immunofluorescence test for the detection of latent brown rot infection, with a new antiserum. Annual report of the Plantenziekten-kundige dienst, Diagnostic center (pp. 6). Wageningen, The Netherlands.
Bach, H. J., Jessen, I., Schloter, M., & Munch, J. C. (2003). A TaqMan-PCR protocol for quantification and differentiation of the phytopathogenic Clavibacter michiganensis subspecies. Journal of Microbiological Methods, 52(1), 85–91. doi:10.1016/S0167-7012(02)00152-5.
Baric, S., & Dalla-Via, J. (2004). A new approach to apple proliferation detection: a highly sensitive real-time PCR assay. Journal of Microbiological Methods, 57(1), 135–145.
Boivin, G., Côté, S., Déry, P., De Serres, G., & Bergeron, M. G. (2004). Multiplex real-time PCR assay for detection of influenza and human respiratory syncytial viruses. Journal of Clinical Microbiology, 42(1), 45–51.
Ciampi, L., Sequeira, L., & French, E. R. (1981). Pseudomonas solanacearum. Distribution in potato plants and the establishment of latent infections. In C. Lozano (Ed.), Proceedings of the 5th International Conference on Plant Pathogenic Bacteria (pp. 148–161). Cali, Colombia: Centro Internacional de Agricultura Tropical.
Elphinstone, J. G., Hennessy, J., Wilson, J. K., & Stead, D. E. (1996). Sensitivity of different methods for the detection of Ralstonia solanacearum in potato tuber extracts. EPPO Bulletin, 26, 15.
Fernández-Pinero, J., Gallardo, C., Elizalde, M., Robles, A., Gómez, C., Bishop, R., & Arias, M. (2013). Molecular diagnosis of African swine fever by a new real-time PCR using universal probe library. Transboundary and Emerging Diseases, 60(1), 48–58.
Fucikovsky, L., & Duveiller, E. (1997). Other plant pathogenic bacteria reported on wheat. In E. Duveiller, L. Fucikovsky, & K. Rudolph (Eds.), The bacterial diseases of wheat. Concept and methods of disease management (pp. 59–64). Mexico: CIMMYT.
Hoorfar, J., Cook, N., Malorny, B., Wagner, M., De Medici, D., Abdulmawjood, A., & Fach, P. (2003). Making internal amplification control mandatory for diagnostic PCR [2]. Journal of Clinical Microbiology, 41(12), 5835.
James, D., Varga, A., Pallas, V., & Candresse, T. (2006). Strategies for simultaneous detection of multiple plant viruses. Canadian Journal of Plant Pathology, 28(1), 16–29.
Li, W., Teixeira, D. C., Hartung, J. S., Huang, Q., Duan, Y., Zhou, L., & Levy, L. (2013). Development and systematic validation of qPCR assays for rapid and reliable differentiation of Xylella fastidiosa strains causing citrus variegated chlorosis. Journal of Microbiological Methods, 92(1), 79–89.
Mackay, I. M. (2004). Real-time PCR in the microbiology laboratory. Clinical Microbiology and Infection, 10(3), 190–212. doi:10.1111/j.1198-743X.2004.00722.x.
Malorny, B., Paccassoni, E., Fach, P., Bunge, C., Martin, A., & Helmuth, R. (2004). Diagnostic real-time PCR for detection of Salmonella in food. Applied and Environmental Microbiology, 70(12), 7046–7052.
Massart, S., De Clercq, D., Salmon, M., Dickburt, C., & Jijakli, M. H. (2005). Development of real-time PCR using Minor Groove Binding probe to monitor the biological control agent Candida oleophila (strain O). Journal of Microbiological Methods, 60(1), 73–82.
Massart, S., Brostaux, Y., Barbarossa, L., César, V., Cieslinska, M., Dutrecq, O., & Jijakli, M. H. (2008). Inter-laboratory evaluation of a duplex RT-PCR method using crude extracts for the simultaneous detection of Prune dwarf virus and Prunus necrotic ringspot virus. European Journal of Plant Pathology, 122(4), 539–547.
Mumford, R., Boonham, N., Tomlinson, J., & Barker, I. (2006). Advances in molecular phytodiagnostics—new solutions for old problems. European Journal of Plant Pathology, 116(1), 1–19.
Ozakman, M., & Schaad, N. W. (2003). A real-time BIO-PCR assay for detection of Ralstonia solanacearum race 3, biovar 2, in asymptomatic potato tubers. Canadian Journal of Plant Pathology, 25(3), 232–239. doi:10.1080/07060660309507075.
Pastrik, K. H. (2000). Detection of Clavibacter michiganensis subsp. sepedonicus in potato tubers by multiplex PCR with coamplification of host DNA. European Journal of Plant Pathology, 106(2), 155–165. doi:10.1023/A:1008736017029.
Pastrik, K. H., & Maiss, E. (2000). Detection of Ralstonia solanacearum in potato tubers by polymerase chain reaction. Journal of Phytopathology, 148(11–12), 619–626.
Pastrik, K. H., Elphinstone, J. G., & Pukall, R. (2002). Sequence analysis and detection of Ralstonia solanacearum by multiplex PCR amplification of 16S-23S ribosomal intergenic spacer region with internal positive control. European Journal of plant pathology/European Foundation for Plant Pathology, 108(9), 831–842.
Schaad, N. W., Berthier-Schaad, Y., Sechler, A., & Knorr, D. (1999). Detection of Clavibacter michiganensis subsp. sepedonicus in potato tubers by BIO-PCR and an automated real-time fluorescence detection system. Plant Disease, 83(12), 1095–1100.
Seal, S., Taghavi, M., Fegan, N., Hayward, A. C., & Fegan, M. (1999). Determination of Ralstonia (Pseudomonas) solanacearum rDNA subgroups by PCR tests. Plant Pathology, 48, 115–120. doi:10.1046/j.1365-3059.1999.00322.x.
Van Brunschot, S. L., Gambley, C. F., De Barro, P. J., Grams, R., Thomas, J. E., Henderson, J., et al. (2013). Panel of real-time PCRs for the multiplexed detection of two tomato-infecting begomoviruses and their cognate whitefly vector species. Plant Pathology.
Van Hoeyveld, E., Houtmeyers, F., Massonet, C., Moens, L., Van Ranst, M., Blanckaert, N., & Bossuyt, X. (2004). Detection of single nucleotide polymorphisms in the mannose-binding lectin gene using minor groove binder-DNA probes. Journal of Immunological Methods, 287(1–2), 227–230.
Weller, S. A., Elphinstone, J. G., Smith, N. C., Boonham, N., & Stead, D. E. (2000). Detection of Ralstonia solanacearum strains with a quantitative, multiplex, real-time, fluorogenic PCR (TaqMan) assay. Applied and Environmental Microbiology, 66(7), 2853–2858. doi:10.1128/aem.66.7.2853-2858.2000.
Yao, Y., Nellåker, C., & Karlsson, H. (2006). Evaluation of minor groove binding probe and TaqMan probe PCR assays: influence of mismatches and template complexity on quantification. Molecular and Cellular Probes, 20(5), 311–316.
Zielke, R., & Naumann, K. (1984). Untersuchungen zur Erfassung des latenten Befallsstadiums von Corynebacterium sepedonicum (Spieck. et Kotth.) Skapt. et Burkh. im Kartoffelgewebe. Zentralblatt für Mikrobiologie, 139(4), 267–280.
Acknowledgments
We thank Dr. Johan Van Vaerenbergh and Dr. Rachid Tahzima (ILVO, Gent, Belgium) for fruitful collaboration and for sending the blinded samples. We are grateful to Sylvie Van Mellaert for excellent technical assistance. We also thank MM. Walter Van Ormelingen et David Michelante from the Federal Agency for the safety of the Food Chain (AFSCA - Belgium) for the external coordination of the blind-test and for fruitful discussions throughout the project. This work was supported by AFSCA and the General Management of Agriculture (DGA) of the Walloon Region Ministry (Belgium).
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Massart, S., Nagy, C. & Jijakli, M.H. Development of the simultaneous detection of Ralstonia solanacearum race 3 and Clavibacter michiganensis subsp. sepedonicus in potato tubers by a multiplex real-time PCR assay. Eur J Plant Pathol 138, 29–37 (2014). https://doi.org/10.1007/s10658-013-0294-4
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10658-013-0294-4