Abstract
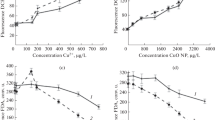
Metal pollution can produce many biological effects on aquatic environments. The marine diatom Amphora subtropica and the green alga Dunaliella sp. possess a high metal absorption capacity. Nickel (Ni) removal by living cells of A. subtropica and Dunaliella sp. was tested in cultures exposed to different Ni concentrations (100, 200, 300, and 500 mg L−1). The amount of Ni removed by the microalgae increased with the time of exposure and the initial Ni concentration in the medium. The metal, which was mainly removed by bioadsorption to Dunaliella sp. cell surfaces (93.63% of total Ni (for 500 mg Ni L−1) and by bioaccumulation (80.82% of total Ni (for 300 mg Ni L−1) into Amphora subtropica cells, also inhibited growth. Exposure to Ni drastically reduced the carbohydrate and protein concentrations and increased total lipids from 6.3 to 43.1 pg cell−1, phenolics 0.092 to 0.257 mg GAE g−1 (Fw), and carotenoid content, from 0.08 to 0.59 mg g−1 (Fw), in A. subtropica. In Dunaliella sp., total lipids increased from 26.1 to 65.3 pg cell−1, phenolics from 0.084 to 0.289 mg GAE g−1 (Fw), and carotenoid content from 0.41 to 0.97 mg g−1 (Fw). These compounds had an important role in protecting the algae against ROS generated by Ni. In order to cope with Ni stress shown by the increase of TBARS level, enzymatic (SOD, CAT, and GPx) ROS scavenging mechanisms were induced.



Similar content being viewed by others
References
Adey WH, Kangas PC, Mulbry W (2011) Algal turf scrubbing: cleaning surface waters with solar energy while producing a biofuel. Bioscience 61:434–441
Aebi H (1984) Catalase in vitro. Meth Enzymol 105:121–126
Alscher RG, Erturk N, Heath LS (2002) Role of superoxide dismutases (SODs) in controlling oxidative stress in plants. J Exp Bot 53:1331–1341
Anantharaj K, Govindasamy C, Natanamurugaraj G, Jeyachandran S (2011) Effect of heavy metals on marine diatom Amphora coffeaeformis (Agardh. Kutz). Glob J Environ Res 5:112–117
Apel K, Hirt H (2004) Reactive oxygen species: metabolism, oxidative stress, and signal transduction. Annu Rev Plant Biol 55:373–399
Belghith T, Athmouni K, Bellassoued K, El Feki A, Ayadi H (2016) Physiological and biochemical response of Dunaliella salina to cadmium pollution. J Appl Phycol 28:991–999
Beyer WF, Fridovich I (1987) Assaying for superoxide dismutase activity: some large consequences of minor changes in conditions. Anal Biochem 161:559–566
Bligh EG, Dyer WJ (1959) A rapid method of total lipid extraction and purification. Can J Biochem Physiol 37:911–917
Branco D, Lima A, Almeida SF, Figueira E (2010) Sensitivity of biochemical markers to evaluate cadmium stress in the freshwater diatom Nitzschia palea (Kützing) W. Smith. Aquat Toxicol 99:109–117
Carr H, Carino F, Yang M, Wong M (1998) Characterization of the cadmium-binding capacity of Chlorella vulgaris. Bull Environ Contam Toxicol 60:433–440
Chekroun KB, Baghour M (2013) The role of algae in phytoremediation of heavy metals: a review. J Mater Environ Sci 4:873–880
Chen H, Zhang Y, He C, Wang Q (2014) Ca2+ signal transduction related to neutral lipid synthesis in an oil-producing green alga Chlorella sp. C2. Plant Cell Physiol 55:634–644
Dahmen Ben Moussa I, Chtourou H, Rezgui F, Sayadi S, Dhouib A (2016) Salinity stress increases lipid, secondary metabolites and enzyme activity in Amphora subtropica and Dunaliella sp. for biodiesel production. Bioresour Technol 218:816–825
Das K, Das S, Dhundasi S (2008) Nickel, its adverse health effects & oxidative stress. Indian J Med Res 128:412
de la Vega L, Fröbius K, Moreno R, Calzado MA, Geng H, Schmitz ML (2011) Control of nuclear HIPK2 localization and function by a SUMO interaction motif. Biochim Biophys Acta 1813:283–297
Dittami SM, Gravot A, Renault D, Goulitquer S, Eggert A, Bouchereau A, Boyen C, Tonon T (2011) Integrative analysis of metabolite and transcript abundance during the short-term response to saline and oxidative stress in the brown alga Ectocarpus siliculosus. Plant Cell Environ 34:629–642
Draper H, Hadley M (1990) Malondialdehyde determination as index of lipid peroxidation. Methods Enzymol 186:421–431
Dubois M, Gilles KA, Hamilton JK, Rebers PA, Smith F (1956) Colorimetric method for determination of sugars and related substances. Anal Chem 28:350–356
Elbaz A, Wei YY, Meng Q, Zheng Q, Yang ZM (2010) Mercury-induced oxidative stress and impact on antioxidant enzymes in Chlamydomonas reinhardtii. Ecotoxicology 19:1285–1293
Ercal N, Gurer Orhan H, Aykin Burns N (2001) Toxic metals and oxidative stress part I: mechanisms involved in metal-induced oxidative damage. Curr Top Med Chem 1:529–539
Flohé L, Günzler WA (1984) Assays of glutathione peroxidase. Methods Enzymol 105:114–120
Gong N, Shao K, Feng W, Lin Z, Liang C, Sun Y (2011) Biotoxicity of nickel oxide nanoparticles and bio-remediation by microalgae Chlorella vulgaris. Chemosphere 83:510–516
Hiscox JT, Israelstam G (1979) A method for the extraction of chlorophyll from leaf tissue without maceration. Can J Bot 57:1332–1334
Jennings J, Rainbow P (1979) Accumulation of cadmium by Dunaliella tertiolecta Butcher. J Plankton Res 1:67–74
Jiang Y, Nunez M, Laverty KS, Quigg A (2015) Coupled effect of silicate and nickel on the growth and lipid production in the diatom Nitzschia perspicua. J Appl Phycol 27:1137–1148
Knothe G (2006) Analyzing biodiesel: standards and other methods. J Am Oil Chem Soc 83:823–833
Liu LN, Bryan SJ, Huang F, Yu J, Nixon PJ, Rich PR, Mullineaux CW (2012) Control of electron transport routes through redox-regulated redistribution of respiratory complexes. Proc Natl Acad Sci 109:11431–11436
Lowry OH, Rosebrough NJ, Farr AL, Randall RJ (1951) Protein measurement with the Folin phenol reagent. J Biol Chem 193:265–275
Luis P, Behnke K, Toepel J, Wilhelm C (2006) Parallel analysis of transcript levels and physiological key parameters allows the identification of stress phase gene markers in Chlamydomonas reinhardtii under copper excess. Plant Cell Environ 29:2043–2054
Martinez Ruiz EB, Martinez Jeronimo F (2015) Nickel has biochemical, physiological, and structural effects on the green microalga Ankistrodesmus falcatus: an integrative study. Aquat Toxicol 169:27–36
Matagi S, Swai D, Mugabe R (1998) A review of heavy metal removal mechanisms in wetlands. Afr J Trop Hydrobiol Fish 8:13–25
Matsunaga T, Takeyama H, Nakao T, Yamazawa A (1999) Screening of marine microalgae for bioremediation of cadmium-polluted seawater. J Biotechnol 70:33–38
Monteiro CM, Castro PM, Malcata FX (2012) Metal uptake by microalgae: underlying mechanisms and practical applications. Biotechnol Prog 28:299–311
Moroney JV, Bartlett SG, Samuelsson G (2001) Carbonic anhydrases in plants and algae. Plant Cell Environ 24:141–153
Murthy KC, Vanitha A, Rajesha J, Swamy MM, Sowmya P, Ravishankar GA (2005) In vivo antioxidant activity of carotenoids from Dunaliella salina—a green microalga. Life Sci 76:1381–1390
Pancha I, Chokshi K, Mishra S (2015a) Enhanced biofuel production potential with nutritional stress amelioration through optimization of carbon source and light intensity in Scenedesmus sp. CCNM 1077. Bioresour Technol 179:565–572
Pancha I, Chokshi K, Maurya R, Trivedi K, Patidar SK, Ghosh A, Mishra S (2015b) Salinity induced oxidative stress enhanced biofuel production potential of microalgae Scenedesmus sp. CCNM 1077. Bioresour Technol 189:341–348
Perales Vela HV, Peña Castro JM, Cañizares Villanueva RO (2006) Heavy metal detoxification in eukaryotic microalgae. Chemosphere 64:1–10
Pérez Rama M, Alonso JA, Lopez CH, Vaamonde ET (2002) Cadmium removal by living cells of the marine microalga Tetraselmis suecica. Bioresour Technol 84:265–270
Pinto E, Sigaud-kutner T, Leitao MA, Okamoto OK, Morse D, Colepicolo P (2003) Heavy metal-induced oxidative stress in algae. J Phycol 39:1008–1018
Provasoli L, McLaughlin J, Droop M (1957) The development of artificial media for marine algae. Arch Mikrobiol 25:392–428
Sabatini SE, Juarez AB, Eppis MR, Bianchi L, Luquet CM, Reos de Molina MC (2009) Oxidative stress and antioxidant defenses in two green microalgae exposed to copper. Ecotoxicol Environ Saf 72:1200–1206
Sacan MT, Oztay F, Bolkent S (2007) Exposure of Dunaliella tertiolecta to lead and aluminum: toxicity and effects on ultrastructure. Biol Trace Elem Res 120:264–272
Shariati M, Hadi MR (2011) Microalgal biotechnology and bioenergy in Dunaliella. In: Carpi A (ed) Progress in molecular and environmental bioengineering—from analysis and modeling to technology applications. InTech, Rijeka, pp 483–506
Singleton VL, Rossi JA (1965) Colorimetry of total phenolics with phosphomolybdic-phosphotungstic acid reagents. Am J Enol Vitic 16:144–158
Stauber J, Florence T (1985) The influence of iron on copper toxicity to the marine diatom, Nitzschia closterium (Ehrenberg) W. Smith. Aquat Toxicol 6:297–305
Tewari RK, Kumar P, Sharma PN, Bisht SS (2002) Modulation of oxidative stress responsive enzymes by excess cobalt. Plant Sci 162:381–388
Torres E, Cid A, Herrero C, Abalde J (1998) Removal of cadmium ions by the marine diatom Phaeodactylum tricornutum Bohlin accumulation and long-term kinetics of uptake. Bioresour Technol 63:213–220
Travieso L, Canizares R, Borja R, Benitez F, Dominguez A, Dupeyrón R, Valiente V (1999) Heavy metal removal by microalgae. Bull Environ Contam Toxicol 62:144–151
Winters C, Guéguen C, Noble A (2017) Equilibrium and kinetic studies of Cu(II) and Ni(II) sorption on living Euglena gracilis. J Appl Phycol 29:1391–1398
Zutshi S, Choudhary M, Bharat N, Abdin MZ, Fatma T (2008) Evaluation of antioxidant defense responses to lead stress in Hapalosiphon fontinalis-3391. J Phycol 44:889–896
Acknowledgements
This study was supported by the Ministry of Higher Education and Scientific Research of Tunisia under Contract Program of the Environmental Bioprocesses Laboratory.
Authors’ contributions
IDB designed and performed the experiments, analyzed the data, and wrote the paper. KA and HC participated in the design and execution of the experiments. HA, SS, and AD critically reviewed the manuscript. All authors read and approved the final manuscript.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Dahmen-Ben Moussa, I., Athmouni, K., Chtourou, H. et al. Phycoremediation potential, physiological, and biochemical response of Amphora subtropica and Dunaliella sp. to nickel pollution. J Appl Phycol 30, 931–941 (2018). https://doi.org/10.1007/s10811-017-1315-z
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10811-017-1315-z