Abstract
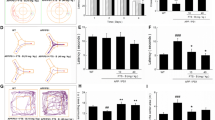
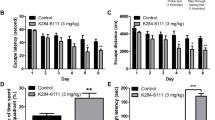
Neuroinflammation can cause multiple neurodegenerative diseases, such as Alzheimer’s disease (AD), Parkinson’s disease (PD), and Huntington’s disease (HD). Recent studies have shown that the artemisinin derivative dihydroartemisinin (DHA) can be used as an immunomodulatory, anti-inflammatory and anti-tumor agent. The anti-neuroinflammatory effects of DHA were evaluated in our study, and the underlying mechanisms were explored using the Morris water maze test (MWMT), Open-field test (OFT) and Closed-field test (CFT), Elevated plus maze test (EPMT), Nissl Staining, Immunofluorescence analysis, RT-PCR, and Western Blot. Our results show that DHA significantly inhibits LPS-induced inflammation and attenuates LPS-induced behavioral and memory disorders. 1. Behavioral test results: 1) in the water maze test, the mice in the LPS group showed increased escape latency and length of the movement path on the third day; they also had a decreased number of crossings of the target quadrant after the platform was removed on the 5th day and remained in the target quadrant for less time; 2) in the open- and closed-field experiment, the number of activities and activities in the open-field were significantly reduced; 3) in the elevated cross maze experiment, LPS-treated mice exhibited a significant reduction in the number of times and the time to enter the open arm; the above behavior was reversed after DHA treatment. 2. Nissl staining results: compared with the Control group, the LPS group showed significant damage, and the number of damaged cells in the hippocampal CA1, CA2, CA3 and DG regions was increased; DHA treatment reduced cell damage. 3. RT-PCR results: compared with the Control group, the LPS group showed increased expression of IL-1β and IL-6 but decreased expression after DHA treatment. 4. GFAP fluorescent staining: compared with the control group, the corresponding reactivity of positive cells in the LPS-induced group was increased in the CA1-CA3 and DG regions of the hippocampus; compared with the LPS-induced mice, cells in the LPS + DHA group showed significantly reduced reactivity (GFAP). 5. Western blot results: compared with the Control group, the LPS group showed increased expression of P-PI3K/PI3K, P-AKT/AKT, IL-6 and TNFα and a decreased expression of P-PI3K/PI3K, IL-6, TNF and P-AKT/AKT after DHA treatment. Our findings provide direct evidence for the potential use of DHA in the treatment of neuroinflammatory diseases.










Similar content being viewed by others
References
Ali T, Badsha H, TH K, Kim MO (2015) Melatonin attenuates D-galactose-induced memory impairment, neuroinflammation and neurodegeneration via RAGE/NF-K B/JNK signaling pathway in aging mouse model. J Pineal Res 58(1):71–85. https://doi.org/10.1111/jpi.12194
Arroyo DS, Gaviglio EA, Peralta Ramos JM, Bussi C, Rodriguez-Galan MC, Iribarren P (2014) Autophagy in inflammation, infection, neurodegeneration and cancer. Int Immunopharmacol 18(1):55–65. https://doi.org/10.1016/j.intimp.2013.11.001
Badshah H, Ali T, Ahmad A, Kim MJ, Abid NB, Shah SA, Yoon GH, Lee HY, Kim MO (2015) Co-treatment with anthocyanins and vitamin C ameliorates ethanol-induced neurodegeneration via modulation of GABAB receptor signaling in the adult rat brain. CNS Neurol Disord Drug Targets 14(6):791–803. https://doi.org/10.2174/1871527314666150225142919
Badshah H, Ali T, Kim MO et al (2016) Osmotin attenuates LPS-induced neuroinflammation and memory impairments via the TLR4/NFκB signaling pathway. Sci Rep 6:24493. https://doi.org/10.1038/srep24493
Barnhart CD, Yang D, Lein PJ (2015) Using the Morris water maze to assess spatial learning and memory in weanling mice. PLoS One 10(4):e0124521. https://doi.org/10.1371/journal.pone.0124521
Biesmans S, Meert TF, Bouwknecht JA, Acton PD, Davoodi N, de Haes P, Kuijlaars J, Langlois X, Matthews LJ, ver Donck L, Hellings N, Nuydens R (2013) Systemic immune activation leads to neuroinflammation and sickness behavior in mice. Mediat Inflamm 2013:271359. https://doi.org/10.1155/2013/271359
Chaudhuri AD, Dastgheyb RM, Yoo SW, Trout A, Talbot CC Jr, Hao H, Witwer KW, Haughey NJ (2018) TNFα and IL-1β modify the miRNA cargo of astrocyte shed extracellular vesicles to regulate neurotrophic signaling in neuro-ns. Cell Death Dis 9(3):363. https://doi.org/10.1038/s41419-018-0369-4
Chen T, Guo Q, Wang H, Zhang H, Wang C, Zhang P, Meng S, Li Y, Ji H, Yan T (2015) E-ffects of esculetin on lipopolysaccharide (LPS)-induced acute lung injury via regulation of RhoA/rho kinase/NF-кB pathways in vivo and in vitro. Free Radic Res 49(12):1459–1468. https://doi.org/10.3109/10715762.2015.108764310.3109/10715762.2015.1087643
Gigante AF, Bruno G, Iliceto G et al (2015) Action tremor in Parkinson’s disease: frequency and relationship to motor and non-motor signs. Neurology 22(2):223–228. https://doi.org/10.1111/ene.12583
Glass CK, Saijo K, Winner B, Marchetto MC, Gage FH (2010) Mechanisms underlying inflammation in neurodegeneration. Cell 140(6):918–934. https://doi.org/10.1016/j.cell.2010.02.016
Khan MS, Ali T, Abid MN, Jo MH et al (2016) Anthocyanins protect against LPS-induced oxidative stress-mediated neuroinflammation and neurodegeneration in the adult mouse cortex. Neurochem Int 100:1–10. https://doi.org/10.1016/j.neuint.2016.08.005
Khan MS, Ali T, Abid MN, Jo MH, Khan A, Kim MW, Yoon GH, Cheon EW, Rehman SU, Kim MO (2017) Lithium ameliorates lipopolysaccharide-induced neurotoxicity in the cortex and hippocampus of the adult rat brain. Neurochem Int 108:343–354. https://doi.org/10.1016/j.neuint.2017.05.008
Yang Lan & Zhang Dong (2017). Summary of dihydroartemisinin and its application for the treatment of lupus erythematosus. Chin Sci Bull 62(18):2007–2012. https://doi.org/10.1360/N972017-00172
Li T, Chen H, Liu XG, Zhou YX, Bai SF (2012) Immunoregulatory effect of artesunate on allergic contact dermatitis and its mechanism. Yao xue xue bao 47(7):884–889
Lindvall O (2016) Clinical translation of stem cell transplantation in Parkinson's disease. Intern Med 279(1):30–40. https://doi.org/10.1111/joim.12415
Okorji UP, Velagapudi R, El-Bakoush A et al (2015) Antimalarial drug artemether inhibits neuroinflammation in BV2 microglia through Nrf2-dependent mechanisms. Mol Neurobiol 53(9):6426–6443. https://doi.org/10.1007/s12035-015-9543-1
Oliveira AC, Yousif NM, Bhatia HS et al (2016) Poly(I:C) increases the expression of mPGES-1 and COX-2 in rat primary microglia. Neuroinflammation 13(1):11. https://doi.org/10.1186/s12974-015-0473-7
Prut L, Belzung C (2003) The open field as a paradigm to measure the effects of drugs on anxiety-like behaviors: a review. Pharmacology 463(1):3–33. https://doi.org/10.1016/s0014-2999(03)01272-x
Rehman SU, Ali T, Alam SI, Ullah R, Zeb A, Lee KW, Rutten BPF, Kim MO (2018) Ferulic acid rescues LPS-induced neurotoxicity via modulation of the TLR4 receptor in the mouse hippocampus. Mol Neurobiol 56(4):2774–2790. https://doi.org/10.1007/s12035-018-1280-9
Sofroniew MV (2009) Molecular dissection of reactive astrogliosis and glial scar formation. Trends Neurosci 32(12):638–647. https://doi.org/10.1016/j.tins.2009.08.002
Sun Z, Yu JT, Jiang T et al (2013) Genome-wide microRNA profiling of rat hippocampus after status epilepticus induced by amygdala stimulation identifies modulators of neuronal apoptosis. PLoS One 8(10):78375. https://doi.org/10.1371/journal.pone.0078375
Tajuddin N, Moon KH, Marshall SA, Nixon K, Neafsey EJ, Kim HY, Collins MA (2014) Neuroinflammation and neurodegeneration in adult rat brain from binge ethanol exposure: abrogation by docosahexaenoic acid. PLoS One 9(7):101223. https://doi.org/10.1371/journal.pone.0101223
Tremblay M-E, Stevens B et al (2011) The role of microglia in thehealthy brain. Neuroscience 31(45):16064–16069. https://doi.org/10.1523/JNEUROSCI.4158-11.2011
Wang J, Markova D, Anderson DG, Zheng Z, Shapiro IM, Risbud MV (2011) TNF-α and IL-1β promotea disintegrin-like and metalloprotease with thrombospondin type I motif-5-mediated aggrecan degradation through syndecan-4 in intervertebral disc. J Biol Chem 286(46):39738–39749. https://doi.org/10.1074/jbc.M111.264549
Xu K, Wang X, Zhang Q, Liang A, Zhu H, Huang D, Li C, Ye W (2016) Sp1 downregulates proinflammatory cytokine-induced catabolic gene expression in nucleus pulposus cells. Mol Med Rep 14(4):3961–3968. https://doi.org/10.3892/mmr.2016.5730
Yin J, Xia W, Zhang Y, Ding G, Chen L, Yang G, Huang S, Jia Z, Zhang A (2018) Role of dihydroartemisinin in regulating prostaglandin E2 synthesis cascade and inflammation in endothelial cells. Heart Vessel 33(11):1411–1422. https://doi.org/10.1007/s00380-018-1190-9
Zeng Q, Han J, Wang B, An S, Duan Y, Dong S, Ma J, Yang L, Cao X (2012) Water maze spatial learning enhances social recognition ability in aged rats. Neurophysiology 44(6):464–469. https://doi.org/10.1007/s11062-012-9319-4
Zhang FX, Xu RS (2017) Juglanin ameliorates LPS-induced neuroinflammation in animal models of Parkinson’s disease and cell culture via inactivating TLR4/NF-KB pathway. Biomed Pharmacother 97:1011–1019. https://doi.org/10.1016/j.biopha.2017.08.132
Zhang K, Liu J, You X, Kong P, Song Y, Cao L, Yang S, Wang W, Fu Q, Ma Z (2016) P2X7 as a new target for chrysophanol to treat lipopolysaccharide-induced depression in mice. Neurosci Lett 613:60–65. https://doi.org/10.1016/j.neulet.2015.12.043
Acknowledgments
This work was supported by the Jiangsu Students’ Platform for Innovation and Entrepreneurship Training Program (201810313040Y), the National Demonstration Center for Experimental Basic Medical Science Education (Xuzhou Medical University).
Author information
Authors and Affiliations
Contributions
YTG, SYJ and YLW contributed to the conception and design of the study. SJZ, NAK and XFM analyzed the data. CYF and BC drafted the manuscript and figures
Corresponding authors
Ethics declarations
The C57 / BL6 mice used in the laboratory were purchased from the Experimental Animal Center of Xuzhou Medical University and used after being approved by medical ethics.
Conflict of interest
The authors have no conflicts of interest to declare.
Ethical approval
All animal procedures were approved by the Ethics Committee of the Catholic University of Korea and were performed in accordance with the National Institutes of Health’s Guide for the Care and Use of Laboratory Animals (NIH Publication No. 80–23). None of the authors performed any human experiments as part of this research.
Additional information
Publisher’s note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Gao, Y., Cui, M., Zhong, S. et al. Dihydroartemisinin ameliorates LPS-induced neuroinflammation by inhibiting the PI3K/AKT pathway. Metab Brain Dis 35, 661–672 (2020). https://doi.org/10.1007/s11011-020-00533-2
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11011-020-00533-2