Abstract
Mitochondrial encephalomyopathy with lactic acidosis and stroke-like episodes (MELAS) is a disease that should be considered as a differential diagnosis to acute ischemic stroke taking into account its onset pattern and neurological symptoms, which are similar to those of an ischemic stroke. Technological advancements in neuroimaging modalities have greatly facilitated differential diagnosis between stroke and MELAS on diagnostic imaging. Stroke-like episodes in MELAS have the following features: (1) symptoms are neurolocalized according to lesion site; (2) epileptic seizures are often present; (3) lesion distribution is inconsistent with vascular territory; (4) lesions are common in the posterior brain regions; (5) lesions continuously develop in adjacent sites over several weeks or months; (6) neurological symptoms and stroke-like lesions tend to be reversible, as presented on magnetic resonance imaging; (7) the rate of recurrence is high; and; (8) brain dysfunction and atrophy are slowly progressive. The m.3243ANG mutation in the MT-TL1 gene encoding the mitochondrial tRNALeu(UUR) is most commonly associated with MELAS. Although the precise pathophysiology is still unclear, one possible hypothesis for these episodes is a neuronal hyperexcitability theory, including neuron–astrocyte uncoupling. Supplementation, such as with L-arginine or taurine, has been proposed as preventive treatments for stroke-like episodes. As this disease is still untreatable and devastating, numerous drugs are being tested, and new gene therapies hold great promise for the future. This article contributes to the understanding of MELAS and its implications for clinical practice, by deepening their insight into the latest pathophysiological hypotheses and therapeutic developments.






Similar content being viewed by others
Data availability
The authors declare that data supporting the findings of this study are available within the article. The datasets are available from the corresponding author upon reasonable request.
Code availability
Not applicable.
Abbreviations
- CT:
-
Computed tomography
- MRI:
-
Magnetic resonance imaging
- MELAS:
-
Mitochondrial encephalomyopathy with lactic acidosis and stroke-like episodes
- LHON:
-
Leber’s hereditary optic neuropathy
- mtDNA:
-
Mitochondrial DNA
- ATP:
-
Adenosine triphosphate
- FLAIR:
-
Fluid-attenuated inversion recovery
- DWI:
-
Diffusion-weighted image
- SPECT:
-
Single-photon emission CT
- ASL-MRI:
-
Arterial spin labeling MRI
- MRA:
-
Magnetic resonance angiography
- ANLS:
-
Astrocyteneuron lactate shuttle
- NO:
-
Nitric oxide
- NOS:
-
Nitric oxide synthase
- ROS:
-
Reactive oxygen species
- iPSCs:
-
Induced pluripotent stem cells
- GC:
-
Guanylate cyclase
- GTP:
-
Converts guanosine triphosphate
- GMP:
-
Guanosine monophosphate
- Cyt c:
-
Cytochrome c
References
Alemdar M, Iseri P, Selekler M et al (2007) MELAS presented with status epilepticus and anton-babinski syndrome; value of adc mapping in MELAS. J Neuropsychiatr 19:482–483. https://doi.org/10.1176/appi.neuropsych.19.4.482
Almasi M, Motamed MR, Mehrpour M et al (2017) A mitochondrial disorder in a middle age iranian patient: report of a rare case. Basic Clin Neurosci 8:337–343. https://doi.org/10.18869/nirp.bcn.8.4.337
Asano K, Suzuki T, Saito A et al (2018) Metabolic and chemical regulation of tRNA modification associated with taurine deficiency and human disease. Nucleic Acids Res 46:1565–1583. https://doi.org/10.1093/nar/gky068
Attwell D, Buchan AM, Charpak S et al (2010) Glial and neuronal control of brain blood flow. Nature 468:232–243. https://doi.org/10.1038/nature09613
Bladin CF (2000) Seizures after stroke. Arch Neurol 57:1617. https://doi.org/10.1001/archneur.57.11.1617
Chen J, Zhang S, Wu J et al (2020) Essential role of nonessential amino acid glutamine in atherosclerotic cardiovascular disease. DNA Cell Biol 39:8–15. https://doi.org/10.1089/dna.2019.5034
Ciafaloni E, Ricci E, Shanske S et al (1992) MELAS: Clinical features, biochemistry, and molecular genetics. Ann Neurol 31:391–398. https://doi.org/10.1002/ana.410310408
Davis DP, Robertson T, Imbesi SG (2006) Diffusion-weighted magnetic resonance imaging versus computed tomography in the diagnosis of acute ischemic stroke. J Emerg Med 31:269–277. https://doi.org/10.1016/j.jemermed.2005.10.003
Demarest ST, Whitehead MT, Turnacioglu S et al (2014) Phenotypic analysis of epilepsy in the mitochondrial encephalomyopathy, lactic acidosis, and strokelike episodes-associated mitochondrial DNA A3243G mutation. J Child Neurol 29:1249–1256. https://doi.org/10.1177/0883073814538511
Desquiret-Dumas V, Gueguen N, Barth M et al (2012) Metabolically induced heteroplasmy shifting and l-arginine treatment reduce the energetic defect in a neuronal-like model of MELAS. Biochim Biophys Acta - Mol Basis Dis 1822:1019–1029. https://doi.org/10.1016/j.bbadis.2012.01.010
El-Hattab AW, Hsu JW, Emrick LT et al (2012) Restoration of impaired nitric oxide production in MELAS syndrome with citrulline and arginine supplementation. Mol Genet Metab 105:607–614. https://doi.org/10.1016/j.ymgme.2012.01.016
El-Hattab AW, Emrick LT, Chanprasert S et al (2014) Mitochondria: Role of citrulline and arginine supplementation in MELAS syndrome. Int J Biochem Cell Biol 48:85–91. https://doi.org/10.1016/j.biocel.2013.12.009
El-Hattab AW, Adesina AM, Jones J, Scaglia F (2015) MELAS syndrome: Clinical manifestations, pathogenesis, and treatment options. Mol Genet Metab 116:4–12. https://doi.org/10.1016/j.ymgme.2015.06.004
Emsley HCA, Tyrrell PJ (2002) Inflammation and infection in clinical stroke. J Cereb Blood Flow Metab 22:1399–1419. https://doi.org/10.1097/01.WCB.0000037880.62590.28
Finsterer J, Aliyev R (2020) Metabolic stroke or stroke-like lesion: Peculiarities of a phenomenon. J Neurol Sci 412:116726. https://doi.org/10.1016/j.jns.2020.116726
Gargalas S, Weeks R, Khan-Bourne N et al (2017) Incidence and outcome of functional stroke mimics admitted to a hyperacute stroke unit. J Neurol Neurosurg Psychiatry 88:2–6. https://doi.org/10.1136/jnnp-2015-311114
Glover EI, Martin J, Maher A et al (2010) A randomized trial of coenzyme Q10 in mitochondrial disorders. Muscle Nerve 42:739–748. https://doi.org/10.1002/mus.21758
Gorman GS, Schaefer AM, Ng Y et al (2015) Prevalence of nuclear and mitochondrial DNA mutations related to adult mitochondrial disease. Ann Neurol 77:753–759. https://doi.org/10.1002/ana.24362
Goto YI, Nonaka I, Horai S (1990) A mutation in the tRNALeu(UUR) gene associated with the MELAS subgroup of mitochondrial encephalomyopathies. Nature 348:651–653
Gramegna LL, Cortesi I, Mitolo M, et al (2021) Major cerebral vessels involvement in patients with MELAS syndrome: Worth a scan? A systematic review. J Neuroradiol = J Neuroradiol. https://doi.org/10.1016/j.neurad.2021.02.002
Guy J, Feuer WJ, Davis JL et al (2017) Gene therapy for leber hereditary optic neuropathy: low- and medium-dose visual results. Ophthalmology 124:1621–1634. https://doi.org/10.1016/j.ophtha.2017.05.016
Haddy FJ, Vanhoutte PM, Feletou M (2006) Role of potassium in regulating blood flow and blood pressure. Am J Physiol - Regul Integr Comp Physiol 290:546–552. https://doi.org/10.1152/ajpregu.00491.2005
Hirano M, Pavlakis SG (1994) Mitochondrial myopathy, encephalopathy, lactic acidosis, and strokelike episodes (MELAS): Current concepts. J Child Neurol 9:4–13. https://doi.org/10.1177/088307389400900102
Homma K, Toda E, Osada H et al (2021) Taurine rescues mitochondria-related metabolic impairments in the patient-derived induced pluripotent stem cells and epithelial-mesenchymal transition in the retinal pigment epithelium. Redox Biol 41:101921. https://doi.org/10.1016/j.redox.2021.101921
Hongo Y, Kaneko J, Suga H et al (2019) A cluster of disseminated small cortical lesions in MELAS: its distinctive clinical and neuroimaging features. J Neurol 266:1459–1472. https://doi.org/10.1007/s00415-019-09283-3
Iadecola C (2017) The neurovascular unit coming of age: a journey through neurovascular coupling in health and disease. Neuron 96:17–42. https://doi.org/10.1016/j.neuron.2017.07.030
Iizuka T, Sakai F (2005) Pathogenesis of stroke-like episodes in melas: Analysis of neurovascular cellular mechanisms. Curr Neurovasc Res 2:29–45. https://doi.org/10.2174/1567202052773544
Iizuka T, Sakai F (2010) Pathophysiology of stroke-like episodes in MELAS: Neuron-astrocyte uncoupling in neuronal hyperexcitability. Future Neurol 5:61–83. https://doi.org/10.2217/fnl.09.71
Iizuka T, Sakai F, Suzuki N et al (2002) Neuronal hyperexcitability in stroke-like episodes of MELAS syndrome. Neurology 59:816–824. https://doi.org/10.1212/WNL.59.6.816
Iizuka T, Sakai F, Kan S, Suzuki N (2003) Slowly progressive spread of the stroke-like lesions in MELAS. Neurology 61:1238–1244. https://doi.org/10.1212/01.WNL.0000091888.26232.FE
Kim JT, Lee YJ, Lee YM et al (2009) Clinical characteristics of patients with non-specific and non-categorized mitochondrial diseases. Acta Paediatr Int J Paediatr 98:1825–1829. https://doi.org/10.1111/j.1651-2227.2009.01428.x
Koga Y, Akita Y, Nishioka J et al (2005) L-Arginine improves the symptoms of strokelike episodes in MELAS. Neurology 64:710–712. https://doi.org/10.1212/01.WNL.0000151976.60624.01
Koga Y, Povalko N, Inoue E et al (2018) Therapeutic regimen of l-arginine for MELAS: 9-year, prospective, multicenter, clinical research. J Neurol 265:2861–2874. https://doi.org/10.1007/s00415-018-9057-7
Kolb SJ, Costello F, Lee AG et al (2003) Distinguishing ischemic stroke from the stroke-like lesions of MELAS using apparent diffusion coefficient mapping. J Neurol Sci 216:11–15. https://doi.org/10.1016/S0022-510X(03)00218-1
Kotrys AV, Szczesny RJ (2019) Mitochondrial gene expression and beyond-novel aspects of cellular physiology. Cells 9. https://doi.org/10.3390/cells9010017
Kraya T, Neumann L, Paelecke-Habermann Y et al (2019) Cognitive impairment, clinical severity and MRI changes in MELAS syndrome. Mitochondrion 44:53–57. https://doi.org/10.1016/j.mito.2017.12.012
Majamaa K, Moilanen JS, Uimonen S et al (1998) Epidemiology of A3243G, the mutation for mitochondrial encephalomyopathy, lactic acidosis, and strokelike episodes: Prevalence of the mutation in an adult population. Am J Hum Genet 63:447–454. https://doi.org/10.1086/301959
McClelland G, Rodgers H, Flynn D, Price CI (2019) The frequency, characteristics and aetiology of stroke mimic presentations: A narrative review. Eur J Emerg Med 26:2–8. https://doi.org/10.1097/MEJ.0000000000000550
Montano V, Gruosso F, Simoncini C et al (2021) Clinical features of mtDNA-related syndromes in adulthood. Arch Biochem Biophys 697:108689. https://doi.org/10.1016/j.abb.2020.108689
Ng YS, Bindoff LA, Gorman GS et al (2019) Consensus-based statements for the management of mitochondrial stroke-like episodes. Wellcome Open Res 4:1–13. https://doi.org/10.12688/wellcomeopenres.15599.1
Ohsawa Y, Hagiwara H, Nishimatsu SI et al (2019) Taurine supplementation for prevention of stroke-like episodes in MELAS: A multicentre, open-label, 52-week phase III trial. J Neurol Neurosurg Psychiatry 90:529–536. https://doi.org/10.1136/jnnp-2018-317964
Pauls AD, Sandhu V, Young D et al (2020) High rate of hypertension in patients with m.3243A>G MELAS mutations and POLG variants. Mitochondrion 53:194–202. https://doi.org/10.1016/j.mito.2020.05.011
Pavlakis SG, Phillips PC, DiMauro S et al (1984) Mitochondrial myopathy, encephalopathy, lactic acidosis, and strokelike episodes: A distinctive clinical syndrome. Ann Neurol 16:481–488. https://doi.org/10.1002/ana.410160409
Pek NMQ, Phua QH, Ho BX et al (2019) Mitochondrial 3243A > G mutation confers pro-atherogenic and pro-inflammatory properties in MELAS iPS derived endothelial cells. Cell Death Dis 10. https://doi.org/10.1038/s41419-019-2036-9
Pellerin L, Bouzier-Sore A-K, Aubert A et al (2007) Activity-dependent regulation of energy metabolism by astrocytes: An update. Glia 55:1251–1262. https://doi.org/10.1002/glia.20528
Pickett SJ, Grady JP, Ng YS et al (2018) Phenotypic heterogeneity in m.3243A>G mitochondrial disease: The role of nuclear factors. Ann Clin Transl Neurol 5:333–345. https://doi.org/10.1002/acn3.532
Rahman J, Rahman S (2018) Mitochondrial medicine in the omics era. Lancet 391:2560–2574. https://doi.org/10.1016/S0140-6736(18)30727-X
Rikimaru M, Ohsawa Y, Wolf AM et al (2012) Taurine Ameliorates Impaired the Mitochondrial Function and Prevents Stroke-like Episodes in Patients with MELAS. Intern Med 51:3351–3357. https://doi.org/10.2169/internalmedicine.51.7529
Rønning OM, Thommessen B (2005) [Stroke: when the diagnosis is wrong]. Tidsskr den Nor Laegeforening 125:1655–1657. https://tidsskriftet.no/2005/06/aktuelt/nar-hjerneslagdiagnosen-er-feil. Accessed 22 Apr 2021
Schaefer AM, McFarland R, Blakely EL et al (2008) Prevalence of mitochondrial DNA disease in adults. Ann Neurol 63:35–39. https://doi.org/10.1002/ana.21217
Schaffer SW, Jong CJ, Warner D et al (2013) Taurine deficiency and MELAS are closely related syndromes. Adv Exp Med Biol 776:153–165. https://doi.org/10.1007/978-1-4614-6093-0_16
Shaw S, Kim P, Millett D (2012) Status epilepticus amauroticus revisited: Ictal and peri-ictal homonymous hemianopsia. Arch Neurol 69:1504–1507. https://doi.org/10.1001/archneurol.2012.317
Sproule DM, Kaufmann P (2008) Mitochondrial encephalopathy, lactic acidosis, and strokelike episodes: Basic concepts, clinical phenotype, and therapeutic management of MELAS syndrome. Ann N Y Acad Sci 1142:133–158. https://doi.org/10.1196/annals.1444.011
Stuehr DJ (1999) Mammalian nitric oxide synthases. Biochim Biophys Acta - Bioenerg 1411:217–230. https://doi.org/10.1016/S0005-2728(99)00016-X
Tetsuka S, Tagawa A, Ogawa T et al (2017) Importance of distinguishing between mitochondrial encephalomyopathy with elderly onset of stroke-like episodes and cerebral infarction. J Clin Med Res 9:812–819. https://doi.org/10.14740/jocmr3122w
Torres AG, Batlle E, Ribas de Pouplana L (2014) Role of tRNA modifications in human diseases. Trends Mol Med 20:306–314. https://doi.org/10.1016/j.molmed.2014.01.008
Tsujikawa K, Senda J, Yasui K et al (2016) Distinctive distribution of brain volume reductions in MELAS and mitochondrial DNA A3243G mutation carriers: A voxel-based morphometric study. Mitochondrion 30:229–235. https://doi.org/10.1016/j.mito.2016.08.011
Tsutomu S, Asuteka N, Takeo S (2011) Human mitochondrial diseases caused by lack of taurine modification in mitochondrial tRNAs. Wiley Interdiscip Rev RNA 2:376–386. https://doi.org/10.1002/wrna.65
Vilela P (2017) Acute stroke differential diagnosis: Stroke mimics. Eur J Radiol 96:133–144. https://doi.org/10.1016/j.ejrad.2017.05.008
Vos MH, Lipowski G, Lambry JC et al (2001) Dynamics of nitric oxide in the active site of reduced cytochrome c oxidase aa3. Biochemistry 40:7806–7811. https://doi.org/10.1021/bi010060x
Wang R, Li Y, Lin J et al (2020) Altered spontaneous brain activity at attack and remission stages in patients with mitochondrial encephalomyopathy, lactic acidosis and stroke-like episodes (MELAS): Beyond stroke-like lesions. Mitochondrion 54:49–56. https://doi.org/10.1016/j.mito.2020.07.002
Yasukawa T, Suzuki T, Suzuki T et al (2000) Modification defect at anticodon wobble nucleotide of mitochondrial tRNAs(Leu)(UUR) with pathogenic mutations of mitochondrial myopathy, encephalopathy, lactic acidosis, and stroke-like episodes. J Biol Chem 275:4251–4257. https://doi.org/10.1074/jbc.275.6.4251
Yasukawa T, Suzuki T, Ishii N et al (2001) Wobble modification defect in tRNA disturbs codon-anticodon interaction in a mitochondrial disease. EMBO J 20:4794–4802. https://doi.org/10.1093/emboj/20.17.4794
Yatsuga S, Povalko N, Nishioka J et al (2012) MELAS: A nationwide prospective cohort study of 96 patients in Japan. Biochim Biophys Acta - Gen Subj 1820:619–624. https://doi.org/10.1016/j.bbagen.2011.03.015
Acknowledgements
The authors would like to thank Enago (www.enago.jp) for the English language review.
Author information
Authors and Affiliations
Contributions
The concept and idea of the study was designed by ST. The manuscript was written by ST and TO. ST, RH and HK revised the manuscript and edited English. RH and HK critically revised the important content of the revision. RH and HK contributed immensely to the amendments. All authors have approved final article version.
Corresponding author
Ethics declarations
Ethical approval
Not applicable.
Informed consent to participate
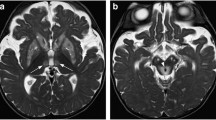
A written parental consent was obtained for publication of the MRI and CT material that is inserted in Figs. 1 and 3.
Consent for publication
All authors have read the manuscript and given their consent for publication.
Conflict of interest
Authors declare no conflicts of interest.
Additional information
Publisher's note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Highlights
• Lesion distribution is inconsistent with vascular territory.
• MELAS tends to present spontaneous reversibility of both neurological symptoms and stroke-like lesions, but both of those are prone to relapse later.
• The m.3243ANG mutation in the MT-TL1 gene encoding the mitochondrial tRNALeu(UUR) is commonly associated with MELAS.
• The mutation affects the taurine modification of tRNA and impairs protein synthesis.
• Neuronal hyperexcitability is a hypothesized cause of these episodes.
• Taurine administration is a new, practical, and therapeutic approach for MELAS.
• New gene therapies hold great promise for the future.
Supplementary Information
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Tetsuka, S., Ogawa, T., Hashimoto, R. et al. Clinical features, pathogenesis, and management of stroke-like episodes due to MELAS. Metab Brain Dis 36, 2181–2193 (2021). https://doi.org/10.1007/s11011-021-00772-x
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11011-021-00772-x