Summary
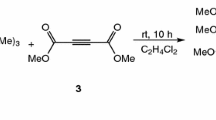
The adduct produced in the reaction between alkyl isocyanides and dialkyl acetylenedicarboxylates was trapped by 2,2,5-trimethyl-1,3-dioxane-4,6-dione (methyl Meldrum's acid), to afford highly functionalized ketenimines in good yields.
Similar content being viewed by others
References
Ugi, I. From isocyanides via four-component condensations to antibiotic syntheses, Angew. Chem. Int. Ed. Eng., 21 (1982) 810–812.
Ugi, I., Lohberger, S. and Karl, R. In: Comprehensive organic synthesis, Trost, B. M. and Fleming, I. (eds) vol 2, Pergamon Oxford, (1991) 1083–1106.
Domling, A. and Ugi, I., Multicomponent reactions with isocyanides, Angew. Chem. Int. Ed. Eng., 39 (2000) 3169–3210
Motoyoshiya, J., Teranishi, A., Mikoshiba, R., Yamamoto, I., Gotoh, H., Enda, J., Ohshiro, Y. and Agawa, T., C-phosphonoketenimines, characterization and synthetic application to heterocycles, J. Org. Chem., 45 (1980) 5385–5387.
Cristau, H.-J., Jouanin, I. and Taillefer, M., New synthesis of diphenyl-N-(substituted)ketenimines from diaminophosphonium diazaylides, J. Organometallic Chem., 584 (1999) 68–72.
Y. Ito, T. Hirao, N. Ohta and T. Saegusa, Synthesis of ketenimine via(N-alkylimino)acylpalladium complex intermediate, Tetrahedron Lett., 18 (1977) 1009–1012.
Ishida, M., Minami, T. and Agawa, T., Synthetic application of methyl (phenylthio) ketene synthesis of vicinal-substituted cyclopentene derivaties, J. Org. Chem., 44 (1979) 2067–2072.
Getzmann, R., Moller, M. H., Rodewald, U., Frohlich, R., Grehl, M. and Wurthwein, E.U., Metallated ketenimines: Deprotonation of n-isopropyl-diphenyl-ketenimine and subsequent trapping reaction with electrophiles. A theoretical and experimental study. Tetrahedron, 51 (1995) 3767–3786.
(a) Yavari, I., Davar-Panah, M., Heydari, M., Najafian, K. and Zonouzi, A. A facil route to highly functionalized ketenimines, Monatsh. Chem., 127 (1996) 963–966; (b) I. Yavari, I. and Nourmohammadian, F. A simple synthesis of highly functionalized ketenimines, J. Chem. Res., (S) (2000) 218–219; (c) Yavari, I., Djahaniani, H. and Nassiri, F. Coll. Czech. Chem. Commun., 69 (2004) 1499–1507; (d) Yavari, I., Djahaniani, H. and Nassiri, F.A simple synthesis of highly functionalized 1-azabutadienes and ketenimines, Monatsh. Chem., 135 (2004) 543–548; (e) Yavari, I., Nassiri, F. and Djahaniani, H. Synthesis and dynamic NMR study of ketenimines derived from tert-butyl isocyanide, alkyl 2-arylamino-2-oxo-acetates, and dialkyl acetylenedicarboxylates, Mol. Diversity, 8 (2004) 431–435; (f) Yavari, I., Nori-Shargh, D., and Fallah-Bagher-Shaidaii, H. Reaction of Meldrum's acid with alkyl isocyanides. synthesis of a novel class of furan derivatives, J. Chem. Res., (S) (1996) 146–147.
Nair, V. J., Rajesh, C., Vinod, A. U., Bindu, S., Sreekanth, A. R., Mathess, J. S. and Balagopal, L. Strategies for heterocyclic construction via novel multicomponent reactions based on isocyanides and nucleophilic carbenes, Acc. Chem. Res., 36 (2003) 899–909.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Yavari, I., Zare, H. & Mohtat, B. Three-Component Synthesis of Dialkyl 2-(Alkylimino-Methylene)3- (2,2,5-Trimethyl-4,6-Dioxo-1,3-Dioxan-5-Yl)-Succinates. Mol Divers 10, 247–250 (2006). https://doi.org/10.1007/s11030-005-9010-4
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11030-005-9010-4