Abstract
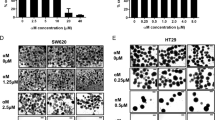
Low sensitivity of cancer stem cells toward regular cancer therapy strategies is an important issue in the field of cancer remedy. The concept of cancer stem cell elimination has been a topic of interest in the field of molecular medicine for a long time. At the current study, it was aimed to elevate the sensitivity of cancer stem-like cells toward radiotherapy by treating with Oct4–Sox2 complex decoy oligodeoxynucleotides (ODNs). After treating HT29 and HT29-ShE cells with Oct4–Sox2 complex decoy ODNs, and analyzing the cellular uptake and localization of decoys, treated cells and control groups were subjected to irradiation by fractionated 6MV X-ray with a final dose of 2 Gy. Thereafter, the influence of radiotherapy on ODNs treated groups and control group was investigated on cell viability, cell cycle, apoptosis, colonosphere formation and scratch assay. Cellular uptake and localization assays demonstrated that decoy ODNs can efficiently be transfected to the cells and reside in subcellular compartment, where they pose their action on gene regulation. Post radiotherapy analysis indicated statistical significance in decoy ODNs treated cells by means of lower cell viability, cell cycle arrest in G2/M phase, increased cellular apoptosis, and reduced cell motility. Also, formed colonospheres were smaller in size and fewer in numbers. Considering the role of Oct4, and Sox2 transcription factors in signaling pathways of preserving stemness and inducing reverse EMT, application of decoy strategy could increase the sensitivity of cancer cells toward irradiation, which has a potential to eliminate the cancerous cells from tumors and support cancer treatment.






Similar content being viewed by others
References
Ferlay J, Shin HR, Bray F, Forman D, Mathers C, Parkin DM (2010) Estimates of worldwide burden of cancer in 2008: GLOBOCAN 2008. Int J Cancer 127(12):2893–2917
Vo D, Julien L, Thorson A (2010) Current controversies in colon and rectal cancer. Minerva Chir 65(6):677–693
Bray F, Ferlay J, Soerjomataram I, Siegel RL, Torre LA, Jemal A (2018) Global cancer statistics 2018: GLOBOCAN estimates of incidence and mortality worldwide for 36 cancers in 185 countries. CA Cancer J Clin 68(6):394–424
Jordan CT, Guzman ML, Noble M (2006) Cancer stem cells. N Engl J Med 355(12):1253–1261
Jiang W, Peng J, Zhang Y, Cho W, Jin K (2012) The implications of cancer stem cells for cancer therapy. Int J Mol Sci 13(12):16636–16657
Reya T, Morrison SJ, Clarke MF, Weissman IL (2001) Stem cells, cancer, and cancer stem cells. Nature 414(6859):105
Clarke MF, Hass AT (2006) Cancer stem cells. Cell Biol. https://doi.org/10.1002/3527600906.mcb.200300130
Koury J, Zhong L, Hao J (2017) Targeting signaling pathways in cancer stem cells for cancer treatment. Stem Cells Int 2017:2925869
Moheb-Alian A, Forouzesh F, Rostami-Nejad M, Rostami K (2016) Mesenchymal stem cells as potential therapeutic approaches in celiac disease. Gastroenterol Hepatol Bed Bench 9(Suppl1):S1
Katoh Y, Katoh M (2008) Hedgehog signaling, epithelial-to-mesenchymal transition and miRNA. Int J Mol Med 22(3):271–275
Basu S, Haase G, Ben-Ze'ev A (2016) Wnt signaling in cancer stem cells and colon cancer metastasis. F1000Res. https://doi.org/10.12688/f1000research.7579.1
Fodde R, Smits R, Clevers H (2001) APC, signal transduction and genetic instability in colorectal cancer. Nat Rev Cancer 1(1):55
Morin PJ, Sparks AB, Korinek V, Barker N, Clevers H, Vogelstein B et al (1997) Activation of β-catenin-Tcf signaling in colon cancer by mutations in β-catenin or APC. Science 275(5307):1787–1790
Ivanova N, Dobrin R, Lu R, Kotenko I, Levorse J, DeCoste C et al (2006) Dissecting self-renewal in stem cells with RNA interference. Nature 442(7102):533
Boyer LA, Lee TI, Cole MF, Johnstone SE, Levine SS, Zucker JP et al (2005) Core transcriptional regulatory circuitry in human embryonic stem cells. Cell 122(6):947–956
Rad SMAH, Bamdad T, Sadeghizadeh M, Arefian E, Lotfinia M, Ghanipour M (2015) Transcription factor decoy against stem cells master regulators, Nanog and Oct-4: a possible approach for differentiation therapy. Tumor Biol 36(4):2621–2629
Chang C-J, Chien Y, Lu K-H, Chang S-C, Chou Y-C, Huang C-S et al (2011) Oct4-related cytokine effects regulate tumorigenic properties of colorectal cancer cells. Biochem Biophys Res Commun 415(2):245–251
Gazouli M, Roubelakis MG, Theodoropoulos GE, Papailiou J, Vaiopoulou A, Pappa KI et al (2012) OCT4 spliced variant OCT4B1 is expressed in human colorectal cancer. Mol Carcinog 51(2):165–173
Dai X, Ge J, Wang X, Qian X, Zhang C, Li X (2013) OCT4 regulates epithelial-mesenchymal transition and its knockdown inhibits colorectal cancer cell migration and invasion. Oncol Rep 29(1):155–160
Avilion AA, Nicolis SK, Pevny LH, Perez L, Vivian N, Lovell-Badge R (2003) Multipotent cell lineages in early mouse development depend on SOX2 function. Genes Dev 17(1):126–140
Takahashi K, Yamanaka S (2006) Induction of pluripotent stem cells from mouse embryonic and adult fibroblast cultures by defined factors. Cell 126(4):663–676
Zheng J, Xu L, Pan Y, Yu S, Wang H, Kennedy D et al (2017) Sox2 modulates motility and enhances progression of colorectal cancer via the Rho-ROCK signaling pathway. Oncotarget 8(58):98635
Campos B, Wan F, Farhadi M, Ernst A, Zeppernick F, Tagscherer KE et al (2010) Differentiation therapy exerts antitumor effects on stem-like glioma cells. Clin Cancer Res 16(10):2715–2728
de Thé H (2018) Differentiation therapy revisited. Nat Rev Cancer 18(2):117
Li Y, Zhou Y, Maris JM, Giaccia AJ, Ye J (2019) Acetate supplementation eliminates hypoxia mediated resistance to differentiation therapy in neuroblastoma cells. AACR. https://doi.org/10.1158/1538-7445
Wang J-s, Wang H-j, Qian H-l (2018) Biological effects of radiation on cancer cells. Military Med Res 5(1):20
Vilalta M, Rafat M, Graves EE (2016) Effects of radiation on metastasis and tumor cell migration. Cell Mol Life Sci 73(16):2999–3007
Moncharmont C, Levy A, Guy J-B, Falk AT, Guilbert M, Trone J-C et al (2014) Radiation-enhanced cell migration/invasion process: a review. Crit Rev Oncol Hematol 92(2):133–142
Chen Y, Zhu X, Zhang X, Liu B, Huang L (2010) Nanoparticles modified with tumor-targeting scFv deliver siRNA and miRNA for cancer therapy. Mol Ther 18(9):1650–1656
Lozano T, Soldevilla MM, Casares N, Villanueva H, Bendandi M, Lasarte JJ et al (2016) Targeting inhibition of Foxp3 by a CD28 2′-fluro oligonucleotide aptamer conjugated to P60-peptide enhances active cancer immunotherapy. Biomaterials 91:73–80
Rad SMAH, Langroudi L, Kouhkan F, Yazdani L, Koupaee AN, Asgharpour S et al (2015) Transcription factor decoy: a pre-transcriptional approach for gene downregulation purpose in cancer. Tumor Biol 36(7):4871–4881
Saltanatpour Z, Johari B, Alizadeh A, Lotfinia M, Majidzadeh AK, Nikbin B et al (2019) Enrichment of cancer stem-like cells by the induction of epithelial-mesenchymal transition using lentiviral vector carrying E-cadherin shRNA in HT29 cell line. J Cell Physiol. https://doi.org/10.1002/jcp.28855
Bigdelou Z, Mortazavi Y, Saltanatpour Z, Asadi Z, Kadivar M, Johari B (2020) Role of Oct4–Sox2 complex decoy oligodeoxynucleotides strategy on reverse epithelial to mesenchymal transition (EMT) induction in HT29-ShE encompassing enriched cancer stem-like cells. Mol Biol Rep 47(3):1859–1869
Terashima S, Hosokawa Y, Tsuruga E, Mariya Y, Nakamura T (2017) Impact of time interval and dose rate on cell survival following low-dose fractionated exposures. J Radiat Res 58(6):782–790
Liu S, Dontu G, Mantle ID, Patel S, Ahn N-s, Jackson KW et al (2006) Hedgehog signaling and Bmi-1 regulate self-renewal of normal and malignant human mammary stem cells. Cancer Res 66(12):6063–6071
Phi LTH, Wijaya YT, Sari IN, Yang YG, Lee YK, Kwon HY (2018) The anti-metastatic effect of ginsenoside Rb2 in colorectal cancer in an EGFR/SOX2-dependent manner. Cancer Med 7(11):5621–5631
Symonds RP, Foweraker K (2006) Principles of chemotherapy and radiotherapy. Current Obstet Gynaecol 16(2):100–106
Kaliberov SA, Buchsbaum DJ (2012) Cancer treatment with gene therapy and radiation therapy. Adv Cancer Res 115:221–263
Milone MC, O’Doherty U (2018) Clinical use of lentiviral vectors. Leukemia 32(7):1529–1541
Breckpot K, Aerts J, Thielemans K (2007) Lentiviral vectors for cancer immunotherapy: transforming infectious particles into therapeutics. Gene Ther 14(11):847–862
Liechtenstein T, Perez-Janices N, Escors D (2013) Lentiviral vectors for cancer immunotherapy and clinical applications. Cancers 5(3):815–837
Wang S-L, Yao H-H, Qin Z-H (2009) Strategies for short hairpin RNA delivery in cancer gene therapy. Expert Opin Biol Ther 9(11):1357–1368
Huang W-S, Wang J-P, Wang T, Fang J-Y, Lan P, Ma J-P (2007) ShRNA-mediated gene silencing of β-catenin inhibits growth of human colon cancer cells. World J Gastroenterol 13(48):6581
Guo W, Chen W, Yu W, Huang W, Deng W (2013) Small interfering RNA-based molecular therapy of cancers. Chin J Cancer 32(9):488
Chalbatani GM, Dana H, Gharagouzloo E, Grijalvo S, Eritja R, Logsdon CD et al (2019) Small interfering RNAs (siRNAs) in cancer therapy: a nano-based approach. Int J Nanomed 14:3111
Sachdeva M, Sachdeva N, Pal M, Gupta N, Khan I, Majumdar M et al (2015) CRISPR/Cas9: molecular tool for gene therapy to target genome and epigenome in the treatment of lung cancer. Cancer Gene Ther 22(11):509
Rahmati M, Johari B, Kadivar M, Rismani E, Mortazavi Y (2020) Suppressing the metastatic properties of the breast cancer cells using STAT3 decoy oligodeoxynucleotides: a promising approach for eradication of cancer cells by differentiation therapy. J Cell Physiol 235(6):5429–5444
Talebi A, Kianersi K, Beiraghdar M (2015) Comparison of gene expression of SOX2 and OCT4 in normal tissue, polyps, and colon adenocarcinoma using immunohistochemical staining. Adv Biomed Res 4:234
Mann MJ, Whittemore AD, Donaldson MC, Belkin M, Conte MS, Polak JF et al (1999) Ex-vivo gene therapy of human vascular bypass grafts with E2F decoy: the PREVENT single-centre, randomised, controlled trial. Lancet 354(9189):1493–1498
Sen M, Thomas SM, Kim S, Yeh JI, Ferris RL, Johnson JT et al (2012) First-in-human trial of a STAT3 decoy oligonucleotide in head and neck tumors: implications for cancer therapy. Cancer Discov 2(8):694–705
Egashira K, Ji S, Ito H, Aoki M, Isobe M, Morishita R (2008) Long-term follow up of initial clinical cases with NF-κB decoy oligodeoxynucleotide transfection at the site of coronary stenting. J Gene Med 10(7):805–809
Johari B, Zargan J (2017) Simultaneous targeted inhibition of Sox2-Oct4 transcription factors using decoy oligodeoxynucleotides to repress stemness properties in mouse embryonic stem cells. Cell Biol Int 41(12):1335–1344
Feng R, Zhou S, Liu Y, Song D, Luan Z, Dai X et al (2013) Sox2 protects neural stem cells from apoptosis via up-regulating survivin expression. Biochem J 450(3):459–468
Zhang Z, Zhu Y, Lai Y, Wu X, Feng Z, Yu Y et al (2013) Follicle-stimulating hormone inhibits apoptosis in ovarian cancer cells by regulating the OCT4 stem cell signaling pathway. Int J Oncol 43(4):1194–1204
Zhang X, Zhang J, Wang L, Wei H, Tian Z (2007) Therapeutic effects of STAT3 decoy oligodeoxynucleotide on human lung cancer in xenograft mice. BMC Cancer 7(1):149
Zhang S-Y, Park K-W, Oh S, Cho H-J, Cho H-J, Park J-S et al (2005) NF-κB decoy potentiates the effects of radiation on vascular smooth muscle cells by enhancing apoptosis. Exp Mol Med 37(1):18–26
Park SI, Park S-J, Lee J, Kim HE, Park SJ, Sohn J-W et al (2016) Inhibition of cyclic AMP response element-directed transcription by decoy oligonucleotides enhances tumor-specific radiosensitivity. Biochem Biophys Res Commun 469(3):363–369
Gambari R (2008) Targeting transcription factors with decoy oligonucleotides: modulation of the expression of genes involved in chemotherapy resistance of tumor cells. In: Bonavida B (ed) Sensitization of Cancer Cells for Chemo/Immuno/Radio-therapy: Cancer Drug Discovery and Development. Springer, Heidelberg, pp 175–187
Zhang X, Liu P, Zhang B, Wang A, Yang M (2010) Role of STAT3 decoy oligodeoxynucleotides on cell invasion and chemosensitivity in human epithelial ovarian cancer cells. Cancer Genet Cytogenet 197(1):46–53
Dritschilo A, Huang CH, Rudin CM, Marshall J, Collins B, Dul JL et al (2006) Phase I study of liposome-encapsulated c-raf antisense oligodeoxyribonucleotide infusion in combination with radiation therapy in patients with advanced malignancies. Clin Cancer Res 12(4):1251–1259
Acknowledgements
This study was supported by Zanjan University of Medical Sciences (Grant Number A-12-1357-1, Ethical Code ZUMS.REC.1378.379).
Author information
Authors and Affiliations
Contributions
Conception and design the study: BJ. Collection, assembly of data and manuscript writing: BJ, HR, MM, ZS, ZT, YM. Final approval of manuscript: BJ and LN.
Corresponding author
Ethics declarations
Conflict of interest
Leila Nasehi received the Grant from Zanjan University of Medical Sciences. All the authors declare no financial or commercial conflict of interests that could negatively influence the study.
Research Involving Human and Animal Participants
No human or animal was involved in this study.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Electronic supplementary material
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Johari, B., Rezaeejam, H., Moradi, M. et al. Increasing the colon cancer cells sensitivity toward radiation therapy via application of Oct4–Sox2 complex decoy oligodeoxynucleotides. Mol Biol Rep 47, 6793–6805 (2020). https://doi.org/10.1007/s11033-020-05737-4
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11033-020-05737-4